Professional Documents
Culture Documents
TISSUE REPAIR Handout
Uploaded by
Geoffrey0 ratings0% found this document useful (0 votes)
13 views3 pagesTissue repair and healing involves two processes - regeneration, where new cells replace damaged tissue, and repair through connective tissue replacement. Healing begins during the inflammatory phase as new blood vessels form (angiogenesis) and granulation tissue develops from endothelial cell migration, proliferation and recruitment of other cells. Fibroblasts are triggered by growth factors to migrate to the injury site and deposit extracellular matrix including collagen to strengthen the wound as it remodels over time. Successful healing depends on factors like nutrition, infection control and wound characteristics.
Original Description:
Original Title
TISSUE REPAIR handout
Copyright
© © All Rights Reserved
Available Formats
DOC, PDF, TXT or read online from Scribd
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentTissue repair and healing involves two processes - regeneration, where new cells replace damaged tissue, and repair through connective tissue replacement. Healing begins during the inflammatory phase as new blood vessels form (angiogenesis) and granulation tissue develops from endothelial cell migration, proliferation and recruitment of other cells. Fibroblasts are triggered by growth factors to migrate to the injury site and deposit extracellular matrix including collagen to strengthen the wound as it remodels over time. Successful healing depends on factors like nutrition, infection control and wound characteristics.
Copyright:
© All Rights Reserved
Available Formats
Download as DOC, PDF, TXT or read online from Scribd
0 ratings0% found this document useful (0 votes)
13 views3 pagesTISSUE REPAIR Handout
Uploaded by
GeoffreyTissue repair and healing involves two processes - regeneration, where new cells replace damaged tissue, and repair through connective tissue replacement. Healing begins during the inflammatory phase as new blood vessels form (angiogenesis) and granulation tissue develops from endothelial cell migration, proliferation and recruitment of other cells. Fibroblasts are triggered by growth factors to migrate to the injury site and deposit extracellular matrix including collagen to strengthen the wound as it remodels over time. Successful healing depends on factors like nutrition, infection control and wound characteristics.
Copyright:
© All Rights Reserved
Available Formats
Download as DOC, PDF, TXT or read online from Scribd
You are on page 1of 3
TISSUE REPAIR & HEALING
• REGENERATION (REPLACEMENT BY N-CELLS)
• REPAIR (REPLACEMENT BY CONNECTIVE TISSUE)
REPAIR BEGINS DURING INFLAMMATION
NEW VESSEL FORMATION (ANGIOGENESIS)
• PROTEOLYTIC ENZYME DIGESTS BASEMENT MEMBRANE
• MIGRATION OF ENDOTHELIAL CELLS
• PROLIFERATION OF ENDOTHELIAL CELLS
• MATURATION OF ENDOTHELIAL CELLS
• RECRUITMENT OF PERIENDOTHELIAL CELLS (PERICYTES & SMOOTH
MUSCLE CELLS)
CONTROLLED BY:
• PROMOTERS (GROWTH FACTORS)
• VASCULAR CELLS & EXTRACELLULAR MATRIX
• CYCLINS
TYPES OF CELLS vis-à-vis THE CELL CYCLE
1. LABILE CELLS: CONTINUOUSLY IN THE CYCLE eg EPITHELIAL CELLS,
SKIN, GIT, CERVIX
2. STABLE CELLS: IN Go PHASE, BUT CAN ENTER CYCLE AS REQUIRED eg.
PARENCHYMAL CELLS, HEPATOCYTES, ENDOTHELIUM
3. PERMANENT CELLS: CELLS THAT HAVE LEFT THE CYCLE eg NEURONS,
CARDIAC MUSCLE
MOLECULAR EVENTS IN CELL GROWTH
AUTOCRINE: SIGNAL FROM SAME CELL
PARACRINE: SIGNAL FROM CELL NEARBY
ENDOCRINE: SIGNAL FROM BLOOD STREAM
CELL SURFACE RECEPTORS
- FIBROBLAST GROWTH FACTOR (FGF)
- EPIDERMAL GROWTH FACTOR (EGF)
- PLATELET DERIVED GROWTH FACTOR (PDGF)
- VASCULAR ENDITHELIAL GROWTH FACTOR (VEGF)
RECEPTORS WITHOUT INTRINSIC CATALYTIC ACTIVITY
- CYTOKINES
G PROTEIN-LINKED RECEPTORS
CHEMOKINES
HEALING
FOLLOWS INFLAMMATION
ACUTE INFLAMMATION CHRONIC INFLAMMATION
(DESTRUCTION OF PARENCHYMAL CELLS AND STROMAL FRAMEWORK)
HEALING BY FIRST OR SECOND INTENTION
1. REGENERATION OF PARENCHYMAL CELLS
2. MIGRATION & PROLIFERATION OF PARENCHYMAL & CONNECTIVE
TISSUE CELLS
3. SYNTHESIS OF ECM
4. REMODELLING OF CONNECTIVE TISSUE & PARENCHYMAL COMPONENTS
5. COLLAGENISATION & ACQUISITION OF WOUND STRENGTH (FIBROSIS /
SCARRING)
MIGRATION OF FIBROBLASTS TO THE SITE OF INJURY AND THEN
PROLIFERATION AT THAT SITE.
TRIGGERED BY PDGF, EGF, FGF,
TGFB, CYTOKINES
DEPOSITION OF EXTRACELULAR MATRIX
FIBROBLASTS LAY DOWN COLLAGEN
TYPES I, II, III = FIBRILLAR COLLAGEN
TYPES IV, V, VI + AMORPHOUS (IV + BASEMENT MEMBRANE)
COLLAGEN TENSILE STRENGTH TYPES I IV
FIBRILS PLEATED & CROSS LINKED TO INCREASE TENSILE STRENGTH
PROLINE HYDROXYPROLINE CROSS LINKAGES (PRO-COLLAGEN -
COLLAGEN)
STIMULATED BY PDGF, FGF, TGF-B AND CYTOKINES SECRETED BY WBC
AND FIBROBLASTS
MATURATION & ORGANISATION (REMODELLING)
USE THE BM AS A “TEMPLATE”, NEED TO BREAK DOWN ECM IN ORDER TO
DEBRIDE WOUND
CONNECTIVE TISSUE REMODELLING
METALLOPROTEINASES:
GELATINASES (TYPE IV)
STROMELYSINS
COLLAGENASES (TYPES I, II & III)
ECM DEGRADATION INHIBITED BY TISSUE INHIBITORS OF
METALLOPROTEINASES
SECRETION OF ABOVE ENZYMES INHIBITED BY TGFβ & STEROIDS
FIBROBLASTS SYNTHESISE ELASTIC FIBRES AND SECRETE ECM
ECM COMPRISES
• COLLAGENS & ELASTINS
• ADHESIVE GLYCOPROTEINS (LAMININ) & INTEGRINS
• PROTEOGLYCANS & HYALURONAN
FACTORS INVOLVED
INFLAMMATORY RESPONSE
GRANULATION TISSUES
PROTEIN
VITAMIN C
ZINC
GROWTH FACTORS
FACTORS AFFECTING WOUND HEALING
SYSTEMIC FACTORS:
• NUTRITION- ZN, PROTEIN & VIT C DEFICIENCIES
• METABOLIC STATUS – DIABETES MELLITUS
• CIRCULATORY STATUS – ARTERIOSCLEROSIS
• HORMONES - GLUCOCORTICOIDS
LOCAL FACTORS
• INFECTION
• MECHANICAL FACTORS – MOBILITY
• FOREIGN BODIES – UNNECESSARY SUTURES, FRAGMENTS OF GLASS,
STELL, SHRAPNEL OR BONE
• SIZE, LOCATION & TYPE OF WOUND
PATHOLOGICAL CONDITIONS
• KELOID
- EXCESS COLLAGEN
- GENETIC
- REPEATED TRAUMA
• PYOGENIC GRANULOMA “PROUD FLESH”
SUMMARY
- EXCESS GRANULATION TISSUE
- FEMALE
- SITES
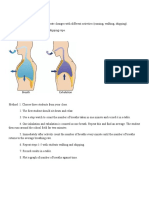
REPAIR BEGINS EARLY ON IN INFLAMMATION (24 HRS).
BEGINS WITH THE FORMATION OF GRANULATION TISSUE (NEW
VESSELS AND FIBROBLASTS)
*GRANULATION TISSUE IS THE HALLMARK OF HEALING*
You might also like
- Wound Healing: Asst. Professor V.D.C. Sravan KumarDocument30 pagesWound Healing: Asst. Professor V.D.C. Sravan KumarKeerthi NeeluforNo ratings yet
- Inflammation and Repair 26-08-22Document86 pagesInflammation and Repair 26-08-22Crystal GamingNo ratings yet
- Pert 43 - Aging in ECM - Dr. Tan Fei Fan, M.Biomed (Dept. Histologi)Document25 pagesPert 43 - Aging in ECM - Dr. Tan Fei Fan, M.Biomed (Dept. Histologi)Juan FeivelNo ratings yet
- Cell Injury, Adaptation and Death: Dr. Syahruddin Saragih, SP - PA Departemen of Pathology UisuDocument27 pagesCell Injury, Adaptation and Death: Dr. Syahruddin Saragih, SP - PA Departemen of Pathology Uisunovriadi suhendraNo ratings yet
- Wound Healing: Mechanism & PhysiologyDocument31 pagesWound Healing: Mechanism & PhysiologyManoj Kumar PasunooriNo ratings yet
- Bio Mod 4 PPT 1Document42 pagesBio Mod 4 PPT 1Yvone PogoyNo ratings yet
- Cellular Basis of DiseaseDocument72 pagesCellular Basis of DiseaseNikkole PhalulaNo ratings yet
- Introduction To Pathology Cmu 605 (Pathology Cmu 605) 2021 Enugu (Autosaved)Document355 pagesIntroduction To Pathology Cmu 605 (Pathology Cmu 605) 2021 Enugu (Autosaved)Ogbuefi PascalNo ratings yet
- Tissue RepairDocument56 pagesTissue RepairZainul Iman Abdul MalekNo ratings yet
- Gaucherdisease 170318151748Document45 pagesGaucherdisease 170318151748shruti kumarNo ratings yet
- Pathology of Urinary SystemDocument384 pagesPathology of Urinary SystemNzau MuangeNo ratings yet
- Pathogenesis of Periodontal DiseaseDocument28 pagesPathogenesis of Periodontal DiseaseAbdullah Seckin ErtugrulNo ratings yet
- Clase Ecm 2021aDocument66 pagesClase Ecm 2021aAndres Camilo Meza AlarconNo ratings yet
- Wound Healing, Tissue Repair, and FibrosisDocument28 pagesWound Healing, Tissue Repair, and FibrosisRibka TheodoraNo ratings yet
- Human Implantation: Embryonic BehaviourDocument33 pagesHuman Implantation: Embryonic BehaviourRaras PraminingrumNo ratings yet
- Bagian Patologi Anatomi Fk-Unsyiah Banda Aceh - Nad 2007Document90 pagesBagian Patologi Anatomi Fk-Unsyiah Banda Aceh - Nad 2007femi_nizaNo ratings yet
- Cedera Sel Dan Adaptasi (Residen)Document77 pagesCedera Sel Dan Adaptasi (Residen)jujuNo ratings yet
- Tissue Repair, Wound Healing and FibrosisDocument48 pagesTissue Repair, Wound Healing and FibrosisIyanAsianaNo ratings yet
- Pathology DKDocument367 pagesPathology DKAshok DaukiyaNo ratings yet
- Wound Healing, Tissue Repair, and FibrosisDocument28 pagesWound Healing, Tissue Repair, and FibrosisRibka Theodora100% (1)
- EmbryoDocument24 pagesEmbryomidhunsekar3541No ratings yet
- Wound and Wound HealingDocument28 pagesWound and Wound HealingObehi Eromosele0% (1)
- Family Micrococcaceae PDFDocument73 pagesFamily Micrococcaceae PDFJean Tsuna KalawNo ratings yet
- CELL PathologyDocument28 pagesCELL PathologySaeed RazaqNo ratings yet
- 05 RepairDocument31 pages05 RepairShameena KnNo ratings yet
- Chap1 Wound HealingDocument35 pagesChap1 Wound HealingAlex HaileyesusNo ratings yet
- Hema Kini BASIC INFLAMMATION RECAP 2022Document49 pagesHema Kini BASIC INFLAMMATION RECAP 2022Shana ShirinNo ratings yet
- Wound HealingDocument76 pagesWound Healingmannkheni209No ratings yet
- Biology of Microfractur E: Dr.V.Chandan NoelDocument25 pagesBiology of Microfractur E: Dr.V.Chandan Noelchandan noelNo ratings yet
- Wound HealingDocument44 pagesWound HealingSorin Niky MocanuNo ratings yet
- First Foundations in Pathology Part 3: Growth and RepairDocument52 pagesFirst Foundations in Pathology Part 3: Growth and Repairlovelyc95No ratings yet
- Wound Healing: Monisha B. Subhashree GDocument35 pagesWound Healing: Monisha B. Subhashree GGayathri MaigandanNo ratings yet
- Pathophysiology of Varicose Vein - Chronic Venous InsufficiencyDocument31 pagesPathophysiology of Varicose Vein - Chronic Venous InsufficiencyDavid Christian100% (1)
- Cor NeaDocument43 pagesCor NeaAdarsh KeshariNo ratings yet
- General Pathology and CytologyDocument110 pagesGeneral Pathology and CytologyJustine Nicole Clavel LachicaNo ratings yet
- Path4 5aDocument89 pagesPath4 5aAbid KhanNo ratings yet
- Biocompatibility: Biological Response Biocompatibility Tests Sterilization IssuesDocument41 pagesBiocompatibility: Biological Response Biocompatibility Tests Sterilization IssuesJASPREETKAUR0410No ratings yet
- Osteogenesis Imperfecta: Vaibhav B K Post Graduate Department of Orthopaedics KVGMCH, SulliaDocument55 pagesOsteogenesis Imperfecta: Vaibhav B K Post Graduate Department of Orthopaedics KVGMCH, Sulliavaibhav gowdaNo ratings yet
- Acute InflammationDocument51 pagesAcute InflammationNayela AkramNo ratings yet
- Physiology of The KidneyDocument35 pagesPhysiology of The Kidneykomalsaharan2001No ratings yet
- Pengantar Patologi AnatomiDocument47 pagesPengantar Patologi AnatomiChristian V. H. TambaNo ratings yet
- Module 1 - Blood Vasculature, Megakaryocytes and PlateletsDocument31 pagesModule 1 - Blood Vasculature, Megakaryocytes and PlateletsI love dem Coffee (Migz)No ratings yet
- Wound HealingDocument70 pagesWound HealingSachin KpNo ratings yet
- Inflammation and Healing: Inflammation "Local Response of Living Mammalian Tissues To Injury Due To Any Agent"Document121 pagesInflammation and Healing: Inflammation "Local Response of Living Mammalian Tissues To Injury Due To Any Agent"Bharath GowdaNo ratings yet
- 4 Regeneration RepairDocument65 pages4 Regeneration RepairMustafa YılmazNo ratings yet
- Woundhealingofcornea 160912145044Document86 pagesWoundhealingofcornea 160912145044Tirza StevanyNo ratings yet
- Cerebro Vascular AccidentDocument55 pagesCerebro Vascular AccidentShalem. KNo ratings yet
- CH 19Document5 pagesCH 19Ryan Carlo IbayanNo ratings yet
- Chronic Inflamation: Bagian Patologi Anatomi FK - UmiDocument35 pagesChronic Inflamation: Bagian Patologi Anatomi FK - UmiTeus FatamorganaNo ratings yet
- Patho Written 108Document11 pagesPatho Written 108Nina Khaled YoussefNo ratings yet
- 2.shedding of ToothDocument30 pages2.shedding of Toothsanofazal786No ratings yet
- Renal Failure en 2Document103 pagesRenal Failure en 2ezz sharabyNo ratings yet
- SAQ Unit 2Document9 pagesSAQ Unit 2Shilpa DuttaNo ratings yet
- Adult Polycystic Kidney Disease: Gerald Schulman, MD, Fasn Vanderbilt University School of MedicineDocument58 pagesAdult Polycystic Kidney Disease: Gerald Schulman, MD, Fasn Vanderbilt University School of MedicineFate ChanNo ratings yet
- Regeneration and Fibrous Repair KuliahDocument16 pagesRegeneration and Fibrous Repair Kuliahakun internetNo ratings yet
- 0828 Introduction and Cellular InjuryDocument74 pages0828 Introduction and Cellular Injury李宜芳No ratings yet
- Cellular Adaptation and NecrosisDocument30 pagesCellular Adaptation and NecrosisHilary Dianne TutoNo ratings yet
- Wound Healing 2022Document53 pagesWound Healing 2022John Patrick Daiz100% (1)
- DR Soekimin SP - PA DR Suryani Eka Mustika SP - PADocument56 pagesDR Soekimin SP - PA DR Suryani Eka Mustika SP - PAAnonymous 7jwKCBRC47No ratings yet
- Haematinics Self-Study - Revision QuestionsDocument1 pageHaematinics Self-Study - Revision QuestionsGeoffreyNo ratings yet
- 4.3.1 MomentumDocument9 pages4.3.1 MomentumIf HassanNo ratings yet
- Notes - Eukaryotic Gene Expression and Regulation & Chromosomal Abberations-1Document80 pagesNotes - Eukaryotic Gene Expression and Regulation & Chromosomal Abberations-1GeoffreyNo ratings yet
- Force QuestionsDocument19 pagesForce QuestionsMohammadTalal277No ratings yet
- Corticosteroids Revision QuestionsDocument2 pagesCorticosteroids Revision QuestionsGeoffreyNo ratings yet
- Revision Questions PharmacologyDocument2 pagesRevision Questions PharmacologyGeoffreyNo ratings yet
- Energy, Work & Power PDFDocument11 pagesEnergy, Work & Power PDFyolanda gaoliNo ratings yet
- 4 PerineumDocument44 pages4 PerineumGeoffreyNo ratings yet
- 2.pelvic Viscera-1Document32 pages2.pelvic Viscera-1GeoffreyNo ratings yet
- Cellular PhysiologyDocument72 pagesCellular PhysiologyGeoffreyNo ratings yet
- Notes - Overview of Genetics - Chromosome Structure & FunctionDocument47 pagesNotes - Overview of Genetics - Chromosome Structure & FunctionGeoffreyNo ratings yet
- 1.pelvis and Perineum-1Document28 pages1.pelvis and Perineum-1GeoffreyNo ratings yet
- 3.pelvis - Peritoneum, Nerves and Blood SupplyDocument30 pages3.pelvis - Peritoneum, Nerves and Blood SupplyGeoffreyNo ratings yet
- Spread of Tumours and EffectsDocument2 pagesSpread of Tumours and EffectsGeoffreyNo ratings yet
- Viral Gastroenteritis ROTA POLIODocument29 pagesViral Gastroenteritis ROTA POLIOGeoffreyNo ratings yet
- Intro. PgyDocument31 pagesIntro. PgyGeoffreyNo ratings yet
- Cell Adaptation and Injury HandoutDocument7 pagesCell Adaptation and Injury HandoutGeoffreyNo ratings yet
- Carcinogenesis HandoutDocument4 pagesCarcinogenesis HandoutGeoffreyNo ratings yet
- Aromaticity: ObjectivesDocument33 pagesAromaticity: ObjectivesGeoffreyNo ratings yet
- Cell Cycle, Growth Factors & Neoplasia HandoutDocument4 pagesCell Cycle, Growth Factors & Neoplasia HandoutGeoffreyNo ratings yet
- Autocoidsmediatorsofinflammation LearningobjectivesDocument1 pageAutocoidsmediatorsofinflammation LearningobjectivesGeoffreyNo ratings yet
- Viral Skin RashesDocument26 pagesViral Skin RashesGeoffreyNo ratings yet
- Respiratory Viruses Influenza and RSVDocument19 pagesRespiratory Viruses Influenza and RSVGeoffreyNo ratings yet
- Brochure COVID-19Document11 pagesBrochure COVID-19GeoffreyNo ratings yet
- Nutrition Temperature Aeration PH: ProvideDocument20 pagesNutrition Temperature Aeration PH: ProvideGeoffreyNo ratings yet
- Sterilisation, Disinfection, and AntisepsisDocument28 pagesSterilisation, Disinfection, and AntisepsisGeoffreyNo ratings yet
- Peptidoglycan: (Also Called Gram's Method)Document20 pagesPeptidoglycan: (Also Called Gram's Method)GeoffreyNo ratings yet
- Definitions: Bacterial Genetics: The Study of Heritable Information inDocument30 pagesDefinitions: Bacterial Genetics: The Study of Heritable Information inGeoffreyNo ratings yet
- Background: Anton Van Leeuwenhoek of Holland Is Known As The Father of Microscopy Simple Microscopes Using Only 1 LensDocument21 pagesBackground: Anton Van Leeuwenhoek of Holland Is Known As The Father of Microscopy Simple Microscopes Using Only 1 LensGeoffreyNo ratings yet
- Understanding Fat OxidationDocument10 pagesUnderstanding Fat OxidationPaulo CesarNo ratings yet
- 1330 Fernando Cervero - Mechanisms of Visceral Pain and HyperalgesiaDocument38 pages1330 Fernando Cervero - Mechanisms of Visceral Pain and HyperalgesiaCaesar Bornie AgustioNo ratings yet
- Career Research Guide WorksheetDocument3 pagesCareer Research Guide Worksheetapi-288548316No ratings yet
- A Ciência Da Esgrima: Implicações Da Performance e Prevenção de LesõesDocument18 pagesA Ciência Da Esgrima: Implicações Da Performance e Prevenção de LesõesDaniel GonçalvesNo ratings yet
- Interstitial Fluid Flow Induces Myofibroblast Differentiation and Collagen Alignment in VitroDocument9 pagesInterstitial Fluid Flow Induces Myofibroblast Differentiation and Collagen Alignment in VitroPedro FonsecaNo ratings yet
- Experiment 1 Cell BioDocument7 pagesExperiment 1 Cell BioWinserng 永森No ratings yet
- Approach To Acid BaseDocument4 pagesApproach To Acid BaseJose Carlos Guerrero AcostaNo ratings yet
- 29 Lab Tech. (MLT)Document17 pages29 Lab Tech. (MLT)Anjali JoshiNo ratings yet
- Modul Bronkoesofagologi - 6. Disfagia OrofaringDocument31 pagesModul Bronkoesofagologi - 6. Disfagia OrofaringNi Putu Apriliantini Arleni Putri 1802511045No ratings yet
- Mitral Valve ProlapseDocument5 pagesMitral Valve ProlapseDerofiez Hana RuhyadinNo ratings yet
- Barotac Nuevo National Comprehensive High SchoolDocument2 pagesBarotac Nuevo National Comprehensive High SchoolVon AlexisNo ratings yet
- Reseptor Kanal Ion Target ObatDocument65 pagesReseptor Kanal Ion Target ObatFitry Ana UtamNo ratings yet
- Qrs Complexes: Fast & Easy Ecgs - A Self-Paced Learning ProgramDocument49 pagesQrs Complexes: Fast & Easy Ecgs - A Self-Paced Learning ProgramMuhammad Hatta HamzahNo ratings yet
- MeiosisDocument20 pagesMeiosisRosalina ColetoNo ratings yet
- The Brain and Cranial NervesDocument82 pagesThe Brain and Cranial NervesImmanuel Ronald LewisNo ratings yet
- CSI Physical ExamDocument3 pagesCSI Physical ExamSympaschoNo ratings yet
- Electrical Double Layer in Biological MembranesDocument6 pagesElectrical Double Layer in Biological MembranesJuan LopezNo ratings yet
- Puberty and Tanner StagesDocument2 pagesPuberty and Tanner StagesDewi LarasatiNo ratings yet
- Head and Neck Block HandbookDocument50 pagesHead and Neck Block HandbookGrace Poon OnionNo ratings yet
- Practice Test 2 1st Sem 2013Document11 pagesPractice Test 2 1st Sem 2013Isabel Barredo Del MundoNo ratings yet
- Chap001 Test Bank Tank PDFDocument45 pagesChap001 Test Bank Tank PDFHeraNo ratings yet
- Respiration Lab 10Document3 pagesRespiration Lab 10Jâmãçy BräthwåítèNo ratings yet
- Insecticide ClassificationDocument10 pagesInsecticide ClassificationAnuragBhatnagarNo ratings yet
- IB Biology HL SyllabusDocument9 pagesIB Biology HL SyllabusjessicaNo ratings yet
- Valorization of CoffeeDocument9 pagesValorization of CoffeeEditor IJTSRDNo ratings yet
- Adaptations of Cellular Growth & Differentiation: @2022 PreclinicalsDocument27 pagesAdaptations of Cellular Growth & Differentiation: @2022 PreclinicalsJames MubitahNo ratings yet
- KaoshikiiDocument24 pagesKaoshikiiapi-238078813No ratings yet
- Biology 209 Lab ManualDocument128 pagesBiology 209 Lab ManualikomalNo ratings yet
- Prostho Vi - Slides 5 - Occlusal - ConsiderationsDocument83 pagesProstho Vi - Slides 5 - Occlusal - Considerationsبراءة أحمد السلاماتNo ratings yet
- Forces in BodyDocument48 pagesForces in Bodytema_racamaNo ratings yet