Professional Documents
Culture Documents
Tejada Miguel
Tejada Miguel
Uploaded by
api-2639095050 ratings0% found this document useful (0 votes)
83 views1 pageFluorine is a nonmetal halogen from the halogens group with 9 protons, 9 electrons, and 10 neutrons. It has 7 valence electrons and forms anions by gaining 1 electron to become F-1. Fluorine is looking for cations to bond with.
Original Description:
Original Title
tejada miguel
Copyright
© © All Rights Reserved
Available Formats
PDF, TXT or read online from Scribd
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentFluorine is a nonmetal halogen from the halogens group with 9 protons, 9 electrons, and 10 neutrons. It has 7 valence electrons and forms anions by gaining 1 electron to become F-1. Fluorine is looking for cations to bond with.
Copyright:
© All Rights Reserved
Available Formats
Download as PDF, TXT or read online from Scribd
0 ratings0% found this document useful (0 votes)
83 views1 pageTejada Miguel
Tejada Miguel
Uploaded by
api-263909505Fluorine is a nonmetal halogen from the halogens group with 9 protons, 9 electrons, and 10 neutrons. It has 7 valence electrons and forms anions by gaining 1 electron to become F-1. Fluorine is looking for cations to bond with.
Copyright:
© All Rights Reserved
Available Formats
Download as PDF, TXT or read online from Scribd
You are on page 1of 1
Fluorine
Home town:
Halogens
Ethnicity:
Nonmetal
Family history:
9p
9e
10n
7 valence electron
F-1
Favorite way to
relax:
add 1 electron
anion
Looking for:
cation
You might also like
- Electronegativity: - The Tendency of An Atom To Attract Electrons From A Neighbouring AtomDocument36 pagesElectronegativity: - The Tendency of An Atom To Attract Electrons From A Neighbouring AtomBharat NimeshNo ratings yet
- Ink ChromatographyDocument9 pagesInk Chromatographyapi-2639095050% (1)
- 17th Group ElementsDocument27 pages17th Group ElementsM. Faisal100% (1)
- AS Level Chemistry Notes: BondingDocument12 pagesAS Level Chemistry Notes: BondingAyesha Ahmed100% (1)
- Polarity & Electronegativity Worksheet SOLVEDDocument1 pagePolarity & Electronegativity Worksheet SOLVEDLili0% (1)
- HalogensDocument20 pagesHalogensbrenda.mboghoNo ratings yet
- Unit 9 P-Block ElementsDocument18 pagesUnit 9 P-Block ElementsfesinNo ratings yet
- Chemistry Chapter 5Document30 pagesChemistry Chapter 5Axad Ali ChaudharyNo ratings yet
- Chemical Bonding: PHD Tamriko SupatashviliDocument112 pagesChemical Bonding: PHD Tamriko SupatashviliCypher Soth ViNo ratings yet
- Org Chap4 BirheDocument42 pagesOrg Chap4 BirhebedadadenbalprosperityNo ratings yet
- Dav School, Adambakkam Class-Xii Chemistry 7. The P-Block Elements (Class Notes)Document18 pagesDav School, Adambakkam Class-Xii Chemistry 7. The P-Block Elements (Class Notes)Maheswari RajnarayananNo ratings yet
- Chemical Bonding NotesDocument23 pagesChemical Bonding Noteskaylee chiaNo ratings yet
- F BlockDocument33 pagesF Blocksnehaa92906No ratings yet
- LANTHANIDEDocument5 pagesLANTHANIDEMahesh100% (1)
- Chapter 16 Lecture NotesDocument15 pagesChapter 16 Lecture NotesJSGINo ratings yet
- PhenolDocument9 pagesPhenolAnuki PereraNo ratings yet
- Ch1 Chem BondingDocument197 pagesCh1 Chem BondingNizarNo ratings yet
- The Groups of The Periodic TableDocument4 pagesThe Groups of The Periodic TableHannahNo ratings yet
- The D and F Block ElementsDocument27 pagesThe D and F Block ElementsPiyush GautamNo ratings yet
- Adobe Scan 02-Jul-2021Document5 pagesAdobe Scan 02-Jul-2021Uppada LaxmiNo ratings yet
- Chapter 9 Chemical Bonding F22Document12 pagesChapter 9 Chemical Bonding F22camila tutasigNo ratings yet
- Teacher Notes Stereochemistry LabDocument5 pagesTeacher Notes Stereochemistry Labjiwanjotmann26No ratings yet
- Chemical Bonding: - The Tendency of An Atom To Attract Electrons From A Neighbouring AtomDocument36 pagesChemical Bonding: - The Tendency of An Atom To Attract Electrons From A Neighbouring AtomRajiv SharmaNo ratings yet
- D - and F-Block Elements NotesDocument9 pagesD - and F-Block Elements NotesDevapriya SUNo ratings yet
- Group 17 Element1Document3 pagesGroup 17 Element1parthaprotimsen6825No ratings yet
- Electrophilic Aromatic Substitution Reaction - by WWW - Learnengineering.inDocument16 pagesElectrophilic Aromatic Substitution Reaction - by WWW - Learnengineering.inKripanshu KaushikNo ratings yet
- Chemistry Notes September 21, 2023Document2 pagesChemistry Notes September 21, 2023jennifer.tranNo ratings yet
- Introduction To The Spectroscopy of Dye MoleculesDocument41 pagesIntroduction To The Spectroscopy of Dye MoleculesYash KumarNo ratings yet
- F Block Elements 2020 21Document3 pagesF Block Elements 2020 21davidweeb2005No ratings yet
- Chem NotesDocument11 pagesChem NotesMuhammad AmmarNo ratings yet
- F BlockDocument9 pagesF BlockJyoti KrishnaNo ratings yet
- CHAPTER 6 - Part II - F-BlockDocument23 pagesCHAPTER 6 - Part II - F-BlockAdam BlerNo ratings yet
- 1-Chemical Bonding (Part 1)Document26 pages1-Chemical Bonding (Part 1)amd279151No ratings yet
- Group 17 Elements: The Halogens Family: P-Block Elements (Halogen & Noble Gases)Document20 pagesGroup 17 Elements: The Halogens Family: P-Block Elements (Halogen & Noble Gases)MoneyNo ratings yet
- Week 2 Lectures PDFDocument62 pagesWeek 2 Lectures PDFanaseini waqaNo ratings yet
- 4.1 Lewis StructureDocument69 pages4.1 Lewis StructureIrena HaisleyNo ratings yet
- The Halogen FamilyDocument4 pagesThe Halogen FamilyTrisha Gabriele LemoncitoNo ratings yet
- F-Block ElementsDocument8 pagesF-Block ElementsSai Sasivardhan GampaNo ratings yet
- 1.6 Bonding Continuum - Covalent BondsDocument4 pages1.6 Bonding Continuum - Covalent BondsPavni ChandaniNo ratings yet
- Chapter 13Document24 pagesChapter 13abubakarabubakarbah563No ratings yet
- Group VII ElementsDocument28 pagesGroup VII ElementsTichafara Paul ShumbaNo ratings yet
- Features of Periodic TableDocument19 pagesFeatures of Periodic TableMichael EumagNo ratings yet
- Easy-To-Use and Secure Digital ExamsDocument4 pagesEasy-To-Use and Secure Digital Examsspammy fammyNo ratings yet
- TEMA 5 Diatomicas 2021Document53 pagesTEMA 5 Diatomicas 2021Armando Diego GasparNo ratings yet
- 5.2 Types of BondingDocument19 pages5.2 Types of BondingAnisha Syazwana Binti RoslyNo ratings yet
- G9 Science Q2-Week 2 - Types of CompoundsDocument79 pagesG9 Science Q2-Week 2 - Types of CompoundsKarina GentonNo ratings yet
- Ionic Bonds: Chemistry Mrs. CoyleDocument35 pagesIonic Bonds: Chemistry Mrs. CoylethetlpxNo ratings yet
- Rules For Assigning Oxidation NumbersDocument1 pageRules For Assigning Oxidation NumbersKyla AngelleNo ratings yet
- Bonding Forces and Energies Al Francis Asuncion EE 3bDocument8 pagesBonding Forces and Energies Al Francis Asuncion EE 3bAce John OliverNo ratings yet
- Covalent NotesDocument6 pagesCovalent Notesjerm655No ratings yet
- Ionic Bonds: Chemistry Mrs. CoyleDocument35 pagesIonic Bonds: Chemistry Mrs. CoyleNeen NaazNo ratings yet
- The D-And F-Block Revision NotesDocument4 pagesThe D-And F-Block Revision NotesMayank Choudhary100% (1)
- P Block 17-18Document23 pagesP Block 17-18Aditya BansalNo ratings yet
- How Many Bonds To Form To Become Stable?: Table 5.1 Number of Covalent Bonds Nonmetal Atoms Usually FormDocument1 pageHow Many Bonds To Form To Become Stable?: Table 5.1 Number of Covalent Bonds Nonmetal Atoms Usually FormkalloliNo ratings yet
- S5 CHEM (The Chemistry of Group VII Elements)Document26 pagesS5 CHEM (The Chemistry of Group VII Elements)SamuelNo ratings yet
- HalogensDocument248 pagesHalogensRicky SantosoNo ratings yet
- Valence Electrons:: Electrons Available FOR BondingDocument14 pagesValence Electrons:: Electrons Available FOR BondingCharl351902 geMINIatureNo ratings yet
- Valence Electrons:: Electrons Available FOR BondingDocument14 pagesValence Electrons:: Electrons Available FOR Bondingacpd75No ratings yet
- P-Block Elements (Halogens and Noble Gases) : ChemistryDocument23 pagesP-Block Elements (Halogens and Noble Gases) : ChemistrythinkiitNo ratings yet
- Track Your LearningDocument1 pageTrack Your Learningapi-263909505No ratings yet
- Group ContractDocument3 pagesGroup Contractapi-263909505No ratings yet
- Density Floating ActivityDocument1 pageDensity Floating Activityapi-263909505No ratings yet
- Group ContractDocument1 pageGroup Contractapi-263909505No ratings yet
- Angel EsquedaDocument1 pageAngel Esquedaapi-263909505No ratings yet
- Delilah GonzalezDocument1 pageDelilah Gonzalezapi-263909505No ratings yet
- Track Your LearningDocument1 pageTrack Your Learningapi-263909505No ratings yet
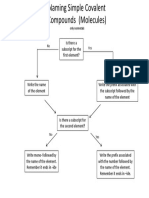
- Naming Simple MoleculesDocument1 pageNaming Simple Moleculesapi-263909505No ratings yet
- Acids and BasesDocument57 pagesAcids and Basesapi-263909505No ratings yet
- Bertha ChavezDocument1 pageBertha Chavezapi-263909505No ratings yet
- Intermolecular ForcesDocument18 pagesIntermolecular Forcesapi-263909505No ratings yet
- Ramirez JoseDocument1 pageRamirez Joseapi-263909505No ratings yet
- Sandoval DemetriDocument1 pageSandoval Demetriapi-263909505No ratings yet
- Ayala AilynDocument1 pageAyala Ailynapi-263909505No ratings yet
- Stoich Quick RefresherDocument1 pageStoich Quick Refresherapi-263909505No ratings yet
- Franco NikkoDocument1 pageFranco Nikkoapi-263909505No ratings yet
- Aldaba JosueDocument1 pageAldaba Josueapi-263909505No ratings yet
- Banda JenniferDocument1 pageBanda Jenniferapi-263909505No ratings yet
- Alavarez ArmandoDocument1 pageAlavarez Armandoapi-263909505No ratings yet
- Chavez VictorDocument1 pageChavez Victorapi-263909505No ratings yet
- Flores SammyDocument1 pageFlores Sammyapi-263909505No ratings yet
- Leyva AriamDocument1 pageLeyva Ariamapi-263909505No ratings yet
- Llanas AlexisDocument1 pageLlanas Alexisapi-263909505No ratings yet
- Marzan MiguelDocument1 pageMarzan Miguelapi-263909505No ratings yet
- Martinez KevinDocument1 pageMartinez Kevinapi-263909505No ratings yet
- Ortega DavidDocument1 pageOrtega Davidapi-263909505No ratings yet
- Brite JimmyDocument1 pageBrite Jimmyapi-263909505No ratings yet
- Hernandez CrystalDocument1 pageHernandez Crystalapi-263909505No ratings yet
- Torres CassieDocument1 pageTorres Cassieapi-263909505No ratings yet