Professional Documents
Culture Documents
Flow Through Packed Beds and Fluidized Beds: R. Shankar Subramanian
Uploaded by
Tapajyoti GhoshOriginal Title
Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
Flow Through Packed Beds and Fluidized Beds: R. Shankar Subramanian
Uploaded by
Tapajyoti GhoshCopyright:
Available Formats
Flow through Packed Beds and Fluidized Beds
R. Shankar Subramanian
Chemical engineering operations commonly involve the use of packed and fluidized beds. These
are devices in which a large surface area for contact between a liquid and a gas (absorption,
distillation) or a solid and a gas or liquid (adsorption, catalysis) is obtained for achieving rapid
mass and heat transfer, and particularly in the case of fluidized beds, catalytic chemical
reactions. You will find a good deal of information about flow through packed and fluidized
beds in the book by McCabe, Smith, and Harriott (2001) and Perrys Handbook (1997). Here,
only a brief summary is given. First, let us consider flow through a packed bed.
Packed Beds
A typical packed bed is a cylindrical column that is filled with a suitable packing material. You
can learn about different types of packing materials from Perrys Handbook. The liquid is
distributed as uniformly as possible at the top of the column and flows downward, wetting the
packing material. A gas is admitted at the bottom, and flows upward, contacting the liquid in a
countercurrent fashion. An example of a packed bed is an absorber. Here, the gas contains some
carrier species that is insoluble in the liquid (such as air) and a soluble species such as carbon
dioxide or ammonia. The soluble species is absorbed in the liquid, and the lean gas leaves the
column at the top. The liquid rich in the soluble species is taken out at the bottom.
From a fluid mechanical perspective, the most important issue is that of the pressure drop
required for the liquid or the gas to flow through the column at a specified flow rate. To calculate
this quantity we rely on a friction factor correlation attributed to Ergun. Other fluid mechanical
issues involve the proper distribution of the liquid across the cross-section, and developing
models of the velocity profile in the liquid film around a piece of packing material so that
heat/mass transfer calculations can be made. Design of packing materials to achieve uniform
distribution of the fluid across the cross-section throughout the column is an important subject as
well. Here, we only focus on the pressure drop issue.
The Ergun equation that is commonly employed is given below.
150
1.75
p
p
f
Re
= +
Here, the friction factor
p
f for the packed bed, and the Reynolds number
p
Re , are defined as
follows.
1
3
2
1
p
p
s
D
p
f
L V
=
( ) 1
p s
p
D V
Re
=
The various symbols appearing in the above equations are defined as follows.
: p Pressure Drop
: L Length of the Bed
:
p
D Equivalent spherical diameter of the particle defined by
Volume of the particle
6
Surface area of the particle
p
D =
: Density of the fluid
: Dynamic viscosity of the fluid
:
s
V Superficial velocity (
s
Q
A
= V where Q is the volumetric flow rate of the fluid and is the
cross-sectional area of the bed)
A
: Void fraction of the bed ( is the ratio of the void volume to the total volume of the bed)
Sometimes, we may use the concept of the interstitial velocity V , which is related to the
superficial velocity by
i
s
i
V
V
= . The interstitial velocity is the average velocity that prevails in
the pores of the column.
Two simpler results, each obtained by ignoring one or the other term in the Ergun equation also
are in use. One is the Kozeny-Carman equation, used for flow under very viscous conditions.
150
p
p
f
Re
= , 1
p
Re
The other is the Burke-Plummer equation, used when viscous effects are not as important as
inertia.
1.75
p
f = , 1, 000
p
Re
It is suggested that the student simply use the Ergun equation. There is no need to use these
other two approximate results, even though they continue to be reported in textbooks.
2
Fluidized Beds
A fluidized bed is a packed bed through which fluid flows at such a high velocity that the bed is
loosened and the particle-fluid mixture behaves as though it is a fluid. Thus, when a bed of
particles is fluidized, the entire bed can be transported like a fluid, if desired. Both gas and liquid
flows can be used to fluidize a bed of particles. The most common reason for fluidizing a bed is
to obtain vigorous agitation of the solids in contact with the fluid, leading to excellent contact of
the solid and the fluid and the solid and the wall. This means that nearly uniform temperatures
can be maintained even in highly exothermic reaction situations where the particles are used to
catalyze a reaction in the species contained in the fluid. In fact, fluidized beds were used in
catalytic cracking in the petroleum industry in the past. The catalyst is suspended in the fluid by
fluidizing a bed of catalytic particles so that intimate contact can be achieved between the
particles and the fluid. Nowadays, you will find fluidized beds used in catalyst regeneration,
solid-gas reactors, combustion of coal, roasting of ores, drying, and gas adsorption operations.
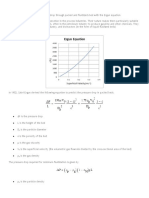
Superficial velocity
Superficial Velocity
V
f
: Minimum Fluidization
Velocity
D
C
C
D
B
A
A
B
E
E
O
O
Pressure
Drop
Bed
Height
3
First, we consider the behavior of a bed of particles when the upward superficial fluid velocity is
gradually increased from zero past the point of fluidization, and back down to zero. Reference is
made to the figure on page 3.
At first, when there is no flow, the pressure drop zero, and the bed has a certain height. As we
proceed along the right arrow in the direction of increasing superficial velocity, tracing the path
ABCD, at first, the pressure drop gradually increases while the bed height remains fixed. This is
a region where the Ergun equation for a packed bed can be used to relate the pressure drop to the
velocity. When the point B is reached, the bed starts expanding in height while the pressure drop
levels off and no longer increases as the superficial velocity is increased. This is when the
upward force exerted by the fluid on the particles is sufficient to balance the net weight of the
bed and the particles begin to separate from each other and float in the fluid. As the velocity is
increased further, the bed continues to expand in height, but the pressure drop stays constant. It
is possible to reach large superficial velocities without having the particles carried out with the
fluid at the exit. This is because the settling velocities of the particles are typically much larger
than the largest superficial velocities used.
Now, if we trace our path backward, gradually decreasing the superficial velocity, in the
direction of the reverse arrows in the figure, we find that the behavior of the bed follows the
curves DCE. At first, the pressure drop stays fixed while the bed settles back down, and then
begins to decrease when the point C is reached. The bed height no longer decreases while the
pressure drop follows the curve CEO. A bed of particles, left alone for a sufficient length of time,
becomes consolidated, but it is loosened when it is fluidized. After fluidization, it settles back
into a more loosely packed state; this is why the constant bed height on the return loop is larger
than the bed height in the initial state. If we now repeat the experiment by increasing the
superficial velocity from zero, well follow the set of curves ECD in both directions. Because of
this reason, we define the velocity at the point C in the figure as the minimum fluidization
velocity V . We can calculate it by balancing the net weight of the bed against the upward force
exerted on the bed, namely the pressure drop across the bed
f
p multiplied by the cross-sectional
area of the bed . In doing this balance, we ignore the small frictional force exerted on the wall
of the column by the flowing fluid.
A
Upward force on the bed p A =
If the height of the bed at this point is and the void fraction is L , we can write
Volume of particles (1 ) AL =
If the acceleration due to gravity is , the net gravitational force on the particles (net weight) is g
Net Weight of the particles ( )( )
1
p f
ALg =
4
Balancing the two yields
( )( )
1
p f
p Lg =
By using an expression relating p to the superficial velocity, which is the fluidization velocity
at this point, we can obtain a result for the latter.
Typically, for a bed of small particles ( 0.1
p
D mm), the flow conditions at this stage are such
that the Reynolds number is relatively small ( ) 10 Re so that we can use the Kozeny-Carman
Equation, applicable to the viscous flow regime, for establishing the point of onset of
fluidization. This yields
( )
2
3
150 1
p f p
f
gD
V
When the superficial velocity
s
V is equal to V , we refer to the state of the bed as one of
incipient fluidization. The void fraction
f
at this state depends upon the material, shape, and
size of the particles. For nearly spherical particles, McCabe, Smith, and Harriott (2001) suggest
that lies in the range 0.40 , increasing a bit with particle size. 0.45
For large particles ( mm), inertial effects are important, and the full Ergun equation must
be used to determine V . When in doubt, use the Ergun equation instead of a simplified version
of it.
1
p
D
f
Now, we consider the condition we must impose on the superficial velocity so that particles are
not carried out with the fluid at the exit. This would occur if the superficial velocity is equal to
the settling velocity of the particles. Restricting attention to small particles so that Stokes law
can be used to calculate their settling velocity, we can write
( )
2
settling
18
p f
gD
V
=
p
If we now use the result for the minimum fluidization velocity for the case of small particles,
given above, we see that the ratio
3
25 1
3
settling
f
V
V
=
For lying in the range 0.40 , this yields a ratio ranging from . McCabe et al.
suggest that it is common to operate fluidized beds at velocities as high as 30 , and values as
large as 100 are used on occasion. Recognizing that not all particles are of the same size and
that is only an average size, we see that fine particles are likely to be carried out with the
0.45 78 50
V
f
f
V
p
D
5
6
exiting fluid in such a situation. They can be recovered by filters or cyclone separators and
returned, in order to obtain the benefits of operating a bed at such large superficial velocities.
Fluidization can be broadly classified into particulate fluidization or bubbling fluidization.
Particulate fluidization occurs in liquids. As the velocity of the liquid is increased past the
minimum fluidization velocity, the bed expands uniformly, and uniform conditions prevail in the
liquid solid mixture. In contrast, bubbling fluidization occurs in gas-fluidized beds. Here, when
the bed is fluidized, large pockets of gas, free of particles, are seen to rise through the bed.
Where there are particles, the bed void fraction is approximately at the value that prevails at the
point of incipient fluidization. The bubbles grow until they fill the cross-section, and then
successive bubbles move up the column, a condition known as slugging.
The above classification should not be interpreted rigidly. Sometimes, very dense particles in a
liquid can show bubbling and gases at high pressure when flowing through beds of fine
particles, can give rise to particulate fluidization. Usually, this occurs at lower velocities, and at
higher velocities, the bed shows bubbling.
References
W.E. McCabe, J.C. Smith, and P.Harriott 2001. Unit Operations of Chemical Engineering,
McGraw Hill, New York.
Perrys Chemical Engineers Handbook, 7
th
Edition. 1997 (Ed: R.H. Perry, D.W. Green, and J.O.
Maloney), McGraw-Hill, New York.
You might also like
- Experiment No.6 Flow Through Fluidized Bed Background and TheoryDocument4 pagesExperiment No.6 Flow Through Fluidized Bed Background and TheoryAayush AgarwalNo ratings yet
- Flow Through Packed Fixed and Fluidized Beds PDFDocument7 pagesFlow Through Packed Fixed and Fluidized Beds PDFelisya diantyNo ratings yet
- Convective Mass TransferDocument46 pagesConvective Mass TransferSandra Enn BahintingNo ratings yet
- Fluid Flow Thru A Bed of Particles: Che 317E Particle TechnologyDocument19 pagesFluid Flow Thru A Bed of Particles: Che 317E Particle TechnologyJulie Anne Cristales100% (1)
- Transport Process (Fluidization Beds)Document6 pagesTransport Process (Fluidization Beds)zujishorok7No ratings yet
- Drag CoefficientDocument5 pagesDrag CoefficientRaghav MainiNo ratings yet
- Fixed Bed and Fluidized BedDocument33 pagesFixed Bed and Fluidized Bedشاكر العاقلNo ratings yet
- Flow in Packed BedsDocument8 pagesFlow in Packed BedsGerry Lou QuilesNo ratings yet
- Fludized BedDocument11 pagesFludized BedHaiqal AzizNo ratings yet
- Exp 4-Fuid MixingDocument26 pagesExp 4-Fuid MixingNawal DaBomb100% (1)
- Drag of A SphereDocument4 pagesDrag of A SphereJean-Franz Baschube100% (1)
- Ergun Equation ValidationDocument18 pagesErgun Equation ValidationAtikur Rahman100% (1)
- CONTINUOUS DistillationDocument5 pagesCONTINUOUS DistillationNaseer SattarNo ratings yet
- Suspension Od Solid Particles (Revised Report)Document7 pagesSuspension Od Solid Particles (Revised Report)michsantosNo ratings yet
- Fluidization: Chemical Engneering PracticeDocument24 pagesFluidization: Chemical Engneering PracticeFarhan M JafrI100% (2)
- Quantum Heat Transfer PDFDocument7 pagesQuantum Heat Transfer PDFPhilip John FuentesNo ratings yet
- Vapor Liquid EquilibriumDocument28 pagesVapor Liquid EquilibriumKhloud MadihNo ratings yet
- Fluid Lab Compiled v2Document26 pagesFluid Lab Compiled v2Kagendren AyanNo ratings yet
- Chemical Engineering in Practise 3: Fluidised BedsDocument12 pagesChemical Engineering in Practise 3: Fluidised BedsYaz Swedan100% (1)
- Pressure Drop For Flow Through Packed Beds PDFDocument19 pagesPressure Drop For Flow Through Packed Beds PDFrohl55No ratings yet
- Flow Through Granular Beds and Packed Beds (Revised 111114)Document38 pagesFlow Through Granular Beds and Packed Beds (Revised 111114)asaya675456No ratings yet
- Boiling Heat TransferDocument26 pagesBoiling Heat TransfercmegmhiNo ratings yet
- Series and Parallel PumpsDocument11 pagesSeries and Parallel PumpsKevin Devastian100% (1)
- Ergun EquationDocument2 pagesErgun EquationJian of LiuNo ratings yet
- SedimentationDocument7 pagesSedimentationgrkhari1100% (2)
- Fluidized Bed ExperimentDocument2 pagesFluidized Bed Experimentdrami94100% (3)
- Diffusion Coefficient Full Report TiqaDocument19 pagesDiffusion Coefficient Full Report TiqaprmzNo ratings yet
- Experimental Analysis of Fluidized Bed Freeze DryingDocument7 pagesExperimental Analysis of Fluidized Bed Freeze DryingMohamadMostafaviNo ratings yet
- Flow Through Packed Lab Report For Chemical EngineeringDocument17 pagesFlow Through Packed Lab Report For Chemical EngineeringManu Kumar60% (5)
- Application of Ergun Equation To Computation of Criticalication of Ergun Equation To Computation of CriticalDocument21 pagesApplication of Ergun Equation To Computation of Criticalication of Ergun Equation To Computation of CriticalNovianto J. SunarnoNo ratings yet
- Heat-Mass Transfer in A Tubular Chemical Reactor: Rehena NasrinDocument11 pagesHeat-Mass Transfer in A Tubular Chemical Reactor: Rehena NasrinNaveed KhanNo ratings yet
- CHAPTER 2 Single Particle in A Fluid - 724666540Document6 pagesCHAPTER 2 Single Particle in A Fluid - 724666540Donna Mae Ramos GalaezNo ratings yet
- Gas Diffusion UnitDocument20 pagesGas Diffusion Unitsolehah misniNo ratings yet
- FluidizationDocument3 pagesFluidizationUmairAhmedNo ratings yet
- Drying: Merry Jessah S. TorresDocument6 pagesDrying: Merry Jessah S. TorresFrancis Val FranciscoNo ratings yet
- Chapter 6a Unsteady-State DiffusionDocument13 pagesChapter 6a Unsteady-State DiffusionrushdiNo ratings yet
- Heat 4e Chap11 LectureDocument32 pagesHeat 4e Chap11 LectureSalim ChohanNo ratings yet
- ERT 313 Bioseparation Engineering Gas Absorption: Prepared By: Pn. Hairul Nazirah Abdul HalimDocument57 pagesERT 313 Bioseparation Engineering Gas Absorption: Prepared By: Pn. Hairul Nazirah Abdul HalimVenkateswara RamakrishnanNo ratings yet
- Fluidized Bed A14Document8 pagesFluidized Bed A14lovelygirl_256No ratings yet
- Diffusivity of Liquid Into LiquidDocument8 pagesDiffusivity of Liquid Into LiquidZahraa GhanemNo ratings yet
- Experiment: Screw Conveyor: Course Number and NameDocument10 pagesExperiment: Screw Conveyor: Course Number and NameMaheshree GohilNo ratings yet
- Chapter 2 - Fundamentals of Fixed-Bed ReactorsDocument21 pagesChapter 2 - Fundamentals of Fixed-Bed Reactorskim haksongNo ratings yet
- Heat Transfer Coefficient PDFDocument57 pagesHeat Transfer Coefficient PDFAnmol Preet SinghNo ratings yet
- Size ReductionDocument16 pagesSize ReductionRA Memije100% (1)
- Transport Phenomena 1Document8 pagesTransport Phenomena 1Al- DhaheriNo ratings yet
- Interphase Mass TransferDocument10 pagesInterphase Mass Transferanilkumar995472No ratings yet
- Unit Operation Laboratory 2 (CCB 3062)Document7 pagesUnit Operation Laboratory 2 (CCB 3062)Carl Erickson100% (1)
- AgitationDocument10 pagesAgitationKenneth Jireh Taboco100% (2)
- Natural Convection Heat Transfer On A Vertical C 2014 International JournalDocument8 pagesNatural Convection Heat Transfer On A Vertical C 2014 International JournalAnonymous vVsidh3nNo ratings yet
- Heat Transfer EnhancementDocument46 pagesHeat Transfer EnhancementShreyas SrinivasanNo ratings yet
- Prelab 3 Response of First and Second Order SystemsDocument7 pagesPrelab 3 Response of First and Second Order SystemsDean Joyce AlborotoNo ratings yet
- LESSON 8 - Shell Momentum BalanceDocument66 pagesLESSON 8 - Shell Momentum BalancePamela MendozaNo ratings yet
- One-Dimensional Compressible Flow: Thermodynamics and Fluid Mechanics SeriesFrom EverandOne-Dimensional Compressible Flow: Thermodynamics and Fluid Mechanics SeriesNo ratings yet
- Bubble Wake Dynamics in Liquids and Liquid-Solid SuspensionsFrom EverandBubble Wake Dynamics in Liquids and Liquid-Solid SuspensionsNo ratings yet
- PSOC Mid 1 QuestionsDocument7 pagesPSOC Mid 1 Questions3nathNo ratings yet
- Robotic SensorsDocument30 pagesRobotic Sensorsmanoj50% (4)
- The Ships Electrical NetworkDocument6 pagesThe Ships Electrical Networktkdrt2166No ratings yet
- Motor BalancingDocument2 pagesMotor BalancingAhmed MenshawyNo ratings yet
- JHS Physics - BOW - MELC PDFDocument12 pagesJHS Physics - BOW - MELC PDFJohnry Guzon ColmenaresNo ratings yet
- Surge CalculationsDocument21 pagesSurge Calculationsger80100% (4)
- Oil Filter Unit, Type OLU, For On-Load Tap-Changers: ManualDocument40 pagesOil Filter Unit, Type OLU, For On-Load Tap-Changers: ManualViệt Hùng Nghiêm100% (1)
- Natural Ventilation Engineering GuideDocument40 pagesNatural Ventilation Engineering GuidePrestoneK100% (1)
- Instruction Manual and Parts List For Compressed Air Filters DDX - DDXP - PDX - PDXP - QDXDocument88 pagesInstruction Manual and Parts List For Compressed Air Filters DDX - DDXP - PDX - PDXP - QDXAlonso GomezNo ratings yet
- DT-7435/DT-7435C: FeaturesDocument2 pagesDT-7435/DT-7435C: FeaturesZamfir GeoNo ratings yet
- Quincy QGV Series: Rotary Screw Air Compressors - 75-200 HPDocument8 pagesQuincy QGV Series: Rotary Screw Air Compressors - 75-200 HPRUN GONo ratings yet
- Kobelco PC 200: Selected DimensionsDocument6 pagesKobelco PC 200: Selected DimensionsGilbert AdityaNo ratings yet
- Miang Besar Coal Terminal: Calculation For Diesel Oil Transfer PumpDocument1 pageMiang Besar Coal Terminal: Calculation For Diesel Oil Transfer PumpPaijo TejoNo ratings yet
- Managing Well CasingDocument18 pagesManaging Well CasingElena Herrera VasquezNo ratings yet
- Parichha Pending Liability 30.09.2016Document43 pagesParichha Pending Liability 30.09.2016Vinay Vinay Ranjan JaiswalNo ratings yet
- Assignment2 NaturalresourcesDocument2 pagesAssignment2 NaturalresourcesAlexandra CarataoNo ratings yet
- CA 305 Air ConditionerDocument15 pagesCA 305 Air ConditionerRedza RashidiNo ratings yet
- I. Choose The Best Answer To The Following Questions!Document8 pagesI. Choose The Best Answer To The Following Questions!Rudi Hendra GunawanNo ratings yet
- Ha Red A 787986240Document30 pagesHa Red A 787986240tarun_aggarwaly5712No ratings yet
- Drag Force On A SphereDocument10 pagesDrag Force On A SphereCameron BrackenNo ratings yet
- Noise Pollution in PalawanDocument30 pagesNoise Pollution in Palawanĸěřbў mŏňNo ratings yet
- Testing Electrochemical CapacitorsDocument8 pagesTesting Electrochemical CapacitorsOlgalycosNo ratings yet
- The Induction Motor - A Short Circuited Rotating Transformer - A Comparative Analysis C1503021014Document5 pagesThe Induction Motor - A Short Circuited Rotating Transformer - A Comparative Analysis C1503021014mkdir911No ratings yet
- Steam Turbine DesignDocument27 pagesSteam Turbine DesignEkta Gupta50% (2)
- Admix Datasheet Dynashear WEBDocument2 pagesAdmix Datasheet Dynashear WEBVishal SalveNo ratings yet
- Finer Orta Oil PaperDocument11 pagesFiner Orta Oil PaperJuanito Sin TierraNo ratings yet
- 30 Equipment Checklists in One Excel WorkbookDocument56 pages30 Equipment Checklists in One Excel WorkbookBorislav VulićNo ratings yet
- StandardDocument2 pagesStandardjopacameNo ratings yet
- 368cffbee6f1Document68 pages368cffbee6f1anilr008No ratings yet
- Electrical Machinery 7th EditionDocument7 pagesElectrical Machinery 7th EditionGreetings Hooman0% (1)