Professional Documents
Culture Documents
Gate Control Method
Uploaded by
Enniah Mlalazi0 ratings0% found this document useful (0 votes)
9 views2 pagesThe Gate Control Model proposes that pain signals enter the spinal cord from the body and are transmitted to the brain. Additional pathways from the spinal cord can open or close a "gate" that determines the strength of the pain signal to the brain. The gate is located in the substantia gelatinosa in the spinal cord. The gate closes from large-fiber tactile signals or signals from the brain, decreasing pain perception, while small pain fiber signals open the gate, increasing pain.
Original Description:
Original Title
Gate control method.docx
Copyright
© © All Rights Reserved
Available Formats
DOCX, PDF, TXT or read online from Scribd
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentThe Gate Control Model proposes that pain signals enter the spinal cord from the body and are transmitted to the brain. Additional pathways from the spinal cord can open or close a "gate" that determines the strength of the pain signal to the brain. The gate is located in the substantia gelatinosa in the spinal cord. The gate closes from large-fiber tactile signals or signals from the brain, decreasing pain perception, while small pain fiber signals open the gate, increasing pain.
Copyright:
© All Rights Reserved
Available Formats
Download as DOCX, PDF, TXT or read online from Scribd
0 ratings0% found this document useful (0 votes)
9 views2 pagesGate Control Method
Uploaded by
Enniah MlalaziThe Gate Control Model proposes that pain signals enter the spinal cord from the body and are transmitted to the brain. Additional pathways from the spinal cord can open or close a "gate" that determines the strength of the pain signal to the brain. The gate is located in the substantia gelatinosa in the spinal cord. The gate closes from large-fiber tactile signals or signals from the brain, decreasing pain perception, while small pain fiber signals open the gate, increasing pain.
Copyright:
© All Rights Reserved
Available Formats
Download as DOCX, PDF, TXT or read online from Scribd
You are on page 1of 2
Gate control method
The Gate Control Model
The gate control model begins with the idea that pain signals
enter the spinal cord from the body and are then transmitted
from the spinal cord to the brain. In addition, the
model proposes that there are additional pathways that
influence the signal sent from the spinal cord to the brain.
The central idea behind the theory is that the signals from
these additional pathways can act to open or close a gate,
located in the spinal cord, which determines the
VL 3 strength of the signal leaving the spinal cord.
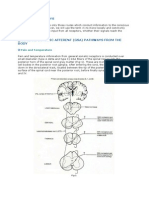
Figure 14.23 shows the circuit that Melzack and Wall
(1965) proposed. The gate control system consists of cells
in the spinal cord called the substantia gelatinosa (Figure
14.23a). These cells are represented by SG_ and SG_ in the
gate control circuit in Figure 14.23b. We can understand
how this circuit functions by considering the following
facts about its operation.
Input to the gate control system occurs along three
pathways:
■ S-fibers. The small-diameter fibers (S-fibers) are associated
with nociceptors—fibers or receptors that fire
to damaging and potentially painful stimuli. Activity
in the S-fibers increases the activity of the transmis-
Pain 345
T-cell
Pain
Gate-control system
SG–
Central
control
Gate closes
Gate opens
SG+ +
+
+
+
+
+
–
L-fiber
(a)
(b)
S-fiber
(from
nociceptors)
Dorsal root
Substantia gelatinosa
Figure 14.23 ❚ (a) Cross section of the spinal cord showing
fibers entering through the dorsal root and the location of the
substantia gelatinosa. (b) The circuit proposed by Melzack
and Wall (1965, 1988) in their gate control model of pain
perception. See text for details.
sion cell (T-cell). The intensity of pain is determined
by the amount of T-cell activity, with more activity
resulting in more pain. You can see how this works
by following the paths along which signals from the
S-fibers travel and noting that all of the synapses are
excitatory. Thus, signals from S-fibers always excite
T-cells, and therefore increase pain.
■ L-fibers. The large-diameter fibers (L-fibers) carry information
about nonpainful tactile stimulation. An
example of this type of stimulus would be signals sent
from rubbing the skin. Activity in the L-fibers can
send inhibition to the T-cells. This occurs because
signals that pass through SG (dashed line) activate
an inhibitory synapse. This closes the gate, which decreases
T-cell activity and decreases pain.
■ Central control. These fibers, which contain information
related to cognitive functions such as expectation,
attention, and distraction, carry signals down
from the cortex. As with the L-fibers, activity coming
down from the brain also closes the gate, decreases
T-cell activity, and decreases pain.
Since the introduction of the gate control model in
1965, researchers have determined that the neural circuits
that control pain are much more complex than what
was proposed in the original model (Perl & Kruger, 1996;
Sufka & Price, 2002). Nonetheless, the idea proposed by the
model—that the perception of pain is determined by a balance
between input from nociceptors in the skin and nonnociceptive
activity from the skin and the brain—stimulated
research that provided a great deal of additional evidence
for the idea that the perception of pain is influenced by more
than just stimulation of the skin (Fields & Basbaum, 1999;
Sufka & Price, 2002; Turk & Flor, 1999; Weissberg, 1999).
We will now consider some examples of how cognition can
influence the perception of pain.
You might also like
- Somatosensory PathwaysDocument36 pagesSomatosensory PathwaysAnonymous w3vYureNo ratings yet
- Spinal NotesDocument7 pagesSpinal NotesUsman Ali AkbarNo ratings yet
- Pain Pathways IN HUMANSDocument189 pagesPain Pathways IN HUMANSAshish VyasNo ratings yet
- Cap II - Curs 5 - 6 - Somatosensory System - 2014 - 2015 - PDFDocument137 pagesCap II - Curs 5 - 6 - Somatosensory System - 2014 - 2015 - PDFMaria MadalinaNo ratings yet
- KNTU NeuroMuscularSys 2018 MuscleSpindle SYariDocument15 pagesKNTU NeuroMuscularSys 2018 MuscleSpindle SYarisantiago mejia100% (1)
- Sensory Input and Responses (Part 2)Document7 pagesSensory Input and Responses (Part 2)Savannah Simone PetrachenkoNo ratings yet
- Anatomy Lecture # 14 (NS1)Document11 pagesAnatomy Lecture # 14 (NS1)Ali Farouk AhmedNo ratings yet
- Human Gait Analysed by An Artificial Neural Network ModelDocument4 pagesHuman Gait Analysed by An Artificial Neural Network Modelstroci4047No ratings yet
- Neuron Writing Ielts Task 1Document2 pagesNeuron Writing Ielts Task 1Ngân NgânNo ratings yet
- Transcutaneous Electrical Nerve StimulationDocument19 pagesTranscutaneous Electrical Nerve StimulationSaleha ZainNo ratings yet
- Sensory PathwaysDocument17 pagesSensory PathwaysNabilah Ani SofianNo ratings yet
- Exer 2 Nerve Conduction Final ShitDocument3 pagesExer 2 Nerve Conduction Final ShitJosephineCarandangNo ratings yet
- Neurofunctional Blueprints Transcripts For Tempe, January 2015 PDFDocument82 pagesNeurofunctional Blueprints Transcripts For Tempe, January 2015 PDFAnonymous oCjRxyBP100% (3)
- Gate ControlDocument2 pagesGate Controla3x5452No ratings yet
- Module 2 - PhysioDocument6 pagesModule 2 - PhysioVidisha KandwalNo ratings yet
- Theories of PainDocument13 pagesTheories of PainAnonymous fOcbAtNo ratings yet
- Structure and Function of NeuronsDocument6 pagesStructure and Function of NeuronsAlkykwyn LiteratoNo ratings yet
- BioDocument4 pagesBioHishanth SuthakaranNo ratings yet
- Nervous System Notes #2Document7 pagesNervous System Notes #2LeoNo ratings yet
- AP Psychology Module 9Document29 pagesAP Psychology Module 9Klein ZhangNo ratings yet
- MovementBrain and WorldDocument14 pagesMovementBrain and WorldCamila HenaoNo ratings yet
- MARCH 19, 2024 The Nervous SystemDocument49 pagesMARCH 19, 2024 The Nervous SystemAlmira ZuñigaNo ratings yet
- MARCH 19, 2024 The Nervous SystemDocument49 pagesMARCH 19, 2024 The Nervous SystemAlmira ZuñigaNo ratings yet
- 03 - The Third-Person View of The MindDocument22 pages03 - The Third-Person View of The MindLuis PuenteNo ratings yet
- Bioactivity study of modified curcumin loaded polymeric nanoparticlesFrom EverandBioactivity study of modified curcumin loaded polymeric nanoparticlesNo ratings yet
- Design Development and Analysis of a Nerve Conduction Study System An Auto Controlled Biofeedback ApproachFrom EverandDesign Development and Analysis of a Nerve Conduction Study System An Auto Controlled Biofeedback ApproachNo ratings yet
- New Microsoft Word DocumentDocument6 pagesNew Microsoft Word DocumentkptrrtNo ratings yet
- Electromyogram - Principles and ApplicationsDocument12 pagesElectromyogram - Principles and ApplicationsIoana PetreNo ratings yet
- Motor Control Theories and PhysiologyDocument7 pagesMotor Control Theories and PhysiologyAaminah NawazNo ratings yet
- NeuronsDocument10 pagesNeuronsEslam abokhalilNo ratings yet
- CH 3 (Biology and Behavior) NotesDocument12 pagesCH 3 (Biology and Behavior) NotesMelanieShiNo ratings yet
- Neuron SystemDocument17 pagesNeuron Systemdibya patraNo ratings yet
- KSG-Brain ActivityDocument10 pagesKSG-Brain ActivitySyahirah ZackNo ratings yet
- Nervous System Lab ReportDocument10 pagesNervous System Lab Reportapi-269985211No ratings yet
- Question 3 & 4Document15 pagesQuestion 3 & 4Arslan Muhammad EjazNo ratings yet
- Tactile PathwayDocument3 pagesTactile PathwayMae Angelie LabayaNo ratings yet
- The Somatosensory System I: Peripheral Mechanisms of Somatic SensationDocument25 pagesThe Somatosensory System I: Peripheral Mechanisms of Somatic SensationSbaa SyedaNo ratings yet
- Anat 6.5 Basal Ganglia - QuijanoDocument6 pagesAnat 6.5 Basal Ganglia - Quijanolovelots1234No ratings yet
- Pain Gate TheoryDocument1 pagePain Gate TheoryamyliaNo ratings yet
- THE Nervous System: Biology ProjectDocument8 pagesTHE Nervous System: Biology ProjectAravind JChandranNo ratings yet
- Neurology II 2.07 Pain Dr. Camara ChuaDocument24 pagesNeurology II 2.07 Pain Dr. Camara ChuaDanica Nicole Seco GabonNo ratings yet
- Skeletal Motor Control HierarchyDocument16 pagesSkeletal Motor Control HierarchyHapy ArdiaNo ratings yet
- Neuroanatomy 1Document65 pagesNeuroanatomy 1maNo ratings yet
- Pharm NotesDocument4 pagesPharm NotesGodfrey kauziNo ratings yet
- SSYA1013 Chapter 3 BiologicalDocument27 pagesSSYA1013 Chapter 3 BiologicalPing Ping TanNo ratings yet
- Nervous SystemDocument20 pagesNervous SystemIsheba Warren100% (3)
- Sept. 17/15 2. Refinement (Of Experiments To Reduce Suffering)Document5 pagesSept. 17/15 2. Refinement (Of Experiments To Reduce Suffering)amirNo ratings yet
- Autonomic Nervous System-NoteDocument13 pagesAutonomic Nervous System-NoteprosperosamegieNo ratings yet
- Gate Control Theory of PainDocument3 pagesGate Control Theory of Paincarmo-netoNo ratings yet
- Power Lab - Index CardDocument5 pagesPower Lab - Index CardKate Alyssa CatonNo ratings yet
- CNS NoteDocument203 pagesCNS NoteAhmad Khair Ahmad BazliNo ratings yet
- 7 Sensitivity and CoordinationDocument19 pages7 Sensitivity and CoordinationVarun GoseinNo ratings yet
- Telelettelelet ReviewerDocument4 pagesTelelettelelet ReviewerninaclaudiadelrosarioNo ratings yet
- Chapter 10Document8 pagesChapter 10Adam TNo ratings yet
- Psy 413 ProjectDocument38 pagesPsy 413 ProjectDumebi AneneNo ratings yet
- The Nervous SystemDocument71 pagesThe Nervous SystemDennis Limosnero MayorNo ratings yet
- NeuropsychologyDocument4 pagesNeuropsychologyBrahmi SathyNo ratings yet
- Sacral Nerve StimulationDocument5 pagesSacral Nerve StimulationAngel Mae Dela VegaNo ratings yet
- Smooth Muscle ExperimentDocument26 pagesSmooth Muscle ExperimentYanuar Dwi PutraNo ratings yet
- Neuro Anatomy, Motor NeuronDocument9 pagesNeuro Anatomy, Motor NeuronMichele Silva PiresNo ratings yet
- Adolescence and Adulthood Course OutlineDocument3 pagesAdolescence and Adulthood Course OutlineEnniah MlalaziNo ratings yet
- Child Development: Multiple ChoiceDocument14 pagesChild Development: Multiple ChoiceEnniah MlalaziNo ratings yet
- University of Zimbabwe Department of Psychology Cognition (PSY 216) Course Outline August 2019 Level 2:2 Second Semester 2019/2020 Academic YearDocument4 pagesUniversity of Zimbabwe Department of Psychology Cognition (PSY 216) Course Outline August 2019 Level 2:2 Second Semester 2019/2020 Academic YearEnniah MlalaziNo ratings yet
- University of Zimbabwe Department of Psychology Community Psychology (PSY 219) Course Outline First Semester 2018Document3 pagesUniversity of Zimbabwe Department of Psychology Community Psychology (PSY 219) Course Outline First Semester 2018Enniah MlalaziNo ratings yet
- Assignment MuchDocument5 pagesAssignment MuchEnniah MlalaziNo ratings yet
- Qualitative Research Methods Course Outline 2018Document3 pagesQualitative Research Methods Course Outline 2018Enniah MlalaziNo ratings yet
- What Are The Physiological Processes Involved in Recognizing Objects by TouchDocument2 pagesWhat Are The Physiological Processes Involved in Recognizing Objects by TouchEnniah MlalaziNo ratings yet
- University of Zimbabwe: Question: As A Zimbabwean Psychology Student Discuss HowDocument6 pagesUniversity of Zimbabwe: Question: As A Zimbabwean Psychology Student Discuss HowEnniah MlalaziNo ratings yet
- University of Zimbabwe: Question: As A Zimbabwean Psychology Student Discuss HowDocument6 pagesUniversity of Zimbabwe: Question: As A Zimbabwean Psychology Student Discuss HowEnniah MlalaziNo ratings yet
- Similarities and DifferencesDocument7 pagesSimilarities and DifferencesEnniah MlalaziNo ratings yet
- Chapter Two Exploring The LiteratureDocument31 pagesChapter Two Exploring The LiteratureEnniah MlalaziNo ratings yet
- AssignmentDocument1 pageAssignmentEnniah MlalaziNo ratings yet
- University of Zimbabwe: Learning Principles (Psy 213)Document5 pagesUniversity of Zimbabwe: Learning Principles (Psy 213)Enniah MlalaziNo ratings yet
- Institute of Metallurgy and Materials Engineering Faculty of Chemical and Materials Engineering University of The Punjab LahoreDocument10 pagesInstitute of Metallurgy and Materials Engineering Faculty of Chemical and Materials Engineering University of The Punjab LahoreMUmairQrNo ratings yet
- School: Grade Level: Teacher: Section Teaching Dates and Time: QuarterDocument3 pagesSchool: Grade Level: Teacher: Section Teaching Dates and Time: QuarterZeny Aquino DomingoNo ratings yet
- Aabroo Recyclable Waste Program Survey: 1. Demographic InformationDocument2 pagesAabroo Recyclable Waste Program Survey: 1. Demographic InformationIqra KhaliqNo ratings yet
- Audio Spot LightingDocument20 pagesAudio Spot LightingLavanya Vaishnavi D.A.No ratings yet
- TSBDocument3 pagesTSBnoe dela vegaNo ratings yet
- Responsible Living: Mantri DevelopersDocument15 pagesResponsible Living: Mantri Developersnadaf8No ratings yet
- Quiz Business MathDocument5 pagesQuiz Business MathMA. JEMARIS SOLISNo ratings yet
- Course For Loco Inspector Initial (Diesel)Document239 pagesCourse For Loco Inspector Initial (Diesel)Hanuma Reddy93% (14)
- Cigarettes and AlcoholDocument1 pageCigarettes and AlcoholHye Jin KimNo ratings yet
- Solid State Controller of Drives - ExperimentDocument37 pagesSolid State Controller of Drives - ExperimentRakesh Singh LodhiNo ratings yet
- The Six Types of Simple MachinesDocument4 pagesThe Six Types of Simple MachinesmarroNo ratings yet
- Tyre Industry in IndiaDocument14 pagesTyre Industry in IndiaH Janardan PrabhuNo ratings yet
- Maharashtra Brochure (2023)Document4 pagesMaharashtra Brochure (2023)assmexellenceNo ratings yet
- Vanilla Farming: The Way Forward: July 2019Document6 pagesVanilla Farming: The Way Forward: July 2019mituNo ratings yet
- Oplan Tumbler ProposalDocument3 pagesOplan Tumbler ProposalKay Tuppil ManzanillaNo ratings yet
- BÀI TẬP TA 9 THEO CHUYÊN ĐỀ NGỮ PHÁPDocument213 pagesBÀI TẬP TA 9 THEO CHUYÊN ĐỀ NGỮ PHÁPhoangmaiNo ratings yet
- Planetary Characteristics: © Sarajit Poddar, SJC AsiaDocument11 pagesPlanetary Characteristics: © Sarajit Poddar, SJC AsiaVaraha Mihira100% (11)
- SCM 025-108 M2Document8 pagesSCM 025-108 M2kazdanoNo ratings yet
- AP Human Geography Review Unit 2Document18 pagesAP Human Geography Review Unit 2BaselOsman50% (2)
- Site AnalysisDocument20 pagesSite AnalysisCarlo RosaioNo ratings yet
- Cooking Off The Clock by Elizabeth Falkner - Recipes and ExcerptDocument16 pagesCooking Off The Clock by Elizabeth Falkner - Recipes and ExcerptThe Recipe ClubNo ratings yet
- PT3 Guided WritingDocument8 pagesPT3 Guided WritingDayang Haffizah Abg AbdillahNo ratings yet
- 520l0553 PDFDocument52 pages520l0553 PDFVasil TsvetanovNo ratings yet
- Atlantis Is Discovered - Atlantis in Middle Danubian Depression (Pannonian Basin) A New Location of Atlantis IslandDocument31 pagesAtlantis Is Discovered - Atlantis in Middle Danubian Depression (Pannonian Basin) A New Location of Atlantis Islandpaulct2004721260% (10)
- ME 175A Midterm SolutionsDocument4 pagesME 175A Midterm SolutionsDavid ChonNo ratings yet
- Appetizer Summative TestDocument36 pagesAppetizer Summative TestArgelynPadolinaPedernalNo ratings yet
- Stratigraphy MCQs With AnswerDocument10 pagesStratigraphy MCQs With Answerkumar Harsh67% (3)
- Manual TR420 enDocument38 pagesManual TR420 enMari Sherlin Salisi-ChuaNo ratings yet
- Research Report 2011 Asses Water ChambalDocument41 pagesResearch Report 2011 Asses Water Chambalsamyak sambhareNo ratings yet
- UNDP NP Dhangadhi SWM TOR FinalDocument4 pagesUNDP NP Dhangadhi SWM TOR FinalNirmal K.c.No ratings yet