Professional Documents
Culture Documents
Practice Problems On Net Ionic Equations
Uploaded by
Maddison LilyOriginal Title
Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
Practice Problems On Net Ionic Equations
Uploaded by
Maddison LilyCopyright:
Available Formats
PRACTICE PROBLEMS ON NET IONIC EQUATIONS
Show the complete ionic and net ionic forms of the following equations. If all species are spectator ions, please
indicate that no reaction takes place. Note: you need to make sure the original equation is balanced before
proceeding! A set of solubility rules are given at the end of this document.
1. AgNO3(aq) + KCl(aq) AgCl(s) + KNO3(aq)
2. Mg(NO3)2(aq) + Na2CO3(aq) MgCO3(s) + NaNO3(aq)
3. strontium bromide(aq) + potassium sulfate(aq) strontium sulfate(s) + potassium bromide(aq)
4. manganese(II)chloride(aq) + ammonium carbonate(aq) manganese(II)carbonate(s) + ammonium chloride(aq)
5. chromium(III)nitrate(aq) + iron(II)sulfate(aq) chromium(III)sulfate(aq) + iron(II)nitrate(aq)
Please complete the following reactions, and show the complete ionic and net ionic forms of the equation:
6. K3PO4(aq) + Al(NO3)3(aq)
7. BeI2(aq) + Cu2SO4(aq)
8. Ni(NO3)3(aq) + KBr(aq)
9. cobalt(III)bromide + potassium sulfide
10. barium nitrate + ammonium phosphate
11. calcium hydroxide + iron(III)chloride
12. rubidium fluoride + copper(II)sulfate
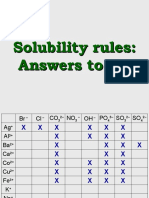
Solubility Rules
1. All salts of Group IA, and ammonium are soluble.
2. All salts of nitrates, chlorates and acetates are soluble.
3. All salts of halides are soluble except those of silver(I), copper(I), lead(II), and mercury(I).
4. All salts of sulfate are soluble except for barium sulfate, lead(II) sulfate, and strontium sulfate.
5. All salts of carbonate, phosphate and sulfite are insoluble, except for those of group IA and ammonium.
6. All oxides and hydroxides are insoluble except for those of group IA, calcium, strontium and barium.
7. All salts of sulfides and insoluble except for those of Group IA and IIA elements and of ammonium.
Author: Georgia Perimeter College page 1 of 3
PRACTICE PROBLEMS ON NET IONIC EQUATIONS
**NOTES: "Total ionic equation" means "complete ionic equation." Also, you don't have to write slashes through the spectator ions as shown below.
Answer Key to Practice Problems on Net Ionic Equations:
1. Molecular: AgNO3 (aq) + KCl (aq) AgCl (s) + KNO3 (aq)
Total Ionic: Ag+ (aq) + NO3¯ (aq) + K+ (aq) + Cl¯ (aq) AgCl (s) + K+ (aq) + NO3¯ (aq)
Net Ionic: Ag+ (aq) + Cl¯ (aq) AgCl (s)
2. Molecular: Mg(NO3)2 (aq) + Na2CO3 (aq) MgCO3 (s) + 2 NaNO3 (aq)
Total Ionic: Mg2+ (aq) + 2 NO3¯ (aq) + 2 Na+ (aq) + CO32- (aq) MgCO3 (s) + 2 Na+ (aq) + 2 NO3¯ (aq)
Net Ionic: Mg2+ (aq) + CO32- (aq) MgCO3 (s)
3. Molecular: SrBr2 (aq) + K2SO4 (aq) SrSO4 (s) + 2 KBr (aq)
Total Ionic: Sr2+ (aq) + 2 Br¯ (aq) + 2 K+ (aq) + SO42- (aq) SrSO4 (s) + 2 K+ (aq) + 2 Br¯ (aq)
Net Ionic: Sr2+ (aq) + SO42- (aq) SrSO4 (s)
4. Molecular: MnCl2 (aq) + (NH4)2CO3 (aq) MnCO3 (s) + 2 NH4Cl (aq)
Total Ionic: Mn2+ (aq) + 2 Cl¯ (aq) + 2 NH4+ (aq) + CO32- (aq) MnCO3 (s) + 2 NH4+ (aq) + 2 Cl¯ (aq)
Net Ionic: Mn2+ (aq) + CO32- (aq) MnCO3 (s)
5. Molecular: 2 Cr(NO3)3 (aq) + 3 FeSO4 (aq) 3 Fe(NO3)2 (aq) + Cr2(SO4)3 (aq)
Total Ionic: 2 Cr3+ (aq) + 6 NO3¯ (aq) + 3 Fe2+ (aq) + 3 SO42- (aq) 2 Cr3+ (aq) + 6 NO3¯ (aq) + 3 Fe2+ (aq) +
3 SO42- (aq)
Net Ionic: No Reaction
6. Molecular: K3PO4 (aq) + Al(NO3)3 (aq) AlPO4 (s) + 3 KNO3 (aq)
Total Ionic: 3 K+ (aq) + PO43- (aq) + Al3+ (aq) + 3 NO3¯ (aq) AlPO4 (s) + 3 K+ (aq) + 3 NO3¯ (aq)
Net Ionic: Al3+ (aq) + PO43- (aq) AlPO4 (s)
7. Molecular: BeI2 (aq) + Cu2SO4 (aq) BeSO4 (aq) + 2 CuI (s)
Total Ionic: Be2+ (aq) + 2 I¯ (aq) 2 Cu+ (aq) + SO42- (aq) Be2+ (aq) + SO42- (aq) + 2 CuI (s)
Net Ionic: 2 Cu2+ (aq) + 2 I¯ (aq) 2 CuI (s)
8. Molecular: Ni(NO3)3 (aq) + 3 KBr (aq) NiBr3 (aq) + 3 KNO3 (aq)
Total Ionic: Ni3+ (aq) + 3 NO3¯ (aq) + 3 K+ (aq) + 3 Br¯ (aq) Ni3+ (aq) + 3 NO3¯ (aq) + 3 K+ (aq) + 3 Br¯ (aq)
Author: Georgia Perimeter College page 2 of 3
PRACTICE PROBLEMS ON NET IONIC EQUATIONS
Net Ionic: No Reaction
9. Molecular: 2 CoBr3 (aq) + 3 K2S(aq) Co2S3 (s) + 6 KBr (aq)
Total Ionic: 2 Co3+ (aq) + 6 Br¯ (aq) + 6 K+ (aq) + 3 S2- (aq) Co2S3 (s) + 6 K+ (aq) + 6 Br¯ (aq)
Net Ionic: 2 Co3+ (aq) + 3 S2- (aq) Co2S3 (s)
10. Molecular: 3 Ba(NO3)2 (aq) + 2 (NH4)3PO4 (aq) Ba3(PO4)2 (s) + 6 NH4NO3 (aq)
Total Ionic: 3 Ba2+ (aq) + 6 NO3¯ (aq) + 6 NH4+ (aq) + PO43- (aq) Ba3(PO4)2 (s) + 6 NH4+ (aq) + 6 NO3¯ (aq)
Net Ionic: 3 Ba2+(aq) + 2 PO43- (aq) Ba3(PO4)2 (s)
11. Molecular: 3 Ca(OH)2 (aq) + 2 FeCl3 (aq) 3 CaCl2 (aq) + 2 Fe(OH)3 (s)
Total Ionic: 3 Ca2+ (aq) + 6 OH¯ (aq) + 2 Fe3+ (aq) + 6 Cl¯ (aq) 3 Ca2+ (aq) + 6 Cl¯ (aq) + 2 Fe(OH)3 (s)
Net Ionic: 2 Fe3+ (aq) + 6 OH¯ (aq) 2 Fe(OH)3 (s) simplifies to: Fe3+ (aq) + 3 OH¯ (aq) Fe(OH)3 (s)
12. Molecular: 2 RbF (aq) + CuSO4 (aq) Rb2SO4 (aq) + CuF2 (aq)
Total Ionic: 2 Rb+(aq) + 2 F¯ (aq) + Cu2+ (aq) + SO42- (aq) 2 Rb+ (aq) + SO42- (aq) + Cu2+ (aq) + 2 F¯ (aq)
Net Ionic: No Reaction
Author: Georgia Perimeter College page 3 of 3
You might also like
- Scholten Jan Homoeopathy and The ElementsDocument98 pagesScholten Jan Homoeopathy and The ElementsKarthik100% (4)
- 6 Precipitation ReactionsDocument2 pages6 Precipitation ReactionsJacob DaughertyNo ratings yet
- EXPERIMENT 5 - Double Displacement Reactions: Report FormDocument6 pagesEXPERIMENT 5 - Double Displacement Reactions: Report FormNguyễn Hoàng ĐăngNo ratings yet
- Lecture 3 Chemical ReactionsDocument63 pagesLecture 3 Chemical ReactionsHiep NguyenNo ratings yet
- Nelson Grade 11 Chemistry ReviewDocument10 pagesNelson Grade 11 Chemistry Reviewexhalait67% (3)
- Solution Manual For Introduction To Programming Using Python 1st Edition Schneider 0134058224 9780134058221Document7 pagesSolution Manual For Introduction To Programming Using Python 1st Edition Schneider 0134058224 9780134058221jennifersmithdesgyzqjni100% (17)
- Net Ionic Equations-ProblemsDocument3 pagesNet Ionic Equations-ProblemsChikuta ShingaliliNo ratings yet
- Net Ionic EquationDocument3 pagesNet Ionic Equationsara bdeirNo ratings yet
- Practice Problems On Net Ionic EquationsDocument3 pagesPractice Problems On Net Ionic EquationsZainabNo ratings yet
- Answer Key To Practice Problems On Net Ionic EquationsDocument4 pagesAnswer Key To Practice Problems On Net Ionic EquationsmerlindikaNo ratings yet
- Net Ionic EquationsDocument7 pagesNet Ionic EquationscelineNo ratings yet
- NIE - and - Particulate - Drawings - Worksheet - Answers at End - 2017-1Document8 pagesNIE - and - Particulate - Drawings - Worksheet - Answers at End - 2017-1Jane Ivanova100% (1)
- Net Ionic EditedDocument8 pagesNet Ionic EditedMuhammad AbdullahNo ratings yet
- WritingNetIonicEquations 2Document8 pagesWritingNetIonicEquations 2Nico Theodorus SimamoraNo ratings yet
- Francisco Vigil - CHDocument5 pagesFrancisco Vigil - CHapi-528208996No ratings yet
- 110 WS Net Ionic Eqns KeyDocument3 pages110 WS Net Ionic Eqns KeyZaara RyeenNo ratings yet
- 1ionic Reactions2Document2 pages1ionic Reactions2Michael MitchellNo ratings yet
- Writing Ionic Equations MSDocument3 pagesWriting Ionic Equations MSSmahs ZabirNo ratings yet
- Chapter 4Document23 pagesChapter 4V KumarNo ratings yet
- Precipitation Reactions NotesDocument8 pagesPrecipitation Reactions NotessprijayaNo ratings yet
- Ionic Equations wksht2 PDFDocument2 pagesIonic Equations wksht2 PDFBrandeice Barrett0% (1)
- HTTPWWW - occc.edukmbaileyChem1115TutorialsNet Ionic Eqns Answers - HTMDocument1 pageHTTPWWW - occc.edukmbaileyChem1115TutorialsNet Ionic Eqns Answers - HTMVishnu SharmaNo ratings yet
- Soluble Insoluble 6. Ca (NO 3. K Soluble Soluble: Follow This Format For Question B, C and DDocument4 pagesSoluble Insoluble 6. Ca (NO 3. K Soluble Soluble: Follow This Format For Question B, C and DBEST OF ONE PIECENo ratings yet
- Chem 125 07.10.2019Document9 pagesChem 125 07.10.2019nilofar jawadiNo ratings yet
- Redoxanswers PDFDocument2 pagesRedoxanswers PDFAlexander Salado IbrahimNo ratings yet
- Redoxanswers PDFDocument2 pagesRedoxanswers PDFalbi veshiNo ratings yet
- Chapter 5 Answers Practice Examples: ReductionDocument7 pagesChapter 5 Answers Practice Examples: ReductionEmre Enes EdizNo ratings yet
- Edexecel IAL Lesson 1Document20 pagesEdexecel IAL Lesson 1Pevin De silvaNo ratings yet
- Chemical Equations: Preparation For College Chemistry Columbia University Department of ChemistryDocument31 pagesChemical Equations: Preparation For College Chemistry Columbia University Department of ChemistryLakshmi SinghNo ratings yet
- 11.3 Powerpoint - Net Ionic Equations CE 2Document25 pages11.3 Powerpoint - Net Ionic Equations CE 2Lauren BoersmaNo ratings yet
- 05 Petrucci10e CSMDocument45 pages05 Petrucci10e CSMAlexNo ratings yet
- Writing Chemical Equations: Test Yourself 8.1 (Page 130)Document1 pageWriting Chemical Equations: Test Yourself 8.1 (Page 130)Zeeshan MunirNo ratings yet
- 20-21 Net Ionic Eqns HW-1Document1 page20-21 Net Ionic Eqns HW-1Anthony WangNo ratings yet
- Chapter 4Document28 pagesChapter 4Andrea PerezNo ratings yet
- Soal ElectrochemistryDocument3 pagesSoal ElectrochemistryHerlinda OktaNo ratings yet
- Chemistry 30AP Electrochemistry Workbook: Net Ionic EquationsDocument27 pagesChemistry 30AP Electrochemistry Workbook: Net Ionic EquationsDayanul AlamNo ratings yet
- HW 4Document3 pagesHW 4api-368121935No ratings yet
- Cover Page Chemistry 2 AdobeDocument7 pagesCover Page Chemistry 2 AdobeJWAN RA YA3QOBNo ratings yet
- Key3 PDFDocument5 pagesKey3 PDFJC JimenezNo ratings yet
- Chemical Reaction NotesDocument30 pagesChemical Reaction NotesTS WongNo ratings yet
- Introductory Chemistry 8Th Edition Zumdahl Solutions Manual Full Chapter PDFDocument40 pagesIntroductory Chemistry 8Th Edition Zumdahl Solutions Manual Full Chapter PDFmasonfisheribasedgcyx100% (10)
- Electrochemistry: Chemistry 30 WorksheetsDocument49 pagesElectrochemistry: Chemistry 30 Worksheetsdan anna stylesNo ratings yet
- Precipitates and Solubility Rules Pre-Lab Discussion:: Solution Set A Solution Set BDocument7 pagesPrecipitates and Solubility Rules Pre-Lab Discussion:: Solution Set A Solution Set Bapi-296518880No ratings yet
- Solubility Rules: Answers To LabDocument7 pagesSolubility Rules: Answers To LabDeba Jyoti NeogNo ratings yet
- Pitogo, Chanie Experiment 2Document7 pagesPitogo, Chanie Experiment 2Chanie Baguio PitogoNo ratings yet
- Chapter 4 Reactions in Aqueous SolutionDocument35 pagesChapter 4 Reactions in Aqueous Solutionnicole.lippolisNo ratings yet
- Homework 7 KeyDocument4 pagesHomework 7 KeyTinh AppleNo ratings yet
- Post-Lab 4 Qualitative Analysis-SolutionsDocument7 pagesPost-Lab 4 Qualitative Analysis-SolutionsUzo Paul NwabuisiNo ratings yet
- Introduction To Reactions in Aqueous Solutions Practice ExamplesDocument25 pagesIntroduction To Reactions in Aqueous Solutions Practice Exampleskennethleo69No ratings yet
- BATTERYDocument19 pagesBATTERYRaja PRNo ratings yet
- Reacciones Redox: Trabajo Practico N°Document5 pagesReacciones Redox: Trabajo Practico N°GuadahNo ratings yet
- Electrochemistry NotesDocument98 pagesElectrochemistry NotesTaufik Sandi100% (2)
- AP Electro Chem 1515 Ex AnsDocument40 pagesAP Electro Chem 1515 Ex AnsYj TehNo ratings yet
- Reaction in Aqueous SolutionDocument24 pagesReaction in Aqueous SolutionJa FuentesNo ratings yet
- Half and Ionic Equations (GCSE)Document31 pagesHalf and Ionic Equations (GCSE)william.ongeri.tutoringNo ratings yet
- Ions in SolutionDocument8 pagesIons in SolutionNaufal TsabitadzNo ratings yet
- Chemistry 1A Fall 2010 Exam 2 Key Chapters 4 (Part), 5, 6, and 7 (Part)Document7 pagesChemistry 1A Fall 2010 Exam 2 Key Chapters 4 (Part), 5, 6, and 7 (Part)jasminp8No ratings yet
- CBSE Class 10 Chemistry Worksheet - Chemical Reactions and Equations PDFDocument4 pagesCBSE Class 10 Chemistry Worksheet - Chemical Reactions and Equations PDFMalancha high school HS100% (1)
- CBSE Class 10 Chemistry Worksheet - Chemical Reactions and Equations PDFDocument4 pagesCBSE Class 10 Chemistry Worksheet - Chemical Reactions and Equations PDFMalancha high school HS0% (1)
- CBSE Class 10 Chemistry Worksheet - Chemical Reactions and EquationsDocument4 pagesCBSE Class 10 Chemistry Worksheet - Chemical Reactions and EquationsMalancha high school HS100% (1)
- Practice Makes Perfect in Chemistry: Oxidation-ReductionFrom EverandPractice Makes Perfect in Chemistry: Oxidation-ReductionRating: 5 out of 5 stars5/5 (1)
- Practice Makes Perfect in Chemistry: Oxidation-Reduction with AnswersFrom EverandPractice Makes Perfect in Chemistry: Oxidation-Reduction with AnswersNo ratings yet
- Balancing Chemical EquationsDocument4 pagesBalancing Chemical EquationsAnonymous AMmNGgPNo ratings yet
- Chemsheets GCSE 1123 Metal Extraction 1Document2 pagesChemsheets GCSE 1123 Metal Extraction 1Vinay SINGHNo ratings yet
- Recent Developments in The Chloride Processing of Nickel Laterites PDFDocument14 pagesRecent Developments in The Chloride Processing of Nickel Laterites PDFmtanaydinNo ratings yet
- Qualitatile Inorganic AnalysisDocument9 pagesQualitatile Inorganic AnalysisRamanNo ratings yet
- Pre-Prelims Revision PaperDocument6 pagesPre-Prelims Revision PaperaaaaNo ratings yet
- Zirconium 702C and Zirconium 705C PDFDocument2 pagesZirconium 702C and Zirconium 705C PDFgullenariNo ratings yet
- Mechnical Property For Q235B Carbon SteelDocument3 pagesMechnical Property For Q235B Carbon SteelPritom Ahmed100% (1)
- Electron Configuration of All ElementsDocument5 pagesElectron Configuration of All ElementsAnne VillarealNo ratings yet
- Soalan Test 3 Semester 2 Sesi 2022 - 2023Document8 pagesSoalan Test 3 Semester 2 Sesi 2022 - 2023y15zrcdu1823No ratings yet
- Materiales API - Annex G and H From API 610 11th ISO13709Document5 pagesMateriales API - Annex G and H From API 610 11th ISO13709raldelgadomaNo ratings yet
- 1st Periodic Test - Science 7Document4 pages1st Periodic Test - Science 7Divina bentayaoNo ratings yet
- Quarter 1 - Module 3 Writing and Naming Chemical Compounds: Yolanda A. PeñalosaDocument14 pagesQuarter 1 - Module 3 Writing and Naming Chemical Compounds: Yolanda A. PeñalosaShane Tabalba100% (4)
- PijDocument149 pagesPijAduchelab AdamsonuniversityNo ratings yet
- EIGA (2008) - Comparison of EP, USP & JP For Medicinal GasDocument21 pagesEIGA (2008) - Comparison of EP, USP & JP For Medicinal GashuynhhaichauchauNo ratings yet
- November 2022 (9701 - 13) QPDocument16 pagesNovember 2022 (9701 - 13) QPHung Mang ThiNo ratings yet
- Is - 412Document12 pagesIs - 412raje_58No ratings yet
- 001 Shiv Puran NepaliDocument3 pages001 Shiv Puran Nepalisupendra phuyalNo ratings yet
- Astm 1394-2009Document4 pagesAstm 1394-2009Srinivasan KrishnamoorthyNo ratings yet
- H2 Chem Summary of Transition ElementDocument7 pagesH2 Chem Summary of Transition Elementonnoez100% (2)
- Hydrogen Peroxide: Concentration Determination 0.1-5%Document3 pagesHydrogen Peroxide: Concentration Determination 0.1-5%Muzaffar BhatNo ratings yet
- General Chemistry 1: Quarter 3 - WEEK 3-4Document19 pagesGeneral Chemistry 1: Quarter 3 - WEEK 3-4RODEL AZARES100% (1)
- Piping Material Selection GuideDocument6 pagesPiping Material Selection Guideqaisarabbas9002100% (3)
- Key Concept Chapter 10 ElectrolysisDocument20 pagesKey Concept Chapter 10 ElectrolysisKim KatNo ratings yet
- Chemistry Practical File (WSD E-Book)Document36 pagesChemistry Practical File (WSD E-Book)dhruvsinghal679% (14)
- Chemistry NYA Notes and Exercises Part 1Document71 pagesChemistry NYA Notes and Exercises Part 1qwerty514No ratings yet
- Lithium and Lithium CompoundsDocument38 pagesLithium and Lithium CompoundsNancy ParvinaNo ratings yet