Professional Documents
Culture Documents
Equ28-01 Sysmex XE2100 Op SOP
Uploaded by
Wasim AkramOriginal Description:
Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
Equ28-01 Sysmex XE2100 Op SOP
Uploaded by
Wasim AkramCopyright:
Available Formats
Whole Blood Analysis (Full Blood Count-FBC) Using SYSMEX XE-2100
Haematology Automated Analyser.
Non-Smile Contributor
Document Number:
Effective (or Post) Date:
Document Origin: BHHRL
Company:
SMILE Approved by:
Review by
Heidi Hanes
Review date
Author: NA
Equ28-01
13-Jan-09
NA
Heidi Hanes
13-Feb-12
SMILE Comments: This document is provided as an example only. It must be revised to accurately reflect your labs
specific processes and/or specific protocol requirements. Users are directed to countercheck facts when considering
their use in other applications. If you have any questions contact SMILE.
Document No.: BHHRL/005TM-01
TEST METHODS MANUAL
Botswana-Harvard HIV
Reference Laboratory
(BHHRL)
Document
Title
Whole Blood Analysis (Full Blood CountFBC) Using SYSMEX XE-2100 Haematology
Automated Analyser.
Document No.: BHHRL/005EQ-01
SYSMEX XE-2100 HAEMATOLYGY AUTOMATED ANALYSER MAINTENANCE
AND TROUBLESHOOTONG PROCEDURE
Prepared by:
Name, Title
History
Tapologo Nkoane
Current
Version
#
1.0
Supersedes
Version #
Effective
Date
(dd/mm/yy)
0.0
Description Notes
First issue
Review Date
Revision Date
Signature
Annual
Review:
Approved
by:
Effective Date:
Name, Title
Laboratory Director (MOH)
Laboratory Director (HSPH)
Doc #: BHHRL/005TM-01
Page 2 of 16
Signature
Date
Version 2.0
TEST METHODS MANUAL
Botswana-Harvard HIV
Reference Laboratory
(BHHRL)
005TM-01
01.0
01.1
01.2
01.3
01.4
01.5
01.6
01.7
01.8
01.9
01.10
01.11
01.12
01.13
Document No.: BHHRL/005TM-01
Whole Blood Analysis (Full Blood CountFBC) Using SYSMEX XE-2100 Haematology
Automated Analyser.
Whole Blood Analysis (Full Blood Count-FBC) Using
SYSMEX XE-2100 Haematology Automated Analyser.
Purpose
Application Scope
Responsibilities
Principle of the Test
01.3.1
Clinical Aspects
01.3.2
System Overview
01.3.2
Measurement Principles
Specimen
01.4.1
Specimen Type/Stability/Storage
01.4.2
Specimen Rejection Criteria
01.4.3
Specimen Interferences
Safety Precautions
Equipment and Apparatus
Reagents
Procedure
01.8.1
Instrument Start-Up
01.8.2
Running Controls
01.8.3
Running Patient Samples
01.8.4
Performing WBC Differential
Quality Control
01.9.1
Internal Quality Control
01.9.1.1
Running Controls
01.9.1.2
Blinded Samples
01.9.1.3
Inter-Laboratory Comparison
01.9.2
External Quality Control
Validation of Results
01.10.1
Quality Control Data
01.10.2
Patients Results
Maintenance and Troubleshooting
References
Co-Applicable Quality Management Documents
Effective Date:
Doc #: BHHRL/005TM-01
Page 3 of 16
Version 2.0
TEST METHODS MANUAL
Botswana-Harvard HIV
Reference Laboratory
(BHHRL)
01.0
Document No.: BHHRL/005TM-01
Whole Blood Analysis (Full Blood CountFBC) Using SYSMEX XE-2100 Haematology
Automated Analyser.
PURPOSE
This chapter describes the test method (automated) used for whole blood analysis (Full
Blood Count - FBC) using the Sysmex XE-2100 Haematology Analyser.
01.1
APPLICATION SCOPE
This chapter applies to Botswana-Harvard HIV Reference Laboratory
01.2 RESPONSIBILITIES
All Haematology laboratory personnel are required to be knowledgeable of this
procedure. New employees are trained and assessed for competence before they can
handle patient samples. Documentation of training on this procedure can be found in the
Personnel Files.
03.3
PRINCIPLE OF THE TEST
01.3.1 Clinical Aspects
Knowing the proportions of the different components (neutrophils, lymphocytes,
monocytes, red blood cells, basophils, haemoglobin and reticulocytes) of peripheral
blood is vital in distinguishing a diseased and normal blood. Full blood count (FBC) as is
normally termed is a useful tool in detecting toxicity of some drugs administered to
patients.
01.3.2 System Overview
XE-2100 performs haematology analyses according to the RF/DC detection method,
HydroDynamic Focusing (DC Detection), flow cytometry method (using the
semiconductor laser) and SLS-haemoglobin method.
RF/DC Detection Method: This method detects the size of the blood cells by changes in
direct-current resistance, and the density of the blood cell interior by changes in radiofrequency resistance. A blood sample is aspirated and measured, diluted to the specified
ratio, and send to the applicable detector chamber. Inside the chamber is a tint hole called
an aperture, on both sides of which are electrodes. Between the electrodes flow the
direct current and radio-frequency current. Blood cells suspended in the diluted sample
pass through the aperture, changing the direct-current resistance and radio-frequency
Effective Date:
Doc #: BHHRL/005TM-01
Page 4 of 16
Version 2.0
TEST METHODS MANUAL
Botswana-Harvard HIV
Reference Laboratory
(BHHRL)
Document No.: BHHRL/005TM-01
Whole Blood Analysis (Full Blood CountFBC) Using SYSMEX XE-2100 Haematology
Automated Analyser.
resistance between the electrodes. The size of the blood cell is detected via changes in the
direct-current resistance, and the density of the blood cell interior (size of the nucleus) is
detected via changes in the radio0frequency resistance, with such detections coming in
the form of electrical pulses. Based on the size of these pulses, a two dimensional
distribution (scatter gram) of the blood-cell size and internal density can be drawn.
Hydro Dynamic Focusing (DC Detection): Inside the detector, the sample nozzle is
positioned in front of the aperture and in line with the centre. After dilute sample is
forced from the sample nozzle into the conical chamber, it is surrounded by front sheath
reagent and passes through the aperture centre. After passing through the aperture, the
diluted sample is surrounded by back sheath reagent and sent to the catcher tube. This
prevents the blood cells in this area from drifting back, and prevents the generation of
false platelets pulses. This method improves blood count accuracy and reproducibility.
Since the blood passes through the aperture in a line, it also prevents the generation of
abnormal blood call pulses.
Flow Cytometry Method Using Semiconductor Laser: Cytometry is used to analyze
physiological and chemical characteristics of cells and other biological particles. Flow
cytometry is used to analyze those cells and particles as they are passed through
extremely small flows. A blood sample is aspirated and measured, diluted to the specified
ratio and stained. The sample is then fed into the flow cell. A semiconductor laser beam
is emitted to the blood cells passing through the flow cell. The forward scattered light is
received by the photodiode, and the lateral scattered light and lateral fluorescent light are
received by the photomultiplier tube. This light is converted into electrical pulses, thus
making it possible to obtain blood cell information.
Forward Scattered light and lateral Scattered Light: When obstacles such as particles
exist in the way of light, the light beam scatters from each obstacle in various directions
(light scattering). By detecting the scattered light it is possible to obtain information on
cell size and material properties.
When laser beam is emitted to blood cell particles, light scattering occurs. The intensity
of the light scatter depends on factors such as the particle diameter and viewing angle.
Forward scattered light provides information on blood cell size and lateral scattered light
provides information on the cell interior (nucleus).
Lateral Florescent light: When light is emitted to fluorescent material, such as stained
blood cells, light of longer wavelength than the original light is produced. The intensity
of the fluorescent light increases as the concentration of the stain becomes higher. By
measuring the intensity of the fluorescence emitted, you can obtain information on the
degree of blood staining. Fluorescence light is emitted in all directions; the XE2100
detects the fluorescent light that is emitted sideways.
Effective Date:
Doc #: BHHRL/005TM-01
Page 5 of 16
Version 2.0
TEST METHODS MANUAL
Botswana-Harvard HIV
Reference Laboratory
(BHHRL)
Document No.: BHHRL/005TM-01
Whole Blood Analysis (Full Blood CountFBC) Using SYSMEX XE-2100 Haematology
Automated Analyser.
SLS-Haemoglobin Method: SULFOLYSER is added to the red blood cells, and
haemoglobin is converted into SLS-haemoglobin. Concentration of SLS-haemoglobin is
measured as light absorbance, and is calculated by comparison with the absorbance of the
diluent measured before the sample was added.
01.3.3 Measurement Principles
The XE-2100 can analyse and output the results for 32 parameters of blood samples. It
utilizes technology of fluorescence flow cytometry to quantitate the standard five part
differential, immature granulocytes (metamyelocytes, myelocytes and promyelocytes),
nucleated red blood cells (NRBC), reticulocyte count, immature reticulocyte fraction and
optical fluorescent platelet count. The combination of side scatter (inner complexity of
the cell), forward scatter (volume) and fluorescence intensity of nucleated cells gives a
concise but precise image of each cell detected in the peripheral blood. A well-defined
physical description of the different leucocyte populations (clusters) is obtained.
Abnormal and immature cells, with their larger nuclear volume show much higher
fluorescence intensity than normal cells, and are easily distinguishable in the DIFF
scattergram.
Measurement of WBC
WBC/BASO channel
RBCs are lysed with the acid haemolytic reagent STROMATOLYSER-FB. This reagent
selectively suppresses the degranulation of Basophils, resulting in their separation from
other WBC. After this reaction the sample is analyzed by flow cytometry using a
semiconductor laser to detect forward and side scattered light information, based on
which a WBC/BASO scattergram is obtained. By analyzing this scattergram, WBC and
Basophil counts are taken.
WBC 4-part differential
RBCs are lysed with STROMATOLYSER-4DL. At the same time the reagent acts on the
WBC membrane to allow dye passage .STROMATOLYSER-4DS ( dying solution) is
then added to allow the dye to enter WBC at the damages portion of its membrane and
stain the DNA and RNA therein. Following this reaction the sample is then analyzed by
flow cytometry using the s semiconductor laser to detect forward and side scattered light
information, based on which a 4-DIFF scattergram is obtained. By analyzing this
scattergram, 4-parameter counts (lymphocytes, monocytes, Eosinophils, Neutrophils and
Basophils are taken.
The immature information (IMI) channel of the XE-2100 counts human progenitor cells
(HPC). The reagents specifically affect the lipid components of the cell membranes; the
Effective Date:
Doc #: BHHRL/005TM-01
Page 6 of 16
Version 2.0
TEST METHODS MANUAL
Botswana-Harvard HIV
Reference Laboratory
(BHHRL)
Document No.: BHHRL/005TM-01
Whole Blood Analysis (Full Blood CountFBC) Using SYSMEX XE-2100 Haematology
Automated Analyser.
membranes of mature cells, with a higher content of lipid are lysed while immature cells
retain their membranes. In normal samples no intact cells are seen in the IMI area. The
IMI channel is used to discriminate between immature and mature white blood cells. It
utilizes the RF/DC detection method.
Measurement of RET and optical measurement of PLT
Blood is diluted with RET SEARCH (II) to preset concentration. RET SEARCH (II)
staining solution is also added to stain WBC DNA and RNA and RET RNA. The sample
is then analyzed by flow cytometry using semiconductor laser to detect forward scattering
light and side fluorescence information, based on which an RET scattergram is obtained.
By analyzing this scattergram, RET counts, RET ratios for individual fluorescence
intensity zones (LFR, MFR, HFR), immature reticulocyte fraction (IRF) and PLT-O are
determined.
Platelets are also measured with sheath flow DC detection method. By measuring larger
number of platelets this method offers increased accuracy. However, in cases where a
large number of RBC fragments or large Platelets are present, accuracy tends to decrease.
For such samples, the PLT-o measurement ensures higher accuracy than the DC detection
method. XE-2100 presents the platelet counts of high accuracy by adopting the results
from either method that seems more accurate judging from the cell distributions and the
scattergrams obtained.
Measurement of NRBC
Nucleated red blood cells (NRBC) occur in very low concentrations; however are found
in high concentrations in haemolytic diseases of new born, sickle cell anaemia and
thalassaemia major particularly following splenectomy. In NRNC channel the cell
membranes of NRBC are lysed with the STROMATOLYZER_NR diluent to expose the
nuclei of NRBC, the white cells are slightly perforated to allow quick influx of the dye,
but remain intact. Nuclear material is stained with a polymethine based fluorescent dye
and cells and nuclei are hydrodynamically focused. Fluorescence intensity and forward
scatter light intensity of each cell are electronically analyzed. This allows the clear
separation of leucocytes and NRBC.
01.4
SPECIMEN
01.4.1 Specimen Type/Stability/Storage
Some anticoagulants will alter test results due to their effects on haemolysis and
blood platelet agglutination. Therefore, use EDTA-2K, 3K or 2Na as the
anticoagulant.
Effective Date:
Doc #: BHHRL/005TM-01
Page 7 of 16
Version 2.0
TEST METHODS MANUAL
Botswana-Harvard HIV
Reference Laboratory
(BHHRL)
Document No.: BHHRL/005TM-01
Whole Blood Analysis (Full Blood CountFBC) Using SYSMEX XE-2100 Haematology
Automated Analyser.
Specimens should be stored at room temperature of 18 - 26C or in the
refrigerator of 2 - 8C. If stored in a refrigerator, samples should be returned to
room temperature, for approximately 30 minutes, before analysis. Otherwise
correct results may not be obtained.
01.4.2 Specimen Rejection Criteria
For specimen rejection criteria, refer to the following SOP:
*
Procedure for Specimen Rejection Criteria
01.4.3 Specimen Interferences
Some abnormal samples may interfere with automated cell counting methods. The
following is a list of possible substances that may interfere with the listed
parameters.
*
WBC: Cold agglutinins, platelet aggregation, nucleated RBCs,
cryoglobulins, lyse-resistant RBCs in patients with haemoglobinopathies,
severe liver disease or neonates.
*
RBC: Cold agglutinins, severe micryocytosis, fragmented RBCs, large
numbers of giant platelets, in vitro haemolysis.
*
Hgb: Lipemia, abnormal proteins in blood plasma, severe leukocytes
(above 100,000/l). The effect of abnormal proteins and Lipemia may be
removed by plasma replacement or plasma blank procedures.
*
Hct: Cold agglutinins, leukocytosis (above 100,000/l), abnormal red
cell fragility.
*
PLT: Pseudothrombocytopenia,
platelet
aggregation,
increased
micrcrocytosis, megalocyttic platelets
01.5
SAFETY PRECAUTIONS
For information on safety precautions, refer to the following SOPs:
*
Universal Precautions Policy (Document No.: BHHRL/010POL-)
*
Waste Disposal Policy (Document No.: BHHRL/010POL-)
In addition please take care of the following warning:
*
If the instrument emits abnormal odour or smoke, turn off the power
immediately and disconnect the power plug from the wall socket.
*
Do not touch the electrical circuits inside the cover, particularly if your
hands are wet. Electrical shock may result.
*
When discarding waste fluid, or disassembling and assembling the related
parts, always wear protective clothing, gloves, and eyeglasses to prevent
contact with contaminated fluids or parts.
01.6
EQUIPMENT AND APPARATUS
1.
Effective Date:
Sysmex XE-2100
Doc #: BHHRL/005TM-01
Page 8 of 16
Version 2.0
Document No.: BHHRL/005TM-01
TEST METHODS MANUAL
Botswana-Harvard HIV
Reference Laboratory
(BHHRL)
Whole Blood Analysis (Full Blood CountFBC) Using SYSMEX XE-2100 Haematology
Automated Analyser.
For Manual Differential
1.
Microscope
2.
Microscope Slides
3.
Oil Immersion
4.
Stains
5.
Applicator Stick
01.7
REAGENTS
Reagent
CellPack (EPK)
CellSheath (ESE)
Stromatolyser-FB (FBA)
Stromatolyser-4DL (FFD)
Stromatolyser-4DS (FFS)
Stromatolyser-NR (SNR) (lyse reagent, dye solution)
Sulfolyser (SLS)
Stromatolyser-IM (SIM)
Ret Search (II) (RED) (diluent, dye solution)
CellClean (detergent)
Storage Conditions
5 - 30C
5 - 30C
5 - 30C
2 - 35C
2 - 35C
2 - 35C
1 - 30C
5 - 30C
2 - 35C
1 - 30C
These reagents are supplied ready for use and are stable until the stated expiry date when
stored at the recommended storage conditions.
When using reagents be cautious of the following:
After unpacking, be sure not to allow dust, dirt, or bacteria to come in touch
with the reagent.
Do not use the reagents which are out of expiration date.
Handle a reagent gently to prevent formation of bubbles.
Take care not to spill a reagent. If it spills, wipe it off immediately in
accordance with BHHRLs policy.
Refer to the MSDS provided for each reagent.
01.8
PROCEDURE
01.8.1
Instrument Start-Up
Refer to:
SYSMEX XE-2100 Haematology Automated Analyser
Equipment
Operation
Instructions.
Document
No.:
BHHRL/005TM-01.
Effective Date:
Doc #: BHHRL/005TM-01
Page 9 of 16
Version 2.0
Document No.: BHHRL/005TM-01
TEST METHODS MANUAL
Botswana-Harvard HIV
Reference Laboratory
(BHHRL)
01.8.2
Whole Blood Analysis (Full Blood CountFBC) Using SYSMEX XE-2100 Haematology
Automated Analyser.
Running Controls
Refer to Section 01.9.1.1 of this procedure.
01.8.3
Running Patient Samples
Refer to:
SYSMEX XE-2100 Haematology
Equipment Operation Instructions.
01.8.4
Performing WBC Differential
Automated
Analyser
Peripheral Blood Smear
If the machine does not produce WBC differential or when in doubt of some parameters,
WBC Differential Count is performed.
You will need a capillary tube, 1x3 inch glass slides one with a frosted end and the
other non-frosted, a pencil or a diamond pen gloves and whole blood in EDTA tube.
1. Place a clean 3 x 1 inch glass slide on a flat surface (stationary slide), one with a
frosted end.
2. Mix the blood well. With a capillary tube, transfer a drop of blood
approximately 2 to 3 mm in diameter to the stationary slide about - inch from
the end of the frosted on the same side as the writing hand.
3. Hold the end of the stationary slide opposite the blood drop with the non-writing
hand.
4. Position a spreader (non-frosted) slide at a 25 to 30 degree angle to the
stationary slide and bring it back into the drop of blood.
5. Allow the blood to spread along the back of the spreader slide.
6. Immediately push the spreader slide forward with a smooth and rapid stroke,
maintaining the same angle and exerting very little pressure. The blood will be
pulled behind the spreader.
7. If the angle of the spreader slide is proper, the speed of the stroke is moderately
fast, and the size of the drop of blood is as specified, the blood should feather
into nothing somewhere between one-half and three-quarters of the way along
the stationary slide.
8. Label the slide at the frosted end with the Patient identification number and
date, using a pencil or a diamond pen.
9. Air-dry the blood rapidly but thoroughly (several minutes) before staining. You
can do this manually or with a cool air blower.
10. Stain
the
smear
using
a
Hema-Tek
stainer
MAY-GRUNWALD GIEMSA STAIN / MANUAL STAINING
-flood smear with may-grunwald stain
Effective Date:
Doc #: BHHRL/005TM-01
Page 10 of 16
Version 2.0
TEST METHODS MANUAL
Botswana-Harvard HIV
Reference Laboratory
(BHHRL)
Document No.: BHHRL/005TM-01
Whole Blood Analysis (Full Blood CountFBC) Using SYSMEX XE-2100 Haematology
Automated Analyser.
-allow to satin for 5 minutes
-flush and replace the stain with 10% freshly prepared Giemsa stain
-leave for 10 minutes
-wash the smear with tap water
- allow the smear to air dry
11. Perform smear analysis and report the results accordingly.
01.9
QUALITY CONTROL
E-check (the control used on the Sysmex XE-2100 Haematology Analyser) is an XE-diff
control, manufactured by STRECK LABORATORIES for SYSMEX and intended for
use in Haemocytometry to monitor daily accuracy and precision of cell analysers and
manual analytical procedures. These controls contain Human RBC, mammalian WBC
and platelets. All the cells are suspended in a plasma like fluid. XE-Diff control is
manufactured in such a way that it simulates whole blood. When used on automated cell
analysers, the following haemacytometric values may be obtained: tWBC; %NE, %LY,
%MO, %ES, %BAS, RBC, HGB, Haematocrit, MCV, MCH,MCHC,PLT and all other
values directly derived from those listed.
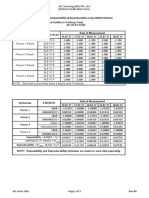
01.9.1 Internal Quality Control
Control samples are analyzed by the X bar or the L-J control programs, and the data is
stored in the quality control file. At least two levels of controls should be run before
analyzing the patient samples. X bar program analyzes the control twice in succession
and the average data is used. L-J program on the other hand, uses the results from one
analysis as one control data. In this lab we use the L-J program.
01.9.1.1Running Controls
1. Bring the controls to room temperature before analysis.
2. Run the controls as per the procedure on Sysmex XE-2100 Operation Instructions.
3. Review the control runs. If all the parameters fall within the manufacturers
recommended ranges, then proceed with patient samples. Otherwise execute the
necessary corrective action for out-of-range parameters until all the parameters
fall within the acceptable range.
01.9.1.2 Internal Control Blinded Samples
During each run, one sample is run every 20 sample using sampler mode. The sample is
given the ID that starts with C then followed by two digits for day, two digits for month
Effective Date:
Doc #: BHHRL/005TM-01
Page 11 of 16
Version 2.0
TEST METHODS MANUAL
Botswana-Harvard HIV
Reference Laboratory
(BHHRL)
Document No.: BHHRL/005TM-01
Whole Blood Analysis (Full Blood CountFBC) Using SYSMEX XE-2100 Haematology
Automated Analyser.
and two digits for year, M or an A for morning and afternoon depending on the time
of the day the test is done, e.g. If sample is run on morning of November 3, 2003, its ID
should appear like; C031103M. The results are filed and reviewed every week by the
haematology technologist and BHHRL Quality Manager to monitor trends and any
deviations.
01.9.2.3 Inter-Laboratory Comparison
Once a week, about 5 samples are run with XE-2100 at BHHRL and PMH laboratory
using Coulter Max by the same technologist. The results are filed and reviewed every
month by the Haematology technologist and BHHRL Quality manager.
01.9.2 External Quality Control
The Botswana-Harvard HIV Reference Laboratory is participating in the CAP
Proficiency Testing Program for this assay, which is performed three times in a year. The
laboratorys performance in this program is reviewed by the Quality Manager and the
personnel running the test every time results are received from CAP and corrective
measures implemented where necessary, for purposes of continuous improvement.
01.10
VALIDATION OF RESULTS
01.10.1
Quality Control Data
After analysis the OK and CANCEL of the function will appear. When you select
OK the analysis data is displayed to the IPU. When you select CANCELL, data is
not transmitted to IPU and the operation ends. If the message CHECK CONTROL
CHART appears in the action message column, the analyzed data has exceeded the
control limit. IF the message REANALYZE CONTROL appears, the analyzed data is
more than three times the limit. The + and - signs in the Flag column indicate which
parameters are outside the control limits and which are above or below the limits. When
the analyzed data is more than 3 times the limit, + and -will be highlighted.
01.10.2
Patients Result
The results are automatically printed. Check if all the tests have been performed and
check for error messages and for tests with no results. Generally RBC count is 3 times the
haemoglobin and the haemoglobin is three times the Haematocrit.
Effective Date:
Doc #: BHHRL/005TM-01
Page 12 of 16
Version 2.0
TEST METHODS MANUAL
Botswana-Harvard HIV
Reference Laboratory
(BHHRL)
Document No.: BHHRL/005TM-01
Whole Blood Analysis (Full Blood CountFBC) Using SYSMEX XE-2100 Haematology
Automated Analyser.
If the machine does not produce WBC differential or when in doubt of some parameters,
make a peripheral blood smear, stain and perform WBC Differential Count.
Review the results, initial and date before passing them to be checked and authorised
(initialled and dated) for release by an authorised laboratory personnel.
For electronic results, authorised laboratory personnel will validate and accept them
online before they are released into the LIMS.
01.11 MAINTENANCE AND TROUBLESHOOTING
Refer to procedure on Sysmex XE-2100 Maintenance and Troubleshooting Procedure.
In cases where there is technical problem with the analyser and therefore cannot be used,
the SYSMEX SF-3000 is used as the back-up, following procedure no. BHHRL/005TM01.
01.12 REFERENCES
Stiene-Martin E.A., Lotspeich-Steininger C.A. and Koepe, J. A. 1998. Clinical
Haematology. 2nd Edition. Lippincott. New York.
Sysmex XE-2100 Operation Manual (February 1999 revision)
Sysmex XE-2100 IPU Operators Manual (June 1999 revision)
01.13 CO-APPLICABLE QUALITY MANAGEMENT DOCUMENTS
Sysmex XE-2100 Maintenance Log Sheet
Haematology (Using Sysmex XE-2100) Reagent Information Log Sheet
Sysmex XE-2100 Maintenance And Troubleshooting Procedure
Sysmex XE-2100 Haematology Automated Analyser Equipment Operation Instructions
Effective Date:
Doc #: BHHRL/005TM-01
Page 13 of 16
Version 2.0
Document No.: BHHRL/005TM-01
TEST METHODS MANUAL
Botswana-Harvard HIV
Reference Laboratory
(BHHRL)
Whole Blood Analysis (Full Blood CountFBC) Using SYSMEX XE-2100 Haematology
Automated Analyser.
APPENDIX I
HAEMATOLOGY ADULT NORMAL RANGES
INSTRUMENT USED: SYSMEX XE 2100
PARAMETER
Red Blood Cell Count (RBC)
Men
Women
Haemoglobin (Hb)
Men
Women
Haematocrit (HCT)
Men
Women
Mean Cell Volume (MCV)
Men
Women
Mean Cell Haemoglobin (MCH)
Men
Women
Mean Cell Haemoglobin Concentration (MCHC)
Men
Women
Red Cell Distribution Width (RDW)
White Blood Cell Count (WBC)
Differential White Cell Count (Diff)
Neutrophils
Lymphocytes
Monocytes
Eosinophils
Basophils
Platelet Count
Effective Date:
REFERENCE RANGE
4.5 5.5 x 1012/l
3.8 4.8 x 1012/l
13 17 g/dl
12 15 g/dl
40 50 %
36 45 %
83 99 fl
83 99 fl
27 32 pg
27 32 pg
32 34 g/dl
32 34 g/dl
11.6 14.0 %
4-10 x 109/l
40 80 % (2 - 7 x 109/l)
20 40 % (1 3 x 109/l)
2- 10 % (0.2 1.0 x 109/l)
1 6 % (0.02 0.5 x 109/l)
< 1- 2 % (0.02 0.1 109/l)
150 400 x 109/l
Doc #: BHHRL/005TM-01
Page 14 of 16
Version 2.0
TEST METHODS MANUAL
Botswana-Harvard HIV
Reference Laboratory
(BHHRL)
Document No.: BHHRL/005TM-01
Whole Blood Analysis (Full Blood CountFBC) Using SYSMEX XE-2100 Haematology
Automated Analyser.
This procedure has been read and understood by the undersigned:
Name of Officer
Effective Date:
Signature
Doc #: BHHRL/005TM-01
Page 15 of 16
Initials
Date
Version 2.0
TEST METHODS MANUAL
Botswana-Harvard HIV
Reference Laboratory
(BHHRL)
Whole Blood Analysis (Full Blood CountFBC) Using SYSMEX XE-2100 Haematology
Automated Analyser.
Name of Officer
Effective Date:
Document No.: BHHRL/005TM-01
Signature
Doc #: BHHRL/005TM-01
Page 16 of 16
Initials
Date
Version 2.0
You might also like
- Sysmex XS-1000i Automated Hematology AnalyzerDocument3 pagesSysmex XS-1000i Automated Hematology AnalyzerAdi Subagio0% (1)
- GPT PDFDocument1 pageGPT PDFHussein N. FarhatNo ratings yet
- Sfri Brochure-Hemix330 Eng BDDocument4 pagesSfri Brochure-Hemix330 Eng BDYair Tigre100% (1)
- Compact Yet Robust: Chemistry AnalyzerDocument4 pagesCompact Yet Robust: Chemistry AnalyzerThiago GalloNo ratings yet
- DXH 520 Implementation Workbook 2.2Document94 pagesDXH 520 Implementation Workbook 2.2Eureca Chara firmanNo ratings yet
- PDFDocument526 pagesPDFjairodiaz88No ratings yet
- Automation UrinalysisDocument36 pagesAutomation UrinalysisThynee Gamalong TagoNo ratings yet
- Sysmex CS 5100Document4 pagesSysmex CS 5100Katamba RogersNo ratings yet
- Precipath HDL - LDL-C.11818171001.V10.en PDFDocument2 pagesPrecipath HDL - LDL-C.11818171001.V10.en PDFARIF AHAMMED PNo ratings yet
- BIO RAD Evolis System ManualDocument4 pagesBIO RAD Evolis System ManualAhmedJuman0% (1)
- Tersaco Santhia 1200Document2 pagesTersaco Santhia 1200yogesh kumarNo ratings yet
- Insert - Elecsys Syphilis - Ms 07802960190.V3.EnDocument5 pagesInsert - Elecsys Syphilis - Ms 07802960190.V3.EnGuneyden GuneydenNo ratings yet
- 3.BC-5800 CompetitorsDocument67 pages3.BC-5800 CompetitorskiryNo ratings yet
- Volume PDFDocument10 pagesVolume PDFBelon Kaler Biyru IINo ratings yet
- NRBCDocument15 pagesNRBCDara VinsonNo ratings yet
- XL Series Parts Concept ListDocument12 pagesXL Series Parts Concept ListIllusive ManNo ratings yet
- Lab 1 Reaserch Assigment Al-Hassan KenaanDocument6 pagesLab 1 Reaserch Assigment Al-Hassan KenaanAlhassan KenaanNo ratings yet
- c111 Operator Manual PDFDocument502 pagesc111 Operator Manual PDFSuponoNo ratings yet
- HydraulicsDocument101 pagesHydraulicsHuseyn aliyevNo ratings yet
- Above and Beyond: BC-760 & BC-780Document3 pagesAbove and Beyond: BC-760 & BC-780Dam L0% (1)
- Leukocytosis: Basics of Clinical Assessment: Production, Maturation and Survival of LeukocytesDocument9 pagesLeukocytosis: Basics of Clinical Assessment: Production, Maturation and Survival of LeukocytesKreshnik HAJDARINo ratings yet
- Cobas C 311 enDocument12 pagesCobas C 311 enCássio Menezes GodóiNo ratings yet
- HM 12 38078v1 WW Ruby Brochure 8.5x11 100112Document8 pagesHM 12 38078v1 WW Ruby Brochure 8.5x11 100112vijayramaswamyNo ratings yet
- Service Manual Coulter GEN S PDFDocument1,192 pagesService Manual Coulter GEN S PDFReynaldo MacarioNo ratings yet
- Acute Myocardial InfarctionDocument45 pagesAcute Myocardial Infarctionwaqas_xsNo ratings yet
- Urinalysis Control - Level 2 (Urnal Control 2) : Cat. No. UC5034 Lot No. 737UC Size: Expiry: 2015-10Document6 pagesUrinalysis Control - Level 2 (Urnal Control 2) : Cat. No. UC5034 Lot No. 737UC Size: Expiry: 2015-10Meiliana ThanNo ratings yet
- Iflash 1200 Operation TrainingDocument118 pagesIflash 1200 Operation Trainingmahmoud boghdadyNo ratings yet
- XN-L - Reference Interval From General Information 2017Document4 pagesXN-L - Reference Interval From General Information 2017widiawaty100% (1)
- HTI CL-500 Urine Analyzer: Quality Products and Service For Healthcare ProfessionalsDocument2 pagesHTI CL-500 Urine Analyzer: Quality Products and Service For Healthcare ProfessionalsTrình BiomedicNo ratings yet
- Section 3 Electronics: June 5, 2008Document12 pagesSection 3 Electronics: June 5, 2008Huseyn aliyevNo ratings yet
- Tosoh Bio Science - G8 ChromatogramsDocument1 pageTosoh Bio Science - G8 ChromatogramsimrecoNo ratings yet
- 0812 in Vitro Blood Gas Analyzers GuideDocument9 pages0812 in Vitro Blood Gas Analyzers GuidedjebrutNo ratings yet
- Manual DXH 560 HEMATOLOGIADocument142 pagesManual DXH 560 HEMATOLOGIAcpdNo ratings yet
- UniCel DXC 600 (Inglés)Document59 pagesUniCel DXC 600 (Inglés)Luis100% (1)
- XS-1000 Automated Hematology Analyzer: Small Footprint. Big DifferenceDocument4 pagesXS-1000 Automated Hematology Analyzer: Small Footprint. Big Differencerizal_aspanNo ratings yet
- K-Lite5 Tech SpecDocument1 pageK-Lite5 Tech SpecNayaRahmaniNo ratings yet
- 3.8 HDSD NeoChem 100Document117 pages3.8 HDSD NeoChem 100huy Lê xuan Thanh100% (1)
- Sysmex CS 2100iDocument3 pagesSysmex CS 2100iKatamba RogersNo ratings yet
- Method Verification of TestDocument2 pagesMethod Verification of TestObak PrithibiNo ratings yet
- XP PM Standard Procedure enDocument7 pagesXP PM Standard Procedure enKarim KarimmNo ratings yet
- 1975ec-2026ec-2028ec 2023-04Document57 pages1975ec-2026ec-2028ec 2023-04Sujit KushwahaNo ratings yet
- KX-21 Training: Main MenuDocument159 pagesKX-21 Training: Main MenuHuseyn aliyev100% (1)
- Abx Pentra400 ManualDocument24 pagesAbx Pentra400 ManualNurNo ratings yet
- BC 760 BC 780 BrochureDocument2 pagesBC 760 BC 780 BrochureBio AllianceNo ratings yet
- SOP05-5002F Service Manual ProlyteDocument41 pagesSOP05-5002F Service Manual ProlyteRogger Ruffini100% (1)
- Operation ManualDocument50 pagesOperation ManualAhmed TawfikNo ratings yet
- Mindray BS120 User ManualDocument6 pagesMindray BS120 User ManualMichael Okeke100% (1)
- Bs 800 Chemistry Analyzer PDFDocument1 pageBs 800 Chemistry Analyzer PDFДимонNo ratings yet
- En EA Cobas 4000 Analyzer SeriesDocument20 pagesEn EA Cobas 4000 Analyzer Seriesatiqa aslamNo ratings yet
- Market Review Blood Gas Analyser S 2010Document43 pagesMarket Review Blood Gas Analyser S 2010Fercalo AndreiNo ratings yet
- Multistix Reagent StripDocument3 pagesMultistix Reagent StripFrancis TorresNo ratings yet
- Control de Calidad 1039Document106 pagesControl de Calidad 1039MARITZA MUÑOZ100% (1)
- Hameg hm5010 hm5011 Spectrum Analyzer Service Manual PDFDocument40 pagesHameg hm5010 hm5011 Spectrum Analyzer Service Manual PDFFrancisco Rodriguez MartinezNo ratings yet
- I-STAT 1 System Manual US English 014331-00 47ADocument676 pagesI-STAT 1 System Manual US English 014331-00 47AHuan Luu Minh100% (1)
- XNL Hydraulic Suga OCT21AMDocument24 pagesXNL Hydraulic Suga OCT21AMOmar GómezNo ratings yet
- PentraDocument8 pagesPentraDV0% (1)
- Xt1800i-Xt2000i Astm Host InterfaceDocument43 pagesXt1800i-Xt2000i Astm Host InterfaceNguyenTueNo ratings yet
- Unit Iv: Community Health Nursing Approaches, Concepts and Roles and Responsibilities of Nursing PersonnelDocument17 pagesUnit Iv: Community Health Nursing Approaches, Concepts and Roles and Responsibilities of Nursing Personnelkles insgkkNo ratings yet
- ABG Practice QuestionsDocument4 pagesABG Practice Questionsbbarnes0912No ratings yet
- Mia Deal or No Deal 2.3.7.A1Document2 pagesMia Deal or No Deal 2.3.7.A1bxnds. zay0% (1)
- Ob MidtermDocument5 pagesOb MidtermSheila Tolentino-BelanioNo ratings yet
- Physical Assessment For Stroke PatientDocument4 pagesPhysical Assessment For Stroke Patientdana75% (4)
- Slu Brochure Ay 2014-2015Document2 pagesSlu Brochure Ay 2014-2015Michael BermudoNo ratings yet
- Aira Noreen Leyva - Resume ReviewDocument2 pagesAira Noreen Leyva - Resume Reviewapi-447475293No ratings yet
- PICS GMP Inspection PackagingDocument8 pagesPICS GMP Inspection PackagingArpita NaikNo ratings yet
- Id ProofDocument3 pagesId ProofSagar MittaNo ratings yet
- 19-CPS Student Induction Presentation-RAZAK FinalDocument18 pages19-CPS Student Induction Presentation-RAZAK FinalbaderNo ratings yet
- Blue Modern Medical Center Z-Fold BrochureDocument2 pagesBlue Modern Medical Center Z-Fold BrochureAngela Shaine CruzNo ratings yet
- Spoon University Nutrition GuideDocument80 pagesSpoon University Nutrition GuidermdelmandoNo ratings yet
- NCP Acute Pain Related To Tissue Ischemia As Manifested by Changes in Level of Consciousness PDFDocument3 pagesNCP Acute Pain Related To Tissue Ischemia As Manifested by Changes in Level of Consciousness PDFOGNTVNo ratings yet
- Noureen Zawar Family Medicine FMH Oct 21, 2017Document32 pagesNoureen Zawar Family Medicine FMH Oct 21, 2017Noureen ZawarNo ratings yet
- Health Promotion and MaintenanceDocument170 pagesHealth Promotion and Maintenancepopota100% (1)
- Pistachio Lush - An Easy One-Pan Dessert For Potlucks or Gatherings!Document2 pagesPistachio Lush - An Easy One-Pan Dessert For Potlucks or Gatherings!Citra RahmadhaniNo ratings yet
- New Mcgraw Hill EducationDocument132 pagesNew Mcgraw Hill EducationAlexandru LeșanNo ratings yet
- Bio MEMSDocument16 pagesBio MEMStronghuynh12No ratings yet
- Special Consideration in Neonatal Resuscitation - 2022 - Seminars in PerinatologDocument8 pagesSpecial Consideration in Neonatal Resuscitation - 2022 - Seminars in PerinatologEduardo Rios DuboisNo ratings yet
- Diare Pada BalitaDocument10 pagesDiare Pada BalitaYudha ArnandaNo ratings yet
- DNAlysis MygeneRx Sample ReportDocument13 pagesDNAlysis MygeneRx Sample ReportTrustNo ratings yet
- LESSON 1 - The Global North and SouthDocument9 pagesLESSON 1 - The Global North and SouthJeff Jeremiah PereaNo ratings yet
- Tdiagnostics - Telangana.gov - in ViewFiles - Aspx ReportId p1s0K7qAzHwmL8S2z98THgDocument3 pagesTdiagnostics - Telangana.gov - in ViewFiles - Aspx ReportId p1s0K7qAzHwmL8S2z98THgyesawovNo ratings yet
- Micro Rapid ReviewDocument6 pagesMicro Rapid ReviewEvan Miller100% (3)
- Attending Physicians Statement - Form CDocument1 pageAttending Physicians Statement - Form CPritam sarkarNo ratings yet
- Harga Obat Generik E-KatalogueDocument4 pagesHarga Obat Generik E-KatalogueMuhammad FarilNo ratings yet
- Alwyn Naicker - Research Proposal Assignment 2 PDFDocument15 pagesAlwyn Naicker - Research Proposal Assignment 2 PDFAlwyn NaickerNo ratings yet
- Nursing Care During Labor and BirthDocument7 pagesNursing Care During Labor and BirthRegine VillanuevaNo ratings yet
- Aurora Juliana Ariel - The QuestDocument215 pagesAurora Juliana Ariel - The QuestCristian Catalina100% (3)
- 9700 w14 QP 21Document16 pages9700 w14 QP 21rashmi_harryNo ratings yet