Professional Documents
Culture Documents
(PFR) : X: FM and The Conversion X Dfa - Fadx
(PFR) : X: FM and The Conversion X Dfa - Fadx
Uploaded by
Naveen Kharb0 ratings0% found this document useful (0 votes)
5 views2 pageshfthdtrt thfg
Original Title
Dfu Bref Ghb
Copyright
© © All Rights Reserved
Available Formats
PDF, TXT or read online from Scribd
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this Documenthfthdtrt thfg
Copyright:
© All Rights Reserved
Available Formats
Download as PDF, TXT or read online from Scribd
0 ratings0% found this document useful (0 votes)
5 views2 pages(PFR) : X: FM and The Conversion X Dfa - Fadx
(PFR) : X: FM and The Conversion X Dfa - Fadx
Uploaded by
Naveen Kharbhfthdtrt thfg
Copyright:
© All Rights Reserved
Available Formats
Download as PDF, TXT or read online from Scribd
You are on page 1of 2
2.3.
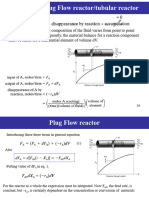
2 Tubular Flow Reactor (FFR)
We model the tubular reactor as having the fluid flowing in plug flow. i.e.. nr
radial gradients in concentration, temperature, or reaction rate.' As the reac
[ant$ enter and flow axially down the reactor, [hey are consumed and the con
version increases along the length of the reactor. To develop the PFR desigr
equation we first multiply both sides of the tubular reactor design equatior
(1- 123 by - I . We then express the mole balance equation for species A in tht
reaction as
For a flow system, FA has previously been given in terms of the entering molat
Row rare FM and the conversion X
differentiating
dFA = -FAdX
and substituting into (7-141, gives the differential form of the design equation
for a plug-flow reactor (PFR):
blgn 4 PFR b, We now separate the variables and integrate with the limits V = 0 when X = 0
cquntlon to obtain the plug-flow reactor voluine necessary to achieve a specified conversion
X:
To carry out the integrations in the batch and plug-Row reactor design
equations (2-9) and (2-16). as welI as to evaluate the CSTR design equation
(2-23), we need to know how the reaction rate - r ~var ies with the concentration
(hence conversion) of the reacting species. This relationship between reaction
rate and concentration is developed in Chapter 3.
This constraint will be removed when we extend our analysis to nonideal (industrial)
reactors in Chapters 13 rind Id.
Sm. 2.4 Apolicatiors cl ths Design Equaticns for Continuous-Flow Fleactors 45
PBR design
equation
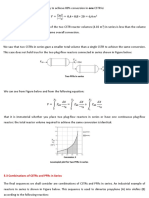
2.3.3 Packed-Bed Reactor
Packed-bed reactors are tubular reactors filled with catalyst particles. The drrivation
of the differential and integral forms of the design equations For
packed-bed reactors are analogous 10 those for a PFR {cf. Equations (2- 15) and
(2- 1611. That is, substituting Equation (2- 12) for FA in Equation ( 1 - 15) gives
The differential Form of the design equation [i.e., Equation (2-17)J must be
used when analyzing reactors that have a pressure drop along the length of the
reactor. We discuss pressure drop in packed-bed reactors in Chapter 4.
In the abserlce of pressure drop, i.e., AP = 0. we can integrate (2- 17)
with Iimits X = 0 at W = 0 to obtain
Equation (2-181 can be used to determine the catalyst weight W necessary to
achieve a conversion X when the total pressure remains constant.
2.4 Applications of the Design Equations
for Continuous-Flow Reactors
In this section. we are going ro show how we can size CSTRs and PFRs (i.e.,
determine their reactor volumes) from knowledge of rbe rate of reaction. -r,.
as n function of conversion, X. The rate of disappearance of A. -r,. is almost
aIways a function of the concentrations of the various species present. When
only one reaction is occurring. each of the concentrations can be expressed as
a function of the conversion X (see Chapter 3); consequently, -r, can be
expressed as a function of X.
A particularly simple functional dependence, yet one that occurs often, is
the first-order dependence
Here. k is the specific reaction rate and is a function only of temperature, and
CA0 is the entering concentration. We note in Equations (2-13) and (2-16) the
reactor volume in a function of the reciprocal of -r,. For this first-order dependence,
a plot of the reciprocal rate of reaction (I/-r,) as a function of conversion
yields a curve similar to the one shown in Figure 2-1, where
46 Conversion
You might also like
- Gas Turbine Training MaterialDocument141 pagesGas Turbine Training MaterialVikas Yerramsetty100% (9)
- Effect PulseDocument33 pagesEffect PulseMohd Sharu Mamat100% (1)
- Medical CertificateDocument2 pagesMedical CertificateNaveen Kharb100% (1)
- Feed PurificationDocument12 pagesFeed PurificationrajuNo ratings yet
- 150 KLPD MEE Mass BalanceDocument8 pages150 KLPD MEE Mass BalancePrathmesh Gujarati100% (5)
- Conversion FouglrDocument21 pagesConversion Fouglrحسام العسلNo ratings yet
- The Limiting Reactant: Lecture 4 /course2 Reactor Design Design Equations For ReactorsDocument16 pagesThe Limiting Reactant: Lecture 4 /course2 Reactor Design Design Equations For Reactorsزهرة ZaraNo ratings yet
- Bioprocess Assignment Derive EquationsDocument12 pagesBioprocess Assignment Derive EquationsRicky LeeNo ratings yet
- Cre - 1Document10 pagesCre - 1dhavalNo ratings yet
- Reactors in ParallelDocument3 pagesReactors in ParallelAdrián D. Rodríguez100% (2)
- Plug Flow Reactor NotesDocument4 pagesPlug Flow Reactor Notesahmad pidotNo ratings yet
- 2 Conversion and Reactor SizingDocument39 pages2 Conversion and Reactor SizingRonaldo Luis Guao BolañoNo ratings yet
- Performance Equations For ReactorsDocument53 pagesPerformance Equations For ReactorsGuhan KANo ratings yet
- CRE Chapter 4-Design For Single Reactions-K192 Part 4.2Document17 pagesCRE Chapter 4-Design For Single Reactions-K192 Part 4.2Huy Huu HuynhNo ratings yet
- Week 6 Chem R Eng 1 09-01-23Document15 pagesWeek 6 Chem R Eng 1 09-01-23Zain Ul AbedinNo ratings yet
- Conversion and Reactor SizingDocument40 pagesConversion and Reactor SizingsiskieoNo ratings yet
- KRD Chapter 2Document39 pagesKRD Chapter 2Reyhan97No ratings yet
- TFR ReportDocument23 pagesTFR ReportJessie Ctiffany EveNo ratings yet
- Non Ideal ReactorsDocument17 pagesNon Ideal Reactorserjayenv7346No ratings yet
- Sir Lal Presentation - pdf1Document19 pagesSir Lal Presentation - pdf1SagheerSiddiquiHydNo ratings yet
- Chemical Reactor Technology Chapter 2Document29 pagesChemical Reactor Technology Chapter 2Salman AlshammariNo ratings yet
- UNIT-3: Department of Chemical Engineering Sathyabama UniversityDocument16 pagesUNIT-3: Department of Chemical Engineering Sathyabama UniversityBT20CME033 Gautam TahilyaniNo ratings yet
- Dispersion ModelDocument9 pagesDispersion ModelBrahma Teja LbNo ratings yet
- CHEE 321: Chemical Reaction Engineering 2. Isothermal Reactor Design Single Reaction in Batch, CSTR, PFR 2a. Levenspiel Plots (Reactor Sizing)Document12 pagesCHEE 321: Chemical Reaction Engineering 2. Isothermal Reactor Design Single Reaction in Batch, CSTR, PFR 2a. Levenspiel Plots (Reactor Sizing)Pranav NakhateNo ratings yet
- Theory - Mixed Flow ReactorDocument4 pagesTheory - Mixed Flow ReactorbaimchemicalNo ratings yet
- Reactores Com RecicloDocument5 pagesReactores Com RecicloAniceto Salomão MachavaNo ratings yet
- CMC091Y1092 Chapter 2Document43 pagesCMC091Y1092 Chapter 2王暐翔No ratings yet
- Plug Flow Reactor ModelDocument4 pagesPlug Flow Reactor ModelMagesh kumarNo ratings yet
- Conversion and Reactor SizingDocument27 pagesConversion and Reactor SizingJam imtiazNo ratings yet
- Chapter 3: Ideal Plug Flow Reactors Design: by Dr. Ramesh RedrouthuDocument25 pagesChapter 3: Ideal Plug Flow Reactors Design: by Dr. Ramesh RedrouthuTsega IsraelNo ratings yet
- Lecture-16: The Ideal Plug Flow ReactorDocument7 pagesLecture-16: The Ideal Plug Flow ReactorGanesh Chandra BaroNo ratings yet
- Experiment 1B - Tubular ReactorDocument14 pagesExperiment 1B - Tubular ReactorNajmul Puda PappadamNo ratings yet
- Week 2Document37 pagesWeek 2HAJEYYNo ratings yet
- CSTRDocument27 pagesCSTRLeslie AmadorNo ratings yet
- Chapter 2 Conversion and Reactor SizingDocument37 pagesChapter 2 Conversion and Reactor SizingAmir NasrinNo ratings yet
- PFRDocument19 pagesPFRKangae IlhamNo ratings yet
- fcc1 PDFDocument7 pagesfcc1 PDFAnonymous jlLBRMAr3ONo ratings yet
- Tubular Reactor bp101bDocument17 pagesTubular Reactor bp101bMuhamad Hafifi AjwadNo ratings yet
- P F RDocument4 pagesP F RMohamed Ragab MohamedNo ratings yet
- CH142L - Experiment 1Document17 pagesCH142L - Experiment 1Allyssa BadilloNo ratings yet
- Reactor of Chemical Engineering: Presented by Nazarudin, PHDDocument69 pagesReactor of Chemical Engineering: Presented by Nazarudin, PHDAsmi RamadhaniNo ratings yet
- FTW1Document52 pagesFTW1pubgkrpg09No ratings yet
- CSTR FinalDocument36 pagesCSTR FinalMuhammad Yar KhanNo ratings yet
- Chapter 3Document13 pagesChapter 3Kev AdiNo ratings yet
- Week 9 Chem R Eng (2) 01-02-2023Document6 pagesWeek 9 Chem R Eng (2) 01-02-2023Zain Ul AbedinNo ratings yet
- Keywords: Steam Reforming, Dynamics, Hydrogen, Fuel Cell, SimulationDocument7 pagesKeywords: Steam Reforming, Dynamics, Hydrogen, Fuel Cell, SimulationMuhammad HarisNo ratings yet
- Kin 6Document126 pagesKin 6fireorion7No ratings yet
- Report With ConclusionDocument30 pagesReport With ConclusionAhmed YounisNo ratings yet
- Optimization of Output Fluctuation For The Model of Non-Ideal CSTR'S in Series With A Sinusoidal Input of ConcentrationDocument7 pagesOptimization of Output Fluctuation For The Model of Non-Ideal CSTR'S in Series With A Sinusoidal Input of ConcentrationNIKHIL SHINDENo ratings yet
- Report PFRDocument21 pagesReport PFRMuhamad Hafifi Ajwad100% (2)
- CH 2Document22 pagesCH 2林哲璋No ratings yet
- Reactors: CHML 404-01 Date: 03/11/2019 Unit Operation: 1 Instructor: Dr. MartinezDocument23 pagesReactors: CHML 404-01 Date: 03/11/2019 Unit Operation: 1 Instructor: Dr. MartinezKevin ThomasNo ratings yet
- FullDocument33 pagesFullEja RotiKeju100% (2)
- Chapter 4Document49 pagesChapter 4Ibrahim Al-HammadiNo ratings yet
- Lecture 3 - Reactor SizingDocument41 pagesLecture 3 - Reactor SizingImran AzmanNo ratings yet
- Single Phase Switching Parameters For Untransposed EHV TLDocument12 pagesSingle Phase Switching Parameters For Untransposed EHV TLCarlos Lino Rojas AgüeroNo ratings yet
- BNBBHBHBHJBJHDocument25 pagesBNBBHBHBHJBJHZati TarhiziNo ratings yet
- CRE1 PAPER - Alif - Farhandi - HannahDocument53 pagesCRE1 PAPER - Alif - Farhandi - HannahFarhandi Fadillah FedrizalNo ratings yet
- Flowing Reactors Continuous Stirred Tank Reactors CstrsDocument3 pagesFlowing Reactors Continuous Stirred Tank Reactors CstrsUkies LNo ratings yet
- Book 13328 PDFDocument23 pagesBook 13328 PDFadilla nitaaNo ratings yet
- Power Electronics Applied to Industrial Systems and Transports, Volume 3: Switching Power SuppliesFrom EverandPower Electronics Applied to Industrial Systems and Transports, Volume 3: Switching Power SuppliesNo ratings yet
- Analysis and Design of Multicell DC/DC Converters Using Vectorized ModelsFrom EverandAnalysis and Design of Multicell DC/DC Converters Using Vectorized ModelsNo ratings yet
- Power Electronics Applied to Industrial Systems and Transports, Volume 2: Power Converters and their ControlFrom EverandPower Electronics Applied to Industrial Systems and Transports, Volume 2: Power Converters and their ControlRating: 5 out of 5 stars5/5 (1)
- PDF NDA Batches For Upload Telegram 09 Jan 13Document10 pagesPDF NDA Batches For Upload Telegram 09 Jan 13Naveen KharbNo ratings yet
- SSB Interviews 10+2 Tes-29 Jul 2013 Course in Army Selection Centre Central, Bhopal (Updated As On 03 Jan 2013 TELE NO 0755-2702223 FAX 0755-2735980Document10 pagesSSB Interviews 10+2 Tes-29 Jul 2013 Course in Army Selection Centre Central, Bhopal (Updated As On 03 Jan 2013 TELE NO 0755-2702223 FAX 0755-2735980Naveen KharbNo ratings yet
- Addendum 21032016Document1 pageAddendum 21032016Naveen KharbNo ratings yet
- MainMenu3NDA 130UPDATEDASON14JAN2013Document20 pagesMainMenu3NDA 130UPDATEDASON14JAN2013Naveen KharbNo ratings yet
- Jngjenjgd Ertjnhjenh Tjbnjbnjy Yhnjynhj Tnn15184841 Dthnh555Document1 pageJngjenjgd Ertjnhjenh Tjbnjbnjy Yhnjynhj Tnn15184841 Dthnh555Naveen KharbNo ratings yet
- (4 5) 1D SS Conduction Part1 PDFDocument24 pages(4 5) 1D SS Conduction Part1 PDFNaveen KharbNo ratings yet
- Lesson 2 HRMDocument21 pagesLesson 2 HRMNaveen KharbNo ratings yet
- CCI Control Valves For Fossil ApplicationsDocument2 pagesCCI Control Valves For Fossil ApplicationsGabrieldiazNo ratings yet
- Optimized Design of A Bagasse Dryer System For Sugar IndustryDocument5 pagesOptimized Design of A Bagasse Dryer System For Sugar IndustryBONFRINGNo ratings yet
- LNG and CNGDocument2 pagesLNG and CNGAli ImranNo ratings yet
- A Sabic - Clariant Partnership Company Confidential Information of Scientific Design Company, IncDocument28 pagesA Sabic - Clariant Partnership Company Confidential Information of Scientific Design Company, IncarifirminoNo ratings yet
- Rapid Sand Filtration DetailedDocument13 pagesRapid Sand Filtration DetailedAbhijith SPNo ratings yet
- Sewage Disposal and Treatment Lecture 1Document3 pagesSewage Disposal and Treatment Lecture 1Tigist Taddese Haile100% (1)
- Booklet NBG, Nbge, NKG, NkgeDocument45 pagesBooklet NBG, Nbge, NKG, NkgeThanh Van LeNo ratings yet
- Airbag Inflator Models in LS-DYNADocument4 pagesAirbag Inflator Models in LS-DYNAAmir IskandarNo ratings yet
- Homework BDocument28 pagesHomework BPravallika Kollipara0% (2)
- 501962-Gate Valves RFQ - Rev.ADocument8 pages501962-Gate Valves RFQ - Rev.AvalveNo ratings yet
- CHEMISTRYDocument26 pagesCHEMISTRYDarshan SahuNo ratings yet
- Mechanical Optional Main by ByjusDocument5 pagesMechanical Optional Main by Byjusayushgupta304No ratings yet
- Notesonhydraulic00owenrich DjvuDocument1,415 pagesNotesonhydraulic00owenrich DjvuAditya KumarNo ratings yet
- HW 3Document7 pagesHW 3Shashwat Chakraborti0% (1)
- Schx4007 Mass Transfer LabDocument60 pagesSchx4007 Mass Transfer LabAhmed AliNo ratings yet
- Stress Analysis Methods For Underground Pipe Lines Part 1 - Basic CalculationsDocument5 pagesStress Analysis Methods For Underground Pipe Lines Part 1 - Basic Calculationskumaran__k100% (3)
- Department of Fluid Mechanics: Delhi Technological UniversityDocument10 pagesDepartment of Fluid Mechanics: Delhi Technological University2K20-ME-161 Mridul AnandNo ratings yet
- Notes DJJ20073 - Fluid Mechanics - Suzilawati Alias PDFDocument74 pagesNotes DJJ20073 - Fluid Mechanics - Suzilawati Alias PDFAisyah SorayaNo ratings yet
- Introduction To Numerical Methods in Chemical Engineering - P. Ahuja PDFDocument99 pagesIntroduction To Numerical Methods in Chemical Engineering - P. Ahuja PDFSarath RNo ratings yet
- 3 Well ProblemsDocument47 pages3 Well ProblemsKarwan DilmanyNo ratings yet
- Incomplete CombustionDocument5 pagesIncomplete CombustionDineshkumarNo ratings yet
- Reservoir Fluid Sampling (Lulav)Document19 pagesReservoir Fluid Sampling (Lulav)Lulav BarwaryNo ratings yet
- New Developments in Hydroprocessing: TopicDocument43 pagesNew Developments in Hydroprocessing: TopicSinh LeNo ratings yet
- ( (Diffusion) ) : Chapter (10) in VolumeDocument9 pages( (Diffusion) ) : Chapter (10) in Volumeashwaq100% (1)
- Ped 13.7 13.7.2Document15 pagesPed 13.7 13.7.2babis1980No ratings yet
- Contoh Itp Mechanical PltuDocument2 pagesContoh Itp Mechanical PltuyusufNo ratings yet
- Diffusion Media FF-500 G (M5) : - Full Penetration of Special Adhesive - Gradient Density StructureDocument2 pagesDiffusion Media FF-500 G (M5) : - Full Penetration of Special Adhesive - Gradient Density StructureDorian HernandezNo ratings yet