Professional Documents
Culture Documents
Liquid Liquid Extraction Example
Liquid Liquid Extraction Example
Uploaded by
brokenpda0 ratings0% found this document useful (0 votes)
70 views2 pagesThis document provides the solution to a homework problem involving calculating the molarity of a solution. The key steps shown are: dividing the moles of solute by the liters of solution to calculate the molarity as 6.67x10^-7 M, and confirming the significant figures by rounding to 0.308 M with three significant figures. The final concentration is reported as 0.3 M.
Original Description:
Example of liquid liquid extraction (water and MiK) in crossflow
Original Title
Liquid Liquid Extraction example
Copyright
© © All Rights Reserved
Available Formats
PDF, TXT or read online from Scribd
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentThis document provides the solution to a homework problem involving calculating the molarity of a solution. The key steps shown are: dividing the moles of solute by the liters of solution to calculate the molarity as 6.67x10^-7 M, and confirming the significant figures by rounding to 0.308 M with three significant figures. The final concentration is reported as 0.3 M.
Copyright:
© All Rights Reserved
Available Formats
Download as PDF, TXT or read online from Scribd
0 ratings0% found this document useful (0 votes)
70 views2 pagesLiquid Liquid Extraction Example
Liquid Liquid Extraction Example
Uploaded by
brokenpdaThis document provides the solution to a homework problem involving calculating the molarity of a solution. The key steps shown are: dividing the moles of solute by the liters of solution to calculate the molarity as 6.67x10^-7 M, and confirming the significant figures by rounding to 0.308 M with three significant figures. The final concentration is reported as 0.3 M.
Copyright:
© All Rights Reserved
Available Formats
Download as PDF, TXT or read online from Scribd
You are on page 1of 2
HO #7 Solution
6.67
=-
6.67x10^-7
0.308
0.308
\\0.3
0.3 m
0.308
0.308
You might also like
- Principles of Foundation Engineering SI Edition 8th Edition Das Solutions Manual 1Document20 pagesPrinciples of Foundation Engineering SI Edition 8th Edition Das Solutions Manual 1barbara100% (54)
- Laboratorio MezclasDocument7 pagesLaboratorio MezclasDuvan GarnicaNo ratings yet
- Example 4 - FlexibilityDocument6 pagesExample 4 - Flexibilityarokia samyNo ratings yet
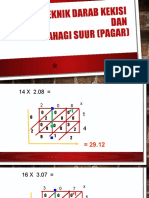
- Teknik Darab Kekisi Dan Bahagi SuurDocument5 pagesTeknik Darab Kekisi Dan Bahagi SuurHarliza Mohamad ArifinNo ratings yet
- 8-Dewey - Phil IriDocument31 pages8-Dewey - Phil IriAllen De La RosaNo ratings yet
- 9-Lotus-Phil IriDocument31 pages9-Lotus-Phil IriAllen De La RosaNo ratings yet
- Descubra A Capacidade Ideal para o Seu Ar-CondicionadoDocument8 pagesDescubra A Capacidade Ideal para o Seu Ar-CondicionadoLeti De RezendeNo ratings yet
- Percentage Conversions by KartikDocument3 pagesPercentage Conversions by KartikanmolsachdevaextraNo ratings yet
- Pembesian Lentur DG Gaya Normal Balok PersegiDocument5 pagesPembesian Lentur DG Gaya Normal Balok PersegiRaditiya PuteraNo ratings yet
- D. Hasil Pengamatan Bahan MR Massa (GR) Konsentrasi (M) PH Warna LarutanDocument2 pagesD. Hasil Pengamatan Bahan MR Massa (GR) Konsentrasi (M) PH Warna LarutanLady Wahyu HapsariNo ratings yet
- Plane TrusDocument31 pagesPlane TrusnurulNo ratings yet
- Luna Duo-Tec 40 GaDocument2 pagesLuna Duo-Tec 40 GaVictor Jerobam Poque RocaNo ratings yet
- 09 Analisis Struktur 02Document2 pages09 Analisis Struktur 02malfoy kdracoNo ratings yet
- Manlab Kelompok6 Xiii Ka 2Document5 pagesManlab Kelompok6 Xiii Ka 2Anissa mdNo ratings yet
- 2B - J310200061 - Garnis Annisa A.M - Analisis Kadar Gula ReduksiDocument3 pages2B - J310200061 - Garnis Annisa A.M - Analisis Kadar Gula ReduksiSaminur FauzanNo ratings yet
- Principles of Foundation Engineering Si Edition 8Th Edition Das Solutions Manual Full Chapter PDFDocument36 pagesPrinciples of Foundation Engineering Si Edition 8Th Edition Das Solutions Manual Full Chapter PDFvernon.yang315100% (15)
- Comic Strip - 4144cfeeDocument1 pageComic Strip - 4144cfeeJoon KauravNo ratings yet
- Worksheet 19 - Weak Acids and Bases: 2 4 2 0, C BX Form The of Equations For + +Document4 pagesWorksheet 19 - Weak Acids and Bases: 2 4 2 0, C BX Form The of Equations For + +Link 08769No ratings yet
- 3.3.5 Solving Problems Involving Sampling Distribution of Sample MeansDocument2 pages3.3.5 Solving Problems Involving Sampling Distribution of Sample MeansJhasmin Jane LagamayoNo ratings yet
- Chapter2 Answers PDFDocument12 pagesChapter2 Answers PDFLalu SuhaimiNo ratings yet
- Matriks A Disebut Matriks Simetrik Jika A ADocument3 pagesMatriks A Disebut Matriks Simetrik Jika A ASuci Indah GuswiraNo ratings yet
- DH F DatosDocument2 pagesDH F DatosBrayan Chaupis GrimaldoNo ratings yet
- ExDocument13 pagesExCatalin CatalinNo ratings yet
- Metode Pendekatan Berurutan:: DiketahuiDocument4 pagesMetode Pendekatan Berurutan:: DiketahuiDinda Sari BintangNo ratings yet
- TreliçasDocument16 pagesTreliçasEmily RufinoNo ratings yet
- Nama: Fransiskus Giling NIM: 2010212397: Metode Pendekatan BerurutanDocument4 pagesNama: Fransiskus Giling NIM: 2010212397: Metode Pendekatan BerurutanDinda Sari BintangNo ratings yet
- Série 7Document6 pagesSérie 7Alya GuesmiNo ratings yet
- Item Analysis 20 6 6: InterpretationDocument7 pagesItem Analysis 20 6 6: InterpretationBernard BanalNo ratings yet
- Untitled DocumentDocument4 pagesUntitled DocumentSarada SanNo ratings yet
- DBO 1 (9:00 Am) : Grado de DilucionDocument3 pagesDBO 1 (9:00 Am) : Grado de DilucionAlonso TinocoNo ratings yet
- Mean and Variance of The Sampling Distribution of The Sample MeanDocument3 pagesMean and Variance of The Sampling Distribution of The Sample MeanCvNo ratings yet
- Ref CAXDocument1 pageRef CAXApply SofttechNo ratings yet
- Viga A: - 2.16 (8) (4) +RBY (6) 0 Rby RBY 11.52 (+) V (-) 5.76 4.32Document3 pagesViga A: - 2.16 (8) (4) +RBY (6) 0 Rby RBY 11.52 (+) V (-) 5.76 4.32Laura CentenoNo ratings yet
- Bisection MethodDocument9 pagesBisection MethodAnonymous 1VhXp1No ratings yet
- Grafik Jar Test 0 DesinfeksiDocument2 pagesGrafik Jar Test 0 DesinfeksihasryNo ratings yet
- Nava Nr. 1 TABEL SEMILĂȚIMI (M) - Pentru Partea I: Calculul de Carene DrepteDocument12 pagesNava Nr. 1 TABEL SEMILĂȚIMI (M) - Pentru Partea I: Calculul de Carene DrepteCiurea Andrei CatalinNo ratings yet
- Test 1 Maths F4Document3 pagesTest 1 Maths F4Thiyaku MaruthaNo ratings yet
- Document From DR - Md.Eleus.-compressedDocument30 pagesDocument From DR - Md.Eleus.-compressedUpgrade StoreNo ratings yet
- Chart Title: 8 9 F (X) 7.7211504037x + 0.050637123 R 0.9991358136Document2 pagesChart Title: 8 9 F (X) 7.7211504037x + 0.050637123 R 0.9991358136arifa nrNo ratings yet