Professional Documents
Culture Documents
Fluid Properties 2: Prof. Balázs M. Fekete
Uploaded by
aceOriginal Title
Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
Fluid Properties 2: Prof. Balázs M. Fekete
Uploaded by
aceCopyright:
Available Formats
Fluid Properties 2
Prof. Balzs M. Fekete
Office: Steinman Room #188
Office Hours: Monday to Wednesday 11am 12:50pm
E-Mail: bfekete@ccny.cuny.edu
2017-02-06 CE 35000 Fluid Mechanics 1
Newtonian Versus Non-Newtonian Fluids
Fluids for which the shearing stress is The dimension of viscosity is FTL-2,
linearly related to the rate of shearing thus, in BG units viscosity is given as
strain (or rate of angular deformation ) lbf s/ft2 and in SI units as N s m-2 .
are designated as Newtonian fluids.
Viscosity is only mildly dependent on
Fluids for which the shearing stress is pressure and the effect of pressure is
not linearly related to the rate of usually neglected.
shearing strain are designated as non-
Newtonian fluids. For a particular fluid the viscosity is
also highly dependent on temperature.
The slope of shearing stress versus For example, as the temperature of
rate of shearing strain graph denoted water changes from 60 to 100 F the
as the apparent viscosity . density decreases by less than 1%, but
the viscosity decreases by about 40% .
For Newtonian fluids the apparent
viscosity is the same as the viscosity Viscosity of liquids decreases with an
and is independent of shear rate. increase in temperature, whereas for
gases an increase in temperature
causes an increase in viscosity.
2017-02-06 CE 35000 Fluid Mechanics 2
Non-Newtonian Fluids and
Their Characteristics
Shear thinning fluids (pseudo-plastic):
The apparent viscosity decreases with increasing
shear rate; the harder the fluid is sheared, the less
viscous it becomes.
Many colloidal suspension and polymer solution
are shear thinning. For instance, Latex paint does
not drip from the brush because the shear rate is
small and the apparent viscosity is large.
However, it flows smoothly onto the wall because
the thin layer of paint between the wall and the
brush causes a large shear rate and a small
apparent viscosity.
( )
n
du
= 0 , n<0
dy
Shear thickening fluids (Dilatant):
The apparent viscosity increases with increasing
shear rate; the harder the fluid is sheared, the more
viscose it becomes. Water-corn starch mixture and
du water-sand mixture (quicksand).
( )
= du n
dy = 0 , n>0
dy
2017-02-06 CE 35000 Fluid Mechanics 3
Non-Newtonian Behavior
Fluids for which shearing stress is not linearly related to the rate of shearing strain are
designated as non-Newtonian fluids.
A mixture of water and corn starch, when placed on a flat surface, flows as a thick, viscous
fluid. However, when the mixture is rapidly disturbed, it appears to fracture and behave more
like a solid. The mixture is a non-Newtonian shear thickening fluid which becomes more
viscous as the shearing rate is suddenly increased through the rapid action of the spoon.
2017-02-06 CE 35000 Fluid Mechanics 4
Bingham Plastic
These types of materials are neither fluid nor a solid. Such material can
withstand a finite, nonezero shear stress, the yield stress ( yield), without
motion (therefore, it is not a fluid), but once the yield stress is exceeded it
flows like a fluid material.
Toothpaste and mayonnaise are common examples of Bingham plastic
materials. For example, mayonnaise can sit in a pile on a slice of bread
(the shear stress less than the yield stress), but it flows smoothly into a
thin layer when the knife increases the stress above the yield stress.
2017-02-06 CE 35000 Fluid Mechanics 5
Effect of Temperature on Viscosity
For gases the Sutherland equation can For liquids an empirical equation that
be expressed as has been used is
B
3
T
CT 2 =D e
=
T +S where D and B are constants and T is
absolute temperature. This equation is
where C and S are empirical constants,
often called Andrade's equation. The
and T is absolute temperature. Thus, if
viscosity must be known at least for two
the viscosity is known at two
temperature so the two constant can be
temperature, C a n d S c a n b e
determined.
determined. Or, if more than two
viscosities are known, the data can be
correlated with previous equation using
some type of curve-fitting scheme.
2017-02-06 CE 35000 Fluid Mechanics 6
Bulk Modulus of Elasticity
Compressibility of the fluid (bulk modulus for short)
dp Ev Bulk Modulus (N m-2, Pa) [FL-2 or ML-1T-2]
E v =
dV /V dp Change in pressure (N m-2, Pa) [FL-2 or ML-1T-2]
V Volume (m3) [L3]
dV Change in Volume (m3) [L3]
dp
E v= Density (kg m-3) [ML-3]
d / d Change in density (kg m-3) [ML-3]
At 15C temperature 2.15 GPa (~21,500m water
depth) could compress a unit volume of water by 1%.
2017-02-06 CE 35000 Fluid Mechanics 7
Ideal Gas Law
p p Absolute pressure (N m-2, Pa) [FL-2 or ML-1T-2]
=R T Density (kg m-3) [ML-3]
Air R = 289.6 [J kg-1 K-1] T Absolute temperature (K) []
R Gas constant (J kg-1 K-1) [L2T-2-1]
Ru
R= Ru Universal gas constant (J kmol-1 K-1) [L2T-2-1]
Mm Mm Molecular mass (kmol g-1)
p
=constant isothermal cp
k=
p cv
k
=constant isentropic
cp Specific heat at constant p (J K-1) [ M L2T21]
cv Specific heat at constant V (J K-1) [ M L2T21]
2017-02-06 CE 35000 Fluid Mechanics 8
Bulk Modulus for Gases
dp dp
Isothermal E v= = = p
d / d
p dp
=C p=C =C
d
dp dp
Isentropic E v= = =kp
d / d
p k dp k 1
=C p=C =k C
k
d
2017-02-06 CE 35000 Fluid Mechanics 9
Tire Pump Example
A 30 l air at an absolute pressure of 101 kPa is
compressed isentropically to 15 l by the tire pump.
What is the final pressure?
pi pf f k
k
i
=
k
f
pf= ( )
i
pi
1.4
p f = ( 2 ) 101[ kPa] p f =266.54 [kPa]
2017-02-06 CE 35000 Fluid Mechanics 10
Speed of Sound
c Speed of sound (m s-1) [LT-1]
dp
c= dp Change in pressure (N m-2, Pa) [FL-2 or ML-1T-2]
d
Density (kg m-3) [ML-3]
Ev
c= Ev
R
Bulk modulus (Pa) [FL-2 or ML-1T-2]
Gas constant (J kg-1 K-1) [L2T-2-1]
T Absolute temperature (C) []
p
c= k c= k RT k The ratio of specific heat at constant pressure (cp)
And specific heat and constant volume (cv).
Air at 15 C k = 1.4 R = 289.6 [J kg-1 K-1] c = 341.8 [m s-1]
Water at 20 C Ev = 2.19 [GPa] = 998.2 [kg m-3] c = 1481 [m s-1]
Steel Y = 210 [GPa] (Young's) = 8050 [kg m-3] c = 5107 [m s-1]
2017-02-06 CE 35000 Fluid Mechanics 11
Speed of Sound
The characteristics of flow past an object depend strongly on whether the object is moving slower (subsonic) or faster (supersonic) than
the speed of sound.
The fact that the artillery shell shown is traveling faster than the speed of sound can be seen by noting the shock wave created by it. This
shock wave shows up as a thin line ahead of the shell and is bent back backwards as it wraps around the top and bottom of the shell to
form a blunt wedge shape. Across the shock waves the fluid properties (pressure, temperature, etc.) change considerably. The "sonic
boom" associated with supersonic flight is actually the pressure difference across the shock wave as it goes past the observer.
2017-02-06 CE 35000 Fluid Mechanics 12
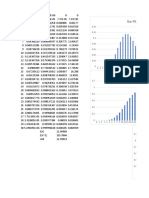
Speed of Sound vs. Temperature
Speed of Sound Mach
370 0.85
0.83
360
0.81
Speed of Sound [m/s]
Mach speed [Ma]
350 0.79
340 0.77
0.75
330 0.73
320 0.71
0.69
310
0.67
300 0.65
-50 -40 -30 -20 -10 0 10 20 30 40 50
Temperature [C]
Jet air craft traveling at 900 km hr-1
2017-02-06 CE 35000 Fluid Mechanics 13
Vapor Pressure
100
95
Boiling Temperature [C]
90
Vapor, pv 85
80
75
70
Liquid Liquid
65
0 1 2 3 4 5 6 7 8 9 10
Altitude [1000 m]
Boiling
Caisson disease
2017-02-06 CE 35000 Fluid Mechanics 14
Surface Tension
0.08
0.075
Surface Tension [N/m]
0.07
0.065
0.06
0.055
0.05
0 10 20 30 40 50 60 70 80 90 100
Temperature
http://en.wikipedia.org/wiki/Surface_tension
2017-02-06 CE 35000 Fluid Mechanics 15
Razor Blade
Surface tension forces exist at liquid/gas or liquid/solid interfaces. Although these forces are relatively small, they are quite important in
many situations.
A heavier-than-water, double-edged steel razor blade can float on water. Without surface tension, the blade would sink because its weight
is greater than its buoyant force. However, surface tension forces are not large enough to support a slightly heavier single-edged blade.
2017-02-06 CE 35000 Fluid Mechanics 16
Pressure Inside a Drop
Surface Tension (N m-1) [FL-1 or MT-2]
R Radius (m) [L]
pi Inside pressure (N m-2, Pa) [FL-2 or ML-1T-2]
pe External pressure (N m-2, Pa) [FL-2 or ML-1T-2]
p Pressure difference (N m-2, Pa) [FL-2 or ML-1T-2]
R
pe
pi 2 2
2 R = p R p= pi p e =
2 R
p R
2017-02-06 CE 35000 Fluid Mechanics 17
Water Strider
2017-02-06 CE 35000 Fluid Mechanics 18
Capillary Action
Munson; et al. 2012
2
Surface Tension (N m-1) [FL-1 or MT-2]
R h=2 R cos
R Radius (m) [L]
2 cos Angle of contact (0 for water 130 for mercury
h=
R Specific weight (N m-3) [FL-3 or ML-2T-2]
h Capillary rise or fall (m) [L]
2017-02-06 CE 35000 Fluid Mechanics 19
Capillary Rise
Although we do not normally think of surface tension effects as being important in the every-day flows that affect our lives, there are many
situations in which surface tension plays a critical role. Most of these situations involve a relatively small size scale.
The rise of a liquid in a small tube, such as the rise of sap in the small diameter channels within the trunk of a tree, is a result of the surface
tension forces developed at the liquid/gas interface within the tube or channel. The height of this capillary rise is inversely proportional to
diameter of the tube. This is shown by the rise of water in glass tubes of various diameters.
2017-02-06 CE 35000 Fluid Mechanics 20
Contact Angle
Surface tension is an important property when two fluids that do not easily mix are placed
next to each other (such as water and air). The surface tension is a force that acts on the
interface between the water and air due to that fact that the molecular attraction from
water molecules on one side of the interface is different from that of air molecules on the
other side.
Surface tension allows drops to maintain a hemispherical shape when placed on a flat
surface, as shown in the video. The contact angle of the drop is the angle that the drop
makes with the solid surface. This angle depends upon the surface tension. In the first
segment of the video, a water drop (colored orange) is placed on a plastic surface. The
contact angle is about 90 degrees. In the second segment, a (blue) drop of alcohol is
placed on the same surface. The lower surface tension of alcohol reduces the contact
angle and cause the alcohol drop to spread more than the water drop (the alcohol wets
the surface more easily). The third droplet is mercury on the same surface. The higher
surface tension of mercury produces less wetting (i.e. the droplet spreads less) and a
higher contact angle (past 90 degrees).
The effect of wetting can also be seen in a meniscus (the curved surface of a liquid
interface in a tube). Orange water wets the tube causing the fluid to rise to a higher
elevation at the wall. However, the much higher surface tension of mercury causes the
interface to be higher at the center of the tube than at the wall.
2017-02-06 CE 35000 Fluid Mechanics 21
Contact Angle
2017-02-06 CE 35000 Fluid Mechanics 22
Pressure at a Point
Munson; et al. 2012
y= s cos z= s sin
y z
p y p s = a y p z p s= ( a z + g )
2 2
x y z
F y = p y x z p s x s sin =2
ay x 0 y 0 z 0
x yz xyz
z z
F = p x y p s y s cos
2
g=
2
az p y= p s p z= p s
Pascal's law: The pressure at a point in a fluid at rest or in motion, is independent of direction as long as there
are no shearing stresses.
2017-02-06 CE 35000 Fluid Mechanics 23
You might also like
- Chapter 1Document41 pagesChapter 1masNo ratings yet
- MI 205 Luid Mechanics: B. K. GandhiDocument28 pagesMI 205 Luid Mechanics: B. K. GandhiAshutosh SinghNo ratings yet
- Characteristics of FluidDocument16 pagesCharacteristics of Fluid24. KrishNo ratings yet
- Lecture 2 - Chap 1Document15 pagesLecture 2 - Chap 1Muneer AhmedNo ratings yet
- Unit 1 Day 3 Class NotesDocument13 pagesUnit 1 Day 3 Class NotesSiddi Sampath Kumar ReddyNo ratings yet
- Lecture 2 - Marine Hydrodynamics I - Physical Properties of FluidsDocument25 pagesLecture 2 - Marine Hydrodynamics I - Physical Properties of FluidsKhalid BaragaNo ratings yet
- Chapter One: Fundamental Concepts inDocument29 pagesChapter One: Fundamental Concepts inNizar AliNo ratings yet
- Informe 6 ViscosityDocument7 pagesInforme 6 ViscosityJuan Jose Castro RicoNo ratings yet
- 48 - 25795 - ME362 - 2020 - 1 - 2 - 1 - Lecture 2 - Fluid 1 - Physical Properties of FluidsDocument29 pages48 - 25795 - ME362 - 2020 - 1 - 2 - 1 - Lecture 2 - Fluid 1 - Physical Properties of FluidsHussien El-masryNo ratings yet
- Chapter 1 NotesDocument10 pagesChapter 1 Notesmadesh1047No ratings yet
- MOF LectnotesDocument84 pagesMOF LectnotesKarar AL-DahlkiNo ratings yet
- 2) Fluid PropertiesDocument27 pages2) Fluid PropertiesAbdul AzizNo ratings yet
- E-Notes PDF All-Units 20052019025142PMDocument410 pagesE-Notes PDF All-Units 20052019025142PMVikram PanchalNo ratings yet
- Fluid Mechanics PDFDocument48 pagesFluid Mechanics PDFrakib hasanNo ratings yet
- Properties of Fluids by S K MondalDocument30 pagesProperties of Fluids by S K MondalLokesh DahiyaNo ratings yet
- Module1: Lecture 1 Basic Concepts and Properties of FluidsDocument18 pagesModule1: Lecture 1 Basic Concepts and Properties of FluidsasathishmctNo ratings yet
- 3.fluid Flow PhenomenaDocument19 pages3.fluid Flow PhenomenaMAHLATSE MULALANo ratings yet
- Fluid Mechanics & Machines Part 1 PDFDocument96 pagesFluid Mechanics & Machines Part 1 PDFsushantNo ratings yet
- PE-209 Lecture-01Document34 pagesPE-209 Lecture-01Adeem AbbasNo ratings yet
- F C N C: Luid Definition and PropertiesDocument25 pagesF C N C: Luid Definition and PropertiesAjay VictorNo ratings yet
- Unit 5: RheologyDocument71 pagesUnit 5: RheologySundar SkNo ratings yet
- (Chapter 1) Fluid Mechanics For Mechanical EngineeringDocument38 pages(Chapter 1) Fluid Mechanics For Mechanical EngineeringAnn Razon0% (1)
- Fluid Mechanics and Fluid Machines IES 2009 GATE 2009Document308 pagesFluid Mechanics and Fluid Machines IES 2009 GATE 2009juan.lNo ratings yet
- Fluid Flow Operations Lecture 1: Introduction: Dr. Srinivas Krishnaswamy Assistant Professor Chemical EngineeringDocument22 pagesFluid Flow Operations Lecture 1: Introduction: Dr. Srinivas Krishnaswamy Assistant Professor Chemical EngineeringPRAJWAL RASTOGINo ratings yet
- Fluid Mechanics 1 ME 321: Dr. M. Ehtisham SiddiquiDocument23 pagesFluid Mechanics 1 ME 321: Dr. M. Ehtisham SiddiquiMahtab IlyasNo ratings yet
- Properties of Fluid: Olids and LuidsDocument14 pagesProperties of Fluid: Olids and LuidsipsitabhattacharyaNo ratings yet
- FM & F Machines 2013 PDFDocument355 pagesFM & F Machines 2013 PDFChandan KumarNo ratings yet
- FM Sample Questions SK MondalDocument372 pagesFM Sample Questions SK Mondalarobin23No ratings yet
- Fluid Mechanics and MachineryDocument6 pagesFluid Mechanics and MachineryBiswadeep Roy ChoudhuryNo ratings yet
- Fluid Mechanics & Fluid MachinesDocument355 pagesFluid Mechanics & Fluid MachinesSatish Kumar100% (6)
- Fluid C1 03 (1) StudentDocument24 pagesFluid C1 03 (1) Studenteric0910160212No ratings yet
- Chpter 2Document36 pagesChpter 2Francis Ian VillacicaNo ratings yet
- Notes of LessonDocument52 pagesNotes of LessonRichards Ðånte WisdomNo ratings yet
- Ammar Hafez Presentation SP2019Document66 pagesAmmar Hafez Presentation SP2019Sunita ChayalNo ratings yet
- 01-Lab 03-Redwoods ViscometerDocument22 pages01-Lab 03-Redwoods ViscometerZero MakerNo ratings yet
- Exp2 Viscosity of Crude OilDocument20 pagesExp2 Viscosity of Crude Oilozknbki.gnydnNo ratings yet
- FLUID MECHANICS 2 Marks QuestionDocument4 pagesFLUID MECHANICS 2 Marks Questionrahul singhNo ratings yet
- Fluid Mechanics FUOYE MEE206 NoteDocument60 pagesFluid Mechanics FUOYE MEE206 NoteSILAS OMONIYINo ratings yet
- Mechanical FluidDocument3 pagesMechanical Fluidshanoon77No ratings yet
- Fluid Mech MarathDocument39 pagesFluid Mech MarathRajneesh KumarNo ratings yet
- Covenant University Nigeria: Tutorial Kit Omega SemesterDocument21 pagesCovenant University Nigeria: Tutorial Kit Omega SemesterthareendaNo ratings yet
- CE2202 Fluid Mechanics NOTESDocument0 pagesCE2202 Fluid Mechanics NOTESGp ChengNo ratings yet
- Lecture 1Document7 pagesLecture 1Tharusha ImalkaNo ratings yet
- Lecture 1: Introduction To Fluid MechanicsDocument26 pagesLecture 1: Introduction To Fluid MechanicsBenidiktus LovianwicahyoNo ratings yet
- Section ADocument14 pagesSection ADarshak KhuntNo ratings yet
- Unit 1Document76 pagesUnit 1ankurparekh2810No ratings yet
- Chapter 1 - Intorduction To Fluid MechanicDocument18 pagesChapter 1 - Intorduction To Fluid MechanicHAFIDZ SYAHMI BIN FAZNEE ANIFFNo ratings yet
- Chapter1 Introduction 1Document37 pagesChapter1 Introduction 1smaras123No ratings yet
- FOOD2003 Food Engineering Rheology: ViscosityDocument14 pagesFOOD2003 Food Engineering Rheology: ViscosityMaria Fernanda Bustamante LayedraNo ratings yet
- CIV 242 Fluid Lec 2 PDFDocument17 pagesCIV 242 Fluid Lec 2 PDFMahaNo ratings yet
- What Are The Newtonian Liquids? - Computer Aided Design & The 118 ElementsDocument8 pagesWhat Are The Newtonian Liquids? - Computer Aided Design & The 118 ElementsEmNo ratings yet
- Hlc-Ii: T P, T T P P T T P P P E Bulk Modulus (Reciprocal of TheDocument5 pagesHlc-Ii: T P, T T P P T T P P P E Bulk Modulus (Reciprocal of ThessriramiitNo ratings yet
- Viscosity of Newtonian and Non-Newtonian Liquids (Rotary Viscometer)Document3 pagesViscosity of Newtonian and Non-Newtonian Liquids (Rotary Viscometer)Thiago SantosNo ratings yet
- Fluids 01 02 PDFDocument10 pagesFluids 01 02 PDFChristian LeeNo ratings yet
- Fluid: Flowing Deforms Continuously External Shearing ForceDocument33 pagesFluid: Flowing Deforms Continuously External Shearing ForcePaul PaulNo ratings yet
- UNit1FFO - PDF (Compatibility Mode)Document122 pagesUNit1FFO - PDF (Compatibility Mode)Pranil KarangaleNo ratings yet
- Compressible Flow PDFDocument38 pagesCompressible Flow PDFApple EmiratessNo ratings yet
- Indian Institute of Technology Guwahati: Fluid MechanicsDocument16 pagesIndian Institute of Technology Guwahati: Fluid MechanicsForam ParekhNo ratings yet
- Fluid Mechanics Chapter1.2 (Rev)Document18 pagesFluid Mechanics Chapter1.2 (Rev)f_ei81No ratings yet
- InstructionDocument1 pageInstructionaceNo ratings yet
- Syllabus 44100 Fall 2017Document2 pagesSyllabus 44100 Fall 2017aceNo ratings yet
- Prob HW For 327Document7 pagesProb HW For 327aceNo ratings yet
- SDP ScheduleDocument1 pageSDP ScheduleaceNo ratings yet
- The Goal of This Case Is To Designate 2 Health Centers To Two Locations in Such A Manner That Average Response Times To Medical Emergencies Is MinimizedDocument1 pageThe Goal of This Case Is To Designate 2 Health Centers To Two Locations in Such A Manner That Average Response Times To Medical Emergencies Is MinimizedaceNo ratings yet
- Test 1 ADocument4 pagesTest 1 AaceNo ratings yet
- Prob HW For 327Document2 pagesProb HW For 327aceNo ratings yet
- Vancouver Paper Torsion v2Document7 pagesVancouver Paper Torsion v2musher95No ratings yet
- Lectures 7 - 02.27.2017Document17 pagesLectures 7 - 02.27.2017aceNo ratings yet
- Mechanics Formula SheetDocument5 pagesMechanics Formula SheetWong Jian MingNo ratings yet
- Rules For Ce 231 Exam 1 Sept 28 2016Document1 pageRules For Ce 231 Exam 1 Sept 28 2016aceNo ratings yet
- 4.6-4.7, 4.12 Members Made of Different Materials, Stress Concentrations, Eccentric LoadingDocument4 pages4.6-4.7, 4.12 Members Made of Different Materials, Stress Concentrations, Eccentric LoadingaceNo ratings yet
- Ecological Pyramids WorksheetDocument5 pagesEcological Pyramids Worksheetace0% (1)
- Quiz 3 TrussDocument1 pageQuiz 3 TrussaceNo ratings yet
- Physics Review SheetDocument18 pagesPhysics Review Sheetnetsam_flixsamNo ratings yet
- Thermodynamics FinalDocument7 pagesThermodynamics FinalaceNo ratings yet
- CH 3 SummaryDocument2 pagesCH 3 SummaryaceNo ratings yet
- P. G. Francis (Auth.) - Mathematics For ChemisDocument202 pagesP. G. Francis (Auth.) - Mathematics For Chemisjunaid0% (1)
- Crystal Field PotentialDocument3 pagesCrystal Field PotentialDubhe3No ratings yet
- King Fahd University of Petroleum & Minerals Mechanical Engineering Department Thermodynamics I - ME 203Document1 pageKing Fahd University of Petroleum & Minerals Mechanical Engineering Department Thermodynamics I - ME 2033bdul78No ratings yet
- PDB - Single R32Document72 pagesPDB - Single R32Duy ChuNo ratings yet
- Example Using The New Model Model With BootDocument9 pagesExample Using The New Model Model With BootMaryJane Ayisha Sado-ObahNo ratings yet
- PCS130 Magnetic Fields 5Document8 pagesPCS130 Magnetic Fields 5Ekan Ahmad100% (1)
- Orbital Parameters of Satellite by Karan Singh PariharDocument21 pagesOrbital Parameters of Satellite by Karan Singh Pariharkaran singh pariharNo ratings yet
- Audio Spotlighting NewDocument30 pagesAudio Spotlighting NewAnil Dsouza100% (1)
- Ce1013 09Document42 pagesCe1013 09Durga SharmaNo ratings yet
- Physical Science Module 1 For Students.Document10 pagesPhysical Science Module 1 For Students.YUH JBWDHGNo ratings yet
- Chapter - 21 Electromagnetic InductionDocument21 pagesChapter - 21 Electromagnetic InductionNafees FarheenNo ratings yet
- Spreading Dynamics of Polymer Nanodroplets: Sandia National Laboratories, Albuquerque, New Mexico 87185, USADocument10 pagesSpreading Dynamics of Polymer Nanodroplets: Sandia National Laboratories, Albuquerque, New Mexico 87185, USAmh123456789No ratings yet
- GTE Micro Project 4th SemDocument6 pagesGTE Micro Project 4th SemNishikant Bhure100% (3)
- VCSELsDocument48 pagesVCSELsmlogan_22No ratings yet
- MD1-0-E-505!06!00001 Generator Transformer Sizing CalculationDocument19 pagesMD1-0-E-505!06!00001 Generator Transformer Sizing Calculationtvpham12350% (2)
- ADOMIAN Decomposition Method For Solvin1Document18 pagesADOMIAN Decomposition Method For Solvin1Susi SusilowatiNo ratings yet
- Exploring Science International ContentsDocument4 pagesExploring Science International ContentsR S67% (6)
- Subject: Quality Management: Reliability Evaluation ModelsDocument8 pagesSubject: Quality Management: Reliability Evaluation ModelsMayank kumawatNo ratings yet
- Effects of Dry Density and Grain Size Distribution On Soil-Water Characteristic Curves of Sandy SoilsDocument12 pagesEffects of Dry Density and Grain Size Distribution On Soil-Water Characteristic Curves of Sandy SoilsmazharNo ratings yet
- Physics 5054/3: MCM Nkana Secondary SchoolDocument5 pagesPhysics 5054/3: MCM Nkana Secondary SchoolJedediah PhiriNo ratings yet
- Inductor and Flyback Transformer DesignDocument20 pagesInductor and Flyback Transformer Designbmmostefa100% (1)
- 9.2 Quantum Theory and The AtomDocument31 pages9.2 Quantum Theory and The AtomRyuusukeNo ratings yet
- Electric and Magnetic States of Matter MagnetismDocument18 pagesElectric and Magnetic States of Matter MagnetismNittam ThakurNo ratings yet
- 06 PlateTheory 08 VibrationsDocument6 pages06 PlateTheory 08 Vibrationsvhj gbhjNo ratings yet
- Concept Check Answers Ch6Document5 pagesConcept Check Answers Ch6Ruben Abreu100% (1)
- A Practical 3D Bounding Surface Plastic Sand Model For Geotechnical Earthquake Engineering ApplicationDocument11 pagesA Practical 3D Bounding Surface Plastic Sand Model For Geotechnical Earthquake Engineering ApplicationMiguel Angel LuceroNo ratings yet
- Determination and Enlarging of The Acoustic Sweet-Spot in An AuditoriumDocument4 pagesDetermination and Enlarging of The Acoustic Sweet-Spot in An AuditoriumInternational Journal of Innovative Science and Research TechnologyNo ratings yet
- TyreCollo2015 GipserDocument30 pagesTyreCollo2015 GipserKoushik SivasubramaniamNo ratings yet
- D M Bose and Cosmic Ray Research: S C Roy and Rajinder SinghDocument18 pagesD M Bose and Cosmic Ray Research: S C Roy and Rajinder Singhdwi johdiNo ratings yet
- Overview:: Book Title:-Engineering Physics, 2E Author:-B. K. Pandey - SDocument2 pagesOverview:: Book Title:-Engineering Physics, 2E Author:-B. K. Pandey - SAvadootha Rajesh NethaNo ratings yet