Professional Documents
Culture Documents
User Com: Measuring Specific Heat Capacity
Uploaded by
rubenssanOriginal Title
Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
User Com: Measuring Specific Heat Capacity
Uploaded by
rubenssanCopyright:
Available Formats
USER COM
Information for users of
METTLER TOLEDO thermal analysis systems
June 1998
Dear customer
We are always being asked what
we do with your ideas and
suggestions. In this edition of
UserCom we would like to
explain how we take note of your
wishes and incorporate them in
new products. We count on your
Measuring specific
heat capacity 7
TA TIP
understanding if we can not 1. Measuring specific heat capacity
immediately take your particular The specific heat capacity, often referred to as the specific heat, is a measure
wish into consideration. We of the amount of energy required to raise the temperature of 1 g (or 1 mole)
welcome your comments because of a substance by 1 K. The symbol used is cp, or Cp for the molar quantity.
we are convinced that with your The ‘p’ in the lower position stands for ‘at constant pressure’. The SI units
cooperation we can improve the for the specific heat capacity are J g-1 K-1 (also J/gK) or J mol-1 K-1 (also
quality of our products even more. J/mol K).
Usually the specific heat is not measured during first order transitions in the
physical state (e.g. melting), where it can be infinitely large.
The values for solids and liquids range from 0.1 to 5 J g-1 K-1. The specific
heat of most substances increases with increasing temperature; the
measurement is therefore often performed over a relatively large
temperature range.
Contents Some examples at 25 °C:
TA-TIP Substance Alumina Aluminum Lead Polystyrene Water
- Measuring specific heat capacity
Wert [J g-1 K-1] 0.895 0.776 0.129 1.3 4.18
NEW in the sales program
- Furnace expander With the STARe Software, 4 different possibilities for the determination of
- IO/OQ the specific heat are available, each with its own advantages and
disadvantages. The various methods will be described later on in detail. First
Applications of all, here is a survey of the various temperature programs:
- Decomposition of ammonium
perchlorate
- Characterization of soil samples
Direct method Steady state ADSC Saphire method ADSC
- Precise measurement of the specific
heat capacity T T T T
- Safety investigations in glass
crucibles
- Determination of curing behaviour
- Platin crucibles in thermogravimetry t t t t
USER COM June 98 1
1.1 General information about mechanical stability. This can lead to Weighing accuracy
measurement conditions a deformation of the crucible when it Weighing errors are important in any
is used repeatedly in the sample cp measurement. A microbalance
DSC cell changer (reference crucible). In such as the MT5, which was used
The FRS5 must be well ‘burned-in’ addition the crucible is unsuitable for for all the measuremnts in this work,
(e.g. 10 minutes at 600 °C with air use with volatile samples because it is best.
flow in order to remove any volatile can not be hermetically sealed.
impurities). After this cleaning Blank curves
process the sensor sits very tightly, Reference crucible All accurate cp measurements should
which is a further requirement for The reference crucible remains in the be blank curve corrected. The
accurate cp measurements. Any ash same position for all measurements. measurement of several blank
or dust is carefully blown away with If it does not have a centering pin curves is recommended before the
compressed air (dust under the then its exact orientation should be sample measurements. The first two
crucible leads to poor reproducibility checked visually from time to time. are to condition the measuring cell.
of DSC curves). With the sample An atmosphere of static air or 50 ml The third is used for the calculation
changer, the 34 recesses on the nitrogen per minute is usual. (the STARe Software always uses
turntable must be freed regularly the most recent blank curve of the
from any dust. Sample preparation relevant method; if this is an outlier
Good contact with the bottom of the it should be deleted from the
Temperature calibration crucible is important. The situation is database).
Since the specific heat is a function ideal with liquid samples and with
of temperature, the temperature flat disks. Powder samples of Heating rate
should be calibrated (checked) in the medium particle size (about 0.1 mm) A heating rate of 10 K/min is
measurement range to be used and if are also good. Very fine powders normally used for classical cp
necessary adjusted. In addition, the often contain a lot of air, which measurements. Time-saving heating
heating rate enters into the result in makes them poor thermal rates of 20 K/min give good results.
both the direct and the steady-state conductors; they can usually be A larger temperature range is
methods. The heating rate is only compacted with a teflon rod. required because about 1 minute is
right if the temperatures are correct. Samples of low thermal conductivity needed to reach a state of dynamic
give cp values that are about 5 % too equilibrium. Reliable cp values can
Tau lag adjustment low. It is important that the bottoms only be obtained in this region.
This is a prerequisite for accurate of the crucibles are flat and not
melting point determinations, but is deformed.
of no great importance for the
accuracy of cp measurements. Sample quantity
The size of the measurement signal
Type of crucible is propotional to the sample amount.
Up until now most experience has This means, in principle, with cp
been gained with the 40 µl standard measurements (except ADSC): the
aluminum crucibles. Previous larger the sample the better. One
measurements with the tall Al should try to obtain a signal of at
crucibles (160 µl) gave poorer cp least 5 mW (maximum about 10
values in spite of the increased signal mW). This requires a 30 mg sample
obtained with the larger sample with a cp of 1 J/gK at a heating rate
amount. The new lighter Al crucibles of 10 K/min.
(20 µl) are advantageous because the For cp measurements with sinusoidal
sample can be pressed down against temperature modulation, signal
the bottom of the crucible, thereby amplitudes of 1 mW are optimal;
improving the thermal contact. This with a temperature program of
is particularly important for amplitude of 1 K and a cycle time of
temperature modulated methods. The 1 min this corresponds to a sample
disadvantage is the poorer of about 15 mg.
2 USER COM June 98
1.2 The direct method 3 %. A more accurate function (reject the first two blanks). The
This is based on the ‘dynamic with a maximum error of 1 % in measurement uncertainty is
definition’ of the the specific heat this temperature range is being therefore ±2 % with a sample
and is equal to the heat flow HF developed. At much lower or signal of 5 mW.
divided by the heating rate ß and the higher temperatures the error is
sample mass m. somewhat larger. Total error (only those that can not
be avoided according to sections 2.
to 4. above): the total error is
Heat Flow, approximately equal to the square
HF, mW zero line root of the sum of the squares of the
0 individual errors i.e. the square root
blank curve of 1 + 22 + 32 = 3.7 % (with the new
Erel temperature function the square
HFraw HF HF root of 1 + 22 + 1 = 2.5 %). In
-2 cp =
m .β addition the sample weighing errors
must also be taken into account.
sample curve, Series of measurements in the range
-4
bs 50 °C to 200 °C with samples of
transient good thermal conductivity show
period sample curve errors of -5.5 % to +2.0 %.
iso dynamic temperature program time, t
1.3 The ‘steady state’ ADSC
Fig. 1: The heat flow HF is obtained from the raw DSC signal HFraw by blank In this method the sample is
curve subtraction (bs). The evaluation should begin after the state of dynamic subjected to a periodic heating and
equilibrium has been reached. cooling program (sawtooth). The
heating and cooling rates lie between
Possible errors: 4. Reproducibility of the sample 2 K/min and 10 K/min and need not
1. Blank curve correction: this is and blank curves: the be identical. A segment should last at
essential because an assymetry reproducibility of the blank least 2 minutes so that the sample is
of up to about 2 mW can occur. curve corrected measurement in a state of dynamic equilibrium
Compared with a sample signal curve is around ±0.1 mW with (steady state). Samples of poor ther-
of 10 mW, for example, an error the FRS5 sensor. These values mal conductivity require 4 to
of 20 % would arise if the blank are valid for temperature range 5 minutes. The evaluation routine
curve correction was not applied. of 50 °C to 200 °C and for a Math/Steady State ADSC Cp
2. Accuracy of the calorimetric preconditioned measuring cell performs a cp determination towards
calibration (EIn): an error limit of
1 % is aimed for. The weighing
error may therefore only be Heat Flow,
about 0.1 % (5 µg in 5 mg). In HF, mW HF
1 cp =
particular, when using the auto- m. β
matic sample changer, it is
recommended that the previously HF HF
blank
flattened indium sample be curve
0
pressed tightly in the middle of
the Al standard crucible with a HF HF HF sample
curve
teflon rod (place the crucible on
a hard, absolutely flat surface, T
e.g. a glass plate). This ensures
that the sample remains in the
middle of the crucible even when Time, t
measurements are performed
repeatedly.
3. Temperature function of Erel: the Fig. 2: The sawtooth temperature program shown below produces the DSC curves
response curve between 0 °C and shown above. The blank curve is subtracted from the sample curve, so that the
250 °C that is stored in the STARe evaluation method Math/Steady State ADSC Cp can convert the difference of the
Software has an error limit of heat flows HF to cp.
USER COM June 98 3
the end of each segment (analogous Possible errors: 1.5 ADSC with a sinusoidal
to the direct method). The blank The saphire cp standard technique temperature program
curve is measured with an empty makes errors in EIn and Erel irrele- In alternating DSC (ADSC) the
crucible of the same mass as for a vant. temperature is varied sinusoidally as
sample in order to avoid additional There is only the reproducibility of a function of time and is
smoothing of the automatic blank the sample and the saphire curves to superimposed on the average heating
curve (identification e.g. blank curve consider. With the FRS5 sensor, the rate. The heat capacity of the sample
sample weight 0 mg). reproducibility of the blank curve gives rise to a periodical
corrected measurement curve is measurement signal. The blank
Possible errors: ±0.1 mW. This value is valid for the curve corrected amplitude is propor-
The possible errors are similar to temperature range 50 °C to 200 °C tional to the heat capacity.
those of the direct method. The with a preconditioned measuring cell A heat capacity calibration standard
measurement signals are somewhat (reject the first blank). The is required just as with the saphire
smaller due to the lower heating measurement uncertainty with a method. It consists of the lid of the
rates. The reproducibility of the DSC sample signal of 5 mW is therefore crucible. The cp temperature
signal is however better because of ±2 %. functions of all Mettler crucible
the shorter segments. With the new The total error (square root of the materials are stored in the STARe
Erel temperature function we expect sum of the squares of the individual database. There are 3 measurements
an error of about 2.5 %. errors) is the square root of (22 + 22) to be performed:
= 2.8 %. In addition, the weighing
1.4 The saphire method errors of the sample and the saphire 1. Blank (crucible without lid on
The saphire method for cp must be taken into account. both sides, the reference crucible
determination has been used for Series of measurements with remains in the same place for all
more than 30 years. The DSC signal samples of good thermal measurements),
of the sample is compared with the conductivity gave errors of -1.4 % to 2. Calibration (crucible with lid,
DSC signal of the calibration sample +0.7 % in the range 50 °C to 200 °C. the difference in mass compared
of known specific heat. Both curves It is advantageous to measure with the crucible of the blank
are blank curve corrected (automatic segments not larger than 100 °C to measurement is entered as the
blank curve correction). 200 °C. If a wider temperature range sample weight),
A total of three measurements are is of interest, then it is better to 3. Sample (in the crucible with lid
made: blank (empty crucible), divide it up into segments of with similar mass (difference <
saphire (3 small saphire disks of maximum 200 °C seperated by 20 µg) as for the calibration).
4.8 mm diameter, ME 17 558 as the isothermal segments of 5 minutes. The evaluation routine Math/
calibration sample) and the sample The evaluation can use as many ADSC Cp determines the ampli-
itself. A blank curve correction is in segments as desired. tude using Fourier analysis. In
this case also essential. addition to the formula shown in
figure 4, the signal damping is
compensated mathematically by
Heat Flow, the phase change of the sample
HF, mW interpolated baselines measurement.
0
HF . m sap Possible errors:
cp = . cp
HFsap m . HF sap
sap The error possibilites are similar to
HF those of the saphire method. The
-2
calibration heat capacity is however
quite small (corresponds to the mass
sample curve,
bs
of the lid). It is better to use smaller
-4 sapph samples with ADSC in order to keep
ire cu
rve, b the signal damping low. For the
s
same reasons high frequencies are
Iso dynamic temperature program iso t not used. Normally the cycle time is
not less than 1 minute. The average
Fig. 3: The blank curve corrected sample and saphire measurements are shown. heating rate lies between -5 K/min
The interpolated baseline between the isothermal levels at the start and end and +5 K/min (the maximum heating
temperatures is automatically calculated in order to improve the accuracy of the and cooling rates of the method must
heat flow HF. The literature values for the cp of saphire as a function of be lower than the maximum heating
temperature function are included in the evaluation program. and cooling rates of the modules).
4 USER COM June 98
An amplitude of 1 K is normally
used.
The longer measurement times (3 to Heat Flow, blank curve (As -AAl) . m Al .
cp = cp
10 times), compared with the saphire HF, mW aluminum curve m . (AAl-Ab ) Al
method, are only justified if the sample curve
specific heat is to be measured (also AAl As
isothermally) during chemical reac- Ab
tions or certain physical transitions. 0
Please note: the Al crucibles are only
suitable for certain chemical
reactions.
-2
Series of samples with good
conductivity gave errors in the range
-2.5 % and +3 %.
Iso modulated temperature program t
Fig. 4: The evaluation routine Math/ADSC Cp determines the phase-correct
amplitude differences ( 1. blank curve, 2. calibration curve, 3. sample curve). The
equation for the cp determination includes the difference of the amplitudes because
the crucible lid that serves as the cp standard is also measured with the sample.
1.6 Comparison of the four different methods
Direct Steady State Saphire ADSC (Sine)
Principle heat flow divided by comparison with a cp standard that is subjected to the
the heating rate and sample mass same temperature program
Accuracy
attainable ±5 % 1) ±4 % 1) ±2 % ±3 %
Disad- • not very • not very • 3 measurements • 3 measurements
vantages accurate accurate required, can be required, long
• long measurement run at 20 K/min measurement time
time however • frequency dependent
Advantages • shortest • yields also • highest accuracy • yields quasi isothermal cp
measurement quasi isothermal cp • for standards: • cp also during non-
time DIN 51007 reversing reactions
ASTM E 1269 • in modern trend
Automatic yes no yes no
blank curve
Software specific Method window, specific heat Method window, Relative
options heat Relative loop, loop, ADSC Cp
required Mathematic, ADSC
1
) With the new Erel temperature function (in version 5.12) of the DSC sensor FRS5, 2.5␣ % to 3␣ % can be attained.
USER COM June 98 5
Furnace expander
With the replacement of the DSC25 safety investigations. The solution to with the automatic furnace lid which
measuring cell, we were faced with the problem is the Expander (ME 51 must be removed for measurements
the problem that that the large 140 735) which can be mounted on with the expander. If the DSC821e
crucibles (height > 7.0 mm) could the DSC821e furnace. This lengthens module is equipped with the sample
no longer be used with the new the furnace by 12.0 mm. A manual changer then this must be set to the
DSC821e module. The large lid of the same height guarantees side. Otherwise the the sample
crucibles had, however, in the past good performance. changer rests on the furnace lid.
proven to be be most useful for The furnace expander does not work
IQ/OQ (Installation Qualification /
Operational Qualification)
An IQ/OQ binder is included with all new software software) are recorded 4. IQ: Proof of Personel Training
our modules from June 1998 in the binder, so that you have up-to-5. OQ: METTLER TOLEDO Stan-
onwards. This binder documents date information on the current state dard Test Procedures
how we tested the instrument of your module. 6. OQ: Short Test of the TA System
together with the corresponding It is of course possible to ‘update’ with General System Suitability
results and adjustment parameters. your existing module with this Test
The module will again be calibrated binder. The IQ/OQ binder is 7. PQ: Logbook
(checked) and, depending on the available in three languages German, 8. PQ: Periodic Testing of the TA
deviation, adjusted at the initial English and French (ME 51709754 System with Specific System
customer installation by METTLER (G), ME 51709755 (E), Suitability Test
TOLEDO. The results are then also ME 51709756 (F)). 9. PQ: Disposal
recorded in the binder. 10. Calibration Certificate
Further calibrations and adjustments 11. Validated Method
will be performed from time to time Table of contents 12. Service Contracts and Contact
depending on your requirements. 1. Purpose of the Thermal Analysis address
The results are then also documented Logbook 13. Document Control
in the binder. 2. CQ: System Test before Deliver 14. Certifications
In addition, any changes to the 3. IQ: Installation of the System and 15. blanc forms
module itself (additional options or Instrument Configuration
Procedure for processing customer
If we receive a comment from you (through a salesman, a service engineer or directly) we decide first of all
whether it is a problem or whether it is something you would like.
• We divide the problems into two main categories: major problems and minor problems. The major problems are
dealt with immediately and a new software version is made.
We consider minor problems to be those that do not lead to false results and for which there is usually an
acceptable short-term solution i.e. a so-called ‘work around’. These problems are set at the top of the list of
specifications for the next version.
• Your wishes and suggestions are also entered in a separate list. We always try to formulate wishes for specific
features in a more general way so that as many users as possible can benefit from the change, once it has been
made.
These general wishes are prioritized and, depending on the resources and time available, also entered in the
specifications for the next version.
We will inform you in each edition of UserCom about the comments and suggestions that have led to
improvements such as, for example, the calculation of Young’s modulus using DLTMA methods (UserCom6).
6 USER COM June 98
Model free kinetic analysis of thermal
decomposition of ammonium
perchlorate
Sergey Vyazovkin and Charles A. Wight
Department of Chemistry , University of Utah, Salt Lake City, Utah, 84112
The kinetics of the thermal of the thermal decomposition of AP The thermogravimetric analysis
decomposition of the cubic were based on the assumption of a (TGA) experiments were carried out
ammonium perchlorate (AP) has constant activation energy. The using a TGA/SDTA851e instrument.
been extensively studied. The isoconversional method, which is the Samples of AP were placed in alu-
reported effective activation energies basis of Mettler-Toledo’s kinetic minum pans and heated in a flowing
vary from about 371 to software “Model Free Kinetics”,4 atmosphere of nitrogen (100 ml/
2602 kJ mol-1. The mechanistic allows one to reveal the dependence min). The TG experiments were
interpretation of the values is also of the activation energy on the extent carried out at heating rates 5, 7.5, 10,
different. The confusing character of of conversion and/or on the 12.5, and 15 °C/min.
the kinetic information is not temperature. The analysis of such a At 240 °C DSC curves (Figure 1)
surprising for the process that is dependence is usually very helpful in show an endothermic peak
known3 to be a tangled interplay of drawing mechanistic conclusions corresponding to the transformation
various chemical (solid-state about the process5. In this study, we of AP from orthorhombic to cubic
decomposition, reaction of gaseous apply the model free kinetic analysis form. It is followed by an
products with the solid, gas-phase to TGA data on the thermal exothermic feature due to the ther-
reactions) and physical (diffusion, decomposition of cubic AP. mal decomposition. The thermal
sublimation, adsorption-desorption) AP (Aldrich) was used as supplied. behavior of AP is markedly different
processes. The effective activation The differential scanning calorimetry in open and closed pans. In a closed
energy of the thermal decomposition (DSC) experiments were performed pan, the thermal decomposition of
of AP is a composite value using a DSC821e. Samples of AP (2 AP is completely exothermic. In an
determined by the activation - 3 mg) were studied in both open open pan, the exothermic
energies of these processes as well as and closed (pierced) aluminum pans. decomposition is overtaken in the
by their relative contributions into
the overall decomposition rate. If
these processes have different
activation energies, the effective
activation energy shows a variation
with the temperature. The kinetics of
such multi-step processes cannot be
characterized by a single constant
value of the activation energy. The
latter is traditionally evaluated by
equation 1:
dα − E
= A exp f (α ) (1)
dt RT
where A (preexponential factor) and
E (the activation energy) are
Arrhenius parameters, alpha is the
extent of conversion, f (α) is the
reaction model, t is the time, T is the
temperature, and R is the gas Fig.1: DSC curves of AP at a heating rate of 5 K/min in hermetically sealed and in
constant. The earlier kinetic Al standard crrucibles with pierced lids. At 250 °C the solid-solid transition to the
evaluations of the activation energies cubic modification occurs. Decomposition reactions follow afterwards.
USER COM June 98 7
later stage by an endothermic event.
Analysis of the literature DTA/DSC
data6,7 shows that the second peak
appears as exothermic under
conditions that retard the escape of
reaction gases. Conversely, under
conditions that facilitate the removal
of gaseous products the second event
appears as an endothermic peak
which is atributed to dissociative
sublimation of AP to ammonia and
perchloric acid. By comparing DSC
and TG curves (Figures 1 and 2) we
can see that the point when
decomposition turns into
endothermic stage is perfectly
correlated with the slowdown
(α = 0.30 to 0.35) observed in TG
experiments. The 30 % to 35 %
decomposed AP has a highly porous Fig. 2: TG curves of AP shown with heating rates of 5 to 15 K/min.
structure8.
Therefore, the solid-state thermal
decomposition of AP involves two
major stages.
The first stage (α < 0.3) is
exothermic decomposition that
results in formation of porous AP.
The second stage is endothermic
dissociative sublimation of AP. Sub-
limation and exothermic
decomposition are most likely to
occur throughout the whole process.
The two major steps of the decom-
position are mirrored in the depen-
dence of the activation energy on the
extent of conversion (Figure 3). The
exothermic decomposition
(α = 0 to 0.3) results in development
of the high surface area that favors
physical processes of diffusion and
adsorption and that hinder the escape Fig. 3: The conversion curves calculated from the TG curves of the various
of gaseous decomposition products. heating rates, and the dependence of the activation energy on conversion
As a result, the exothermic calculated using model free kinetics.
decomposition becomes controlled
by the slow mass-transfer processes
which are characterized by low
activation energies. This explains
slowing the mass loss process
(Figure 2) as well as decreasing the
effective activation energy (Figure 3)
while α increases to 0.3. At small
extents of conversion, the process is
kinetically controlled. The effective
8 USER COM June 98
value of the activation energy E decomposed AP is 1000 times 242 ±0.8 kJ mol -1. Because the
(115 kJ mol -1) is an estimate for the greater than that of the original AP. activation energy of sublimation
activation energy of the thermal Then, we may expect that by α = 0.3 (125 kJ mol -1) is greater than that
decomposition of AP. At α = 0.3, the the subli-mation increases 1000 for the mass-transfer controlled
exothermic decomposition still times as compared to that at the decomposition (about 80 kJ mol -1),
prevails over sublimation but slows beginning of the decomposition. the relative contribution of the sub-
down markedly. Therefore, the This seems to be the major reason limation into the overall process rate
effective value of the activation why sublimation ultimately takes increases with the temperature.
energy E (80 kJ mol -1) can be used over the slow exothermic Consequentl, the effective value of
as an estimate for the activation decomposition controlled by mass- the activation energy E
energy of the mass transfer process transfer. Nevertheless (125 kJ mol -1) that corresponds to
responsible for the removal of the exothermic decomposition is the maximum temperature should be
gaseous products. most likely to accompany used as an estimate for the acti-
The development of the high surface sublimation. The coexistence of vation energy of sublimation. In
area promotes transformation of the these two processes is supported by accord with earlier theoretical and
process from the exothermic to the the fact that the endothermic effect experimental findings11, this value
endothermic regime at α > 0.3. The estimated by DSC (140 kJ mol -1 is about one-half the enthalpy of
rate of sublimation is directly pro- after correcting for the initial mass sublimation.
portional to the surface area. loss) is only about 60 % of the Therefore we can propose a rela-
According to some data9, the surface experimentally determined10 tively simple kinetic scheme for the
area of the 30 % to 35 % enthalpy of sublimation, thermal decomposition of cubic AP:
Gaseous products
Transport controlled decomposition
Decomposition (exo-)
EA= 80 kJ mol-1
EA=115kJ mol-1 Gaseous products +
AP
porous AP Dissociative sublimation
EA= 125 kJ mol-1
NH3 + HCIO4
This example shows how model free kinetics can assist the clarification of reactiom mechanisms.
References
1 Brill, T. B.; Brush, P. J.; Patil, D. G. Combust. Flame 1993, 94, 70.
2 Kishore, K; Pai Verneker, V. R.; Krishna Mohan, V. Thermochim. Acta 1975, 13, 277.
3 Jacobs, P. W. M.; Whitehead, H. M. Chem. Rev. 1969, 69, 551.
4 Kelsey, M. S. Am. Lab., January, 1996, p. 13, Datenblatt Modellfreie Kinetik (ME 51 724 783)
5 Vyazovkin, S. Int. J. Chem. Kinet. 1996, 28, 95.
6 Morisaki, S; Komamiya, K. Thermochim. Acta 1975, 12, 239.
7 Tompa, A. S. Thermochim. Acta 1984, 77, 133.
8 Boggs, T. L.; Kraeulte, K. J., Combust. Sci. Technol. 1969, 1, 75.
9 Chang, F.-M.; Huang, C.-C.; Yeh, T.-F.; Liu, C.-S.; Leu A.-L. Propellants, Explosives, Pyrotechnics 1990, 15, 261.
10 Inami, H. S.; Rosser, W. A.; Wise, H. J. Phys. Chem. 1963, 67, 1077.
11 Jacobs, P. W. M.; Russel-Jones, A. J. Phys. Chem. 1968, 72, 202.
USER COM June 98 9
Prospects for the use of thermo-
gravimetry for the characterization of
soil and ecosystems
Dr. Ch. Siewert, Institute of Ecology and Soil Science, Technical University Berlin
Soil plays a central role in strategy in the evaluation of consists of heating from 25 °C to
agriculture and forestry and also in thermogravimetric results. For about 1000 °C in an air flow of the
problems concerning environmental instance, because of the infinite same humidity. Large sample
protection, waste utilization and variety of chemical components and volumes (> 500 mg to 2 g depending
many ecological problems. One of structures in soils, no individual on the size of the crucible) are
the most important characteristics of components are analyzed from the recommended because of the hetero-
soil is the organic soil substance (in dynamics of weight loss steps from geneous nature of the soil and the
the original article in German, ‘orga- peaks in the OBS quality assessment. limited possibilies for homoge-
nische Bodensubstanz’ OBS). It is This is replaced by relationships nization. The heating rate is chosen
the basis of plant nutrition and is of between weight losses in relatively depending on the sample weight so
decisive importance for many che- large temperature ranges. They that the sample is heated uniformly
mical, physical and biological soil describe particularities of biological over the whole temperature range.
properties. It influences the behavior processes in contact with the mineral The best results up until now have
of pollutants substances and plays a parent materials for soil formation. been achieved with a heating rate of
key role in the material cycle of By this means, a description of 5 K/min with sample weights of
ecosystems. quality corresponding to the laws of about 1 g using the METTLER TO-
For several decades, people have soil development is possible without LEDO TGA/SDTA851e. The latter
been looking hard for ways that are a detailed analysis of the chemical can be especially recommended for
generally applicable for its composition. soil analysis. The combination of
characterization. Up until now all The thermogravimetric analysis of large crucible size (maximum
attempts at determining the quality OBS uses soil samples that have sample volume 900 µl) with a
of soil have proven to be been air dried and sieved at 2 mm sample changer and the high
unsatisfactory. The reasons are the particle size (standard preparation) accuracy and versatility of the data
extremely large soil-specific variety after previous conditioning at handling system make the equipment
of material components, the defined humidity. The analysis itself very suitable for the analysis.
heterogeneity of transformation
processes and the continuously Thermal degradation dynamics of organic soil
Components identified
substance and correlation of components
changing adaptations of soil mean weight loos dynamics
temperature range
organisms. Because of this, methods Carbonates and carbonate
A B C D dependent components
and results of the analysis of OBS loss of water thermal decomposition decomposition of clay dependent components
and its components are hardly free bound of organic substances (OS) carbonates (water and humus substanes)
biologically convertible
Weight loos
comparable with each other. They components
in %
uncertain correlation +
have not been applicable to current
or humified (brown)
difficultly degradable (green)
free
bound
easily degradable
data scatter
tasks of soil protection and are not In comparison with variations
through:
suitable tackling future projects in incubation under
laboratory conditions
ecological (e.g. risk assessment of
seasonal
the use of genetically changed variations
organisms in agriculture). Uncertain correlations
a hagroscopicity of carbonate
A thermogravimetric method for the b buffered exoenzymes
assessment of the quality of organic d clay bound convertible substance
Hygroscopicity of carbonates
substance in soil has now been c variation of the soil bilomass
e vegetation dependent variations of
developed as part of a federal dificultly convertible OS
research project at the Technical
University in Berlin. It is based on a (temperature °C)
modified approach to the analysis of Fig. 1: Thermal decomposition dynamics of soil samples of organic soil substance
soils and makes use of a new and correlation of components
10 USER COM June 98
According to a patented procedure of ecologically low risk technologies thermogravimetric curves includes
(GO 1 N 196 38 731) the recorded for land use which are themslves the recognition of soil specific
weight loss dynamics allow the economically attractive. Further decomposition processes of organic
amount of convertable substance and application possibilities of the OBS soil substances using special test
the amount of humus material to be quality assessment concern the criteria. To do this, characteristic
determined. In addition the effect of detection of foreign carbon (e.g. components of the OBS (amount of
the humus material on the from coal and slag) and of bound water, biologically
convertable substance can be disturbances of the adaptation ability convertable components, humified
quantified using the amount of of transformation processes of components) are determined by
bound water. The amount of ecosystems through xenobiotic means of weight loss in certain
convertable substance and of bound substances, the diagnosis of soil temperature ranges. In natural soils
water are finally combined formation processes and the these components are dependent on
mathematically to give the
thermogravimetric OBS quality
index.
Information concerning the carbon,
nitrogen, clay and carbonate
concentrations of the samples can
also be derived from the weight loss
dynamics. As far as accuracy is
concerned, these thermogravimetric
determinations of the most important
soil properties are - as opposed to for
example ignition loss determination -
comparable with standard methods
(such as elementary analysis) and
much more economical. They have,
however, up until now, only been
applicable to mineral soils.
The application possibilities of
thermogravimetric OBS quality
determination are extremely varied. Fig. 2 TGA curves of three different soil samples measured at 5 K/min
Currently, priority has been given to
the investigation of possibilities of
describing particularities of material worldwide homogenization of one other within given limits. If no
cycles of ecosystems in different classification features of soils, the inadmissible deviations occur
climatic zones. As the first results explanation for the causes and side through the presence of substances
show, these, as well as the changes effects of individual soil formation foreign to soil, then the weight losses
brought about by land and forestry processes, the quality assessment of in other temperature ranges are
use can be characterized by the compost and garden soil and many determined and with the help of
thermogravimetric OBS quality other questions from the research regression parameters converted to
index and will allow an experimental fields of soil science, forestry, values for the C-, N-, clay and
quantification of the lasting effects agriculture, ecology and carbonate content (Fig. 2). Finally
of new technologies for land evolutionary biology. the weight losses of convertable
utilization and soil protection. In this components and the of bound water
way, it should be possible to form Three examples of soil samples are combined mathematically to the
the basis for the rapid development The initial evaluation of the OBS quality index.
Literature:
• Siewert, C. (1994): Ökosystemorientierte Grundlagen der Humusqualitätsbestimmung. Teil 1: Theoretisches Kon-
zept zur Ableitung ökosystemarer Humusfunktionen. Archiv für Acker-, Pflanzenbau und Bodenkunde, 38,
127-147
• Siewert, C. (1995): Ökosystemorientierte Grundlagen der Humusqualitätsbestimmung. Teil 4: Besonderheiten der
Atmungsdynamik unterschiedlicher Böden nach Lufttrocknung. Archiv für Acker-, Pflanzenbau und Bodenkunde,
39, 131-153
• Walter, H. (1968): Die Vegetation der Erde in ökophysiologischer Betrachtung. Band II: Die gemässigten und
arktischen Zonen. Fischer Verlag, Jena
USER COM June 98 11
Precision measurements of the speci-
fic heat capacity with the temperature
modulated DSC (TMDSC) at lower
temperatures
P. Mesquida, S. P. Krüger, J. K. Krüger
Department of Physics, B 38, University of Saarland, D 66123 Saarbrücken
The specific heat capacity cp is one entropy production. TMDSC has temperature range that is not
of the most important quantities used therefore, at least in principle, all the normally accessible to purely
to characterize the thermodynamic characteristics of a precision commercial DSC instruments. PDP
properties of materials in the measurement method. shows a weak first order ferroelectric
condensed phase. This is especially The application possibilities of com- phase transition at Tc = 122.7 K.
true for the static and dynamic mercial DSC instruments for the Precision cp(T) investigations
changes in properties in structural investigation of phase transition performed using adiabatic
phase transitions and glass phenomena are limited mainly by calorimetry in the region of the
transitions. Prominent examples of their thermostat systems. This is true transition temperature are reported in
such transition phenomena that are for the temperature range, the the literature for PDP [2]. Figure 1
in the forefront of current research measurement accuracy and the time shows a comparison of the literature
activities are ferroelectric and during which uninterrupted data with our own data which was
ferromagnetic phase transitions, measurements are possible (time recorded with the time domain
transitions in liquid crystals and domain [1]). TMDSC. Curve (a) was measured
superconductors, glass transitions in Most of the investigations described with the following parameters:
structural and classical (canonical) here were performed with a modified heating rate (r) = 0, amplitude (a) =
glasses etc. The transition Mettler-Toledo DSC821e calorimeter 0.02 K, period (p) = 30 s. It is known
temperatures of interest lie typically with ADSC option and a new FRS5 from the literature that even at nor-
between a few degrees Kelvin and ceramic sensor. The modifications mal pressures Tc lies very close to
many hundreds of degrees Kelvin. (done in our institute) concern only the tricritical point [3]. At the
The cp investigations in the region of the cooling system of the instrument. tricritical point the first order phase
phase changes can be roughly A pre-chamber vaporizer cryostat change changes into a second order
divided into two classes: costly and was developed that could be phase change.
time-consuming precision operated with both liquid ntrogen The marked rise of cp(T) in the low
measurements using adiabatic or and liquid helium. The furnace with temperature phase approaching Tc
‘alternating current’ (AC) its heating system and the DSC indicates the closeness to the
calorimetry, and survey sensor were retained and integrated tricritical point. The sharpness and
measurements using conventional in the new cooling system. The new height of the cp anomaly shown in
DSC. The purpose of this work is to heat exchanger allows the furnace to figure 1 demonstrates both the high
show that ‘temperature modulated’ be cooled down to less than 30 K. At quality of the crystal and also the
DSC (TMDSC) provides an the same time, the temperature of the excellent quality of the measurement
excellent alternative to the above surroundings of the furnace can be technique.
mentioned precision methods. The kept constant to within several Figure 2 demonstrates the ‘mean
measuring principle of TMDSC hundredths of a degree. field’ behavior of the phase transition
allows investigations at quasi Nevertheless, even at 80 K, cooling observed as well as the closeness of
constant temperature (temperature rates of 10 K/min are still possible. Tc to the tricritical point. In this
modulation at a heating rate of zero), Two substances, potassium figure, the square of the ratio of the
i.e. quasi isothermally in the time dihydrogen phosphate (PDP) and temperature to the critical part of the
domain [1] and with small strontium titanate (SrTiO3), were specific heat capacity is shown as a
measurement signals; on top of this chosen as models to test the quality function of temperature[1,4]:
it allows studies of latent heat or of of the new measurement system in a (T/∆cp)2 proportional to (T-To)
12 USER COM June 98
It turns out that Tc and To are the
same (Tc ≈ To = 122.6 K) within the
limits of temperature accuracy. It
follows that the ferroelectric phase
transition of our PDP sample occurs
in the immediate vicinity of the
tricritical point.
The evaluation of a cp(T) curve in
the region of a phase transition is
made more difficult in that both the
crystal quality and also a poorly
chosen measurement scenario can
have a similar effect on the
measurement results. Figure 3 shows
the comparison of two time domain
TMDSC measurements of the same
quality on a quasi defect free PDP
Fig. 1: Adiabatic calorimetry (b) and time domain TMDSC (a) of PDP. For sample (curve a) and on a PDP
further explanations see text. sample with enriched surface defects
brought about by mechanical
polishing (curve b). It is evident that
the enrichment of defects leads to a
significant ‘smearing’ of the phase
transition. On the other hand one
recognizes the power of the TMDSC
method for the detection of such
defects.
In the case of phase transitions that
demonstrate only weak cp anomalies,
a slightly increased concentration of
defects caused by polishing can
make the detection and the
evaluation of a phase transition
appreciably more difficult. Figure 4
Fig. 2: This behavior of the critical part ∆cp of the specific heat capacity of PDP shows two TMDSC investigations in
for T<Tc = To = 122.6 K is known as ‘Landau-like’. the region of the phase transition of
SrTiO3 at Tc = 105 K. Curve (b) was
performed on a mechanically
polished sample. At best, only
indications of a phase transition in
the region of Tc can be made out.
Curve (a) was measured on the same
sample after removing the lattice
defects by etching strongly. The well
known weak second order phase
transition can now be clearly
identified.
The importance of the choice of a
suitable scenario for the
measurement of the behavior of the
specific heat capacity at the phase
transition and therefore for the phase
Fig. 3: Detection of the influence of crystal defects on the behavior of the specific transformation behavior is
heat capacity in the region of the ferroelectric phase transition of PDP (a = 0.02 K, demonstrated again in figure 5 for
p = 30 s). For further explanations see text. the defect-rich PDP sample. As
USER COM June 98 13
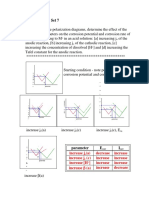
expected, both the position and the
shape of the cp(T) curve depend on
the heating rate, the modulation
frequency and the amplitude of
modulation. By all appearances, the
time domain TMDSC technique
supplies the most reliable cp data.
It can be concluded from these
results that the TMDSC technique is
very suitable, even at medium-low
temperatures, for the investigation of
phase transitions in the condensed
phase, as long as the cooling of the
measurement system is sufficiently
stable. At the same time, it is clear
that the time domain method is to be
preferred to measurements with rates
not equal to zero.
Literature:
[1] K. P. Bohn, A. Prahm,
J. Petersson, J. K. Krüger, Abb. 4: Detection of the influence of crystal defects on the behavior of the specific
Thermochimica Acta heat capacity in the region of the structural phase transition of SrTiO3 at Tc = 105
304/305 (1997) 283-290 K (r = 0.12 K/min, a = 0.2 K, p = 50 s).
[2] W. Reese, L. F. May, Phys. Rev.,
162, (1967) 510-518
[3] W.Gebhardt, U.Krey, „Phasen-
übergänge und kritische Phäno-
mene“, Vieweg & Sohn,
Braunschweig/Wiesbaden 1980
[4] E. K. H. Salje „Phase transitions
in ferroelastic and co-elastic
crystals“, Cambridge University
Press, Cambridge 1990
Fig. 5: Detection of the influence of the applied measurement scenario of
TMDSC measurements on the ‘apparent’ specific heat capacity. Polished (defect-
rich) PDP was used as model substance. Curve (a): r = 0.6 K/min, a = 0.2 K, p =
60 s; curve (b): r = 0.025 K/min, a = 0.1 K, p = 240 s; curve (c): time domains
TMDSC, a = 0.02, p = 30 s.
14 USER COM June 98
Safety investigations in glass
crucibles (100 µl)
Introduction Previously, safety investigations were often performed in glass crucibles. Glass has the
advantage of a noncatalytic surface. In addition, the sample can be inspected visually after
the measurement. Unfortunately the glass crucibles (ME 27 812) are too high for the
DSC821e cell. A furnace expansion with a special lid (known as the DSC furnace expander
ME 51 140 735) is now available, so that the 100 µl glass crucibles can be used once again.
Sample 4-nitrotoluene
Sample preparation Grind the 4-nitrotoluene in a mortar. Fill about 5 mg into the glass crucible. Clean the
narrow part of the crucible with a piece of rolled tissue paper. Seal the crucible with a
microburner. The 4-nitrotoluene remains cold thanks to the crucible stand.
Conditions Measuring cell DSC821e with air cooling and furnace expander
Temperature program Heating from 25 °C to 430 °C at 10 K/min
Interpretation The sharp endothermic peak indicates the melting of the sample. An initial endothermic
reaction occurs over a wide temperature range between 200 °C and 340 °C. Exothermic
decomposition begins at about 340 °C.
Summary One should be aware of the potential dangers when chemicals are stored and used. In
particular, it is important to determine any exothermic reactions. DSC safety investigations
detect such reactions and determine the maximum energy of the reaction, the heat of
reaction and the corresponding temperature range. These investigations have to be
performed in hermetically sealed and pressure resistant crucibles to avoid loss of volatile
materials.
USER COM June 98 15
Determination of curing behavior
using TMA/SDTA
During curing, a duroplast is at first sample and exerts a defined force on device with variable distance
liquid or can at least undergo plastic it. In dilatometric measurements this between the supports
deformation. With increasing force is so small that the expansion (ME 51140755).
crosslinking it becomes gel-like or of the sample is not hindered. On the The relationship for the bending
rubber-like before it finally hardens. other hand, with TMA softening system is given by the equation:
The molecules form a three-dimen- measurements, an appreciable force E = (∆F . l3) / (4 . ∆L . a3 . b) (2)
sional network throughout the entire is exerted on the sample. In DLTMA ∆F change of force of DLTMA
reaction mass. The gelation of normal measurements the force changes, l distance between the supports
epoxy resins occurs with a conversion (in the lengthwise direction of
of about 65 %. The gelation time is the sample)
important for the processing of ∆L flexure of the sample
duroplasts, because afterwards the (DLTMA signal)
material can no longer be plastically a sample thickness (vertical)
formed. An adhesive, for example, can b sample width (horizontal)
only be used up until just before the
gelation (the gelation time is also As with other modulation
Fig. 1: The diagram shows the
known as the ‘pot life’). techniques, a phase angle occurs
alternating rectangular force profile.
Thermal analysis includes several dif- between the exertion of the force and
This in turn leads to the periodic change
ferent techniques that can characterize the resulting signal. The phase angle
in length of the sample.
the curing behavior of such systems: is a measure of the damping
reaction kinetics (curing time and alternating between low and high behavior of the sample.
curing temperature) as well as the values.
measurement of certain physical The modulus of elasticity (Young’s SDTA
properties before, during and after the modulus) is the ratio of the A thermocouple measures the tempe-
curing. DSC is the technique mechanical stress (force per area) to rature at the sample position. The
normally used to follow the the relative change in length: SDTA signal is equal to the
crosslinking reaction by measuring E = (∆F/A) / (∆L/L0) (1) difference between this measured
the heat evolved, and then Only very soft samples such as sample temperature and the
afterwards, to measure the resulting rubber can be measured in this so- calculated reference temperature at
glass transition temperature. DSC called compression mode. In the sample position. It corresponds
can not, of course, determine the comparison, the bending to the temperature difference of a
increase of the viscosity or the arrangement makes the measurement measurement with and without a
modulus of elasticity during the of samples of even very high sample and is, with reproducible
curing, or the expansion coefficient modulus of elasticity possible . thermal resistance, proportional to
of the cured resin. F
the heat flow to the sample.
Thermomechanical analysis (TMA) a b
is then the method of choice, Application example
especially if bending measurements The curing of a two-component
are performed. Ideally both epoxy adhesive was measured with
techniques, i.e. the measurement of Fig. 2: The TMA bending measurement TMA/SDTA in order to demonstrate
the heat of reaction and of bendingThe diagram shows how the sample the possibilites described above.
are combined. This has has been is mounted on two supports. The According to the manufacturer’s
realized in the METTLER TOLEDO support is formed from wire of about information, the curing time was 24
TMA/SDTA840. 1 mm diameter. The probe with the hours at room temperature.
ball point presses in the middle on It is possible to put a drop of the
Bending measurements with the sample. The U-form wire has the freshly mixed adhesive on to the
DLTMA advantage that it need not be cleaned sample holder and let the probe
Normally with TMA, the dimensions after the curing reaction, because a move up and down. The gelation
of a sample are recorded as a new one is used for each time is reached as soon as the
function of time (or temperature). measurement. movement of the probe comes to rest
The probe is in contact with the There is also a quartz glass bending during the isothermal curing. A disad-
16 USER COM June 98
vantage is that, after the measurement,
the probe sticks to the sample holder
and the cured sample must be burned
off (heat cleaning).
It is also possible to measure the
curing of the sample in an aluminum
crucible (as with DSC) without
lowering the probe. This method
records only the SDTA and no
length change.
To measure the change of viscosity
and of Young’s modulus a thin film of
the adhesive (about 0.3 mm) was
applied to the underside of a razor
blade. This had been previously cut so
as to fit the U-form wire serving as the
bending device. Measures were taken
to prevent contact of the adhesive with
the sample holder. Without the
adhesive sample, the elastic behavior
of the razor blade steel can be
measured. With the adhesive layer the
Fig. 3: Curing of a an epoxy resin with TMA/SDTA in the bending mode. The
bending behavior is initially the same
underside of a razor blade suitably cut out is coated with a 0.3 mm layer of freshly
as without the sample. The flexure
mixed adhesive . Heating rate 5 K/min, load 0.02/0.04 N, period 12 s. width of
decreases during the curing process.
blade 3.4 mm, distance between the supports 10 mm. The DLTMA curve was
The DLTMA curve is shown in figure
corrected for the thermal expansion (which is not of interest here) by subtraction of
3. The SDTA measurement curve
the mean curve. SDTA curve: the second measurement was subtracted as ‘blank
yields an exothermic peak in spite of
curve’. The conversion curve results from the SDTA curve. The Young’s modulus
the gap between the adhesive film
was calculated using equation (2).
and the thermocouple in the sample
holder. Integration of the SDTA peak
results in the conversion curve
shown. The dotted line at 65 %
conversion corresponds to the
gelation of the adhesive. It also
intersects the Young’s modulus
curve at two thirds of its change.
The same setup allows the glass
transition temperature of the cured
adhesive to be determined in a second
heating run, since the Young’s
modulus changes to a considerably
lower value at the glass transition.
Figure 4 shows the DLTMA curves of
curing (first heating) and for
comparison the measurement of the
glass transition (second heating). The
corresponding values for the Young’s
modulus are recorded. The glass
transition occurs in the range 40 °C to
60 °C. The curve of the phase angle
between the applied force and the
resulting flexure is also shown. As
Fig. 4: DLTMA of the epoxy adhesive: First measurement (curing) as in Fig. 3. with ADSC, the phase lags noticeably
Second measurement to determine the glass transition of the cured resin was behind during the glass transition. All
performed under the same conditions as in Fig. 3. The values for Young’s modulus the possibilities described above are
are also calculated according to equation (2). The phase angle curve shows that the included in version 5.1 of the STARe
phase lags behind during the glass transition. Software TMA option ‘Evaluation’.
USER COM June 98 17
Tips: metals can also attack platinum performed in an electric oven. If
Platin crucibles in thermogravimetry just as phosphorus, arsenic, this is done by glowing over an
antimony, bismuth, silicon, boron open flame, then only the
Dr Rod Bottom, MT UK, Leicester of free carbon. We therefore oxidizing region should be used.
recommended that you put some Traces of metal oxides could
A potential customer asked us to alumina in the crucible to prevent otherwise be reduced and form an
measure the decomposition profile of these substances coming into direct alloy with the platinum (carbon
dishwashing powder (a mixture of contact with the platinum. black is also a platinum poison).
silicates and organic surfactants), in • Platinum is soft, can easily be • The crucibles are best cleaned by
order to check the suitability of our deformed and is susceptible to boiling in a suitable solvents
thermobalance for his application. scratches (possible centers of (water or acids). A very good
The initial TGA measurements were corrosion). Pay special attention to method is the use of fused
performed under nitrogen in 70 µl a perfectly clean and flat surface in potassium pyrosulfate to dissolve
alumina crucibles in the tempearture order to guarantee the best possible contaminants followed by boiling
range 40 °C to 600 °C at a heating thermal contact. in water. Mechanical cleaning
rate of 10 K/min. The result, a should be done with fine cleaned
weight loss step between 200 °C and • New platinum materials tend to sea sand, so that the surface is not
300 °C as well as a significant stick to each other on contact when damaged. Under no circumstances
residue, was presented to the heated to above 1000 °C (platinum should they be glowed to constant
customer. His comments were that crucible with lid or sample holder). weight before cleaning because
our system did not show the results This effect disappears to a large impurities would then only
be expected and that it was not as extent on repeated use (heating). penetrate further into the metal.
sensitive as other competitive • Heating out platinum is best
instruments. Spurred on by this
reaction, we performed further
measurements at reduced heating
rates, using smaller sample sizes and
lighter aluminum crucibles. Without
success. Finally our experts in
Schwerzenbach gave us the decisive
tip - to test for the possible catalytic
effect of platinum crucibles. A new
series of experiments was started
that infact yielded the expected
results. The catalytic effect of platin-
um led to the decomposition being
completed 30 °C lower and made
three decomposition steps visible.
Today this customer has a Mettler-
Toledo instrument...
A part from possible catalytic
effects, platinum exhibits several
other properties that can in practice
lead to complications.
• ‘Platinum poison’: platinum can
under certain circumstances react
with a number of substances. The- The example shows that the material used for the crucible can have an effect on
se include molten metals which the decomposition reaction and therefore influence the measurement curve. The
lead to the formation of alloys. difference is warefor not due to faulty adjustment of the instrument but is caused
Examples of such metals are tin, by the catalytic effect of platinum on certain reactions. Whether this effect has an
lead, zinc, aluminum, silver and influence on your reactions must be checked from case to case. Actually inert
gold. Salts or oxides of heavy crucibles are preferred for thermal analysis.
18 USER COM June 98
Exhibitions, conferences and courses
Analitica June 30 - July 2, 1998 Sao Paulo (Brasilien)
15th IUPAC Conference on
Chemical Thermodynamics July 26 - August 8, 1998 Porto (Portugal)
ESTAC August 8 - September 4, 1998 Balatonfüred (Hungary)
Euroanalysis 10 September 6 - 11, 1998 Basle (Switzerland)
CTAS September 9 - 12, 1998 Kingston, ONT (Canada)
NATAS September 13 - 15, 1998 Cleveland (USA)
Salon de la Physique September 16 - 19, 1998 Paris (France)
Het Instrument October 4 - 9, 1998 Jaarbeurs Utrecht (Netherlands)
lV Simposio Latino Americano
de Polimeros October 25 - 28, 1998 Viña del mar (Chile)
K October 22 - 29, 1998 Düsseldorf (Germany)
Eurostar PhandTA 4 March 24 - 26, 1999 Karlsruhe (Germany)
13. Ulm-Freiberger Kalorimetrietage March 17 - 19, 1999 Freiberg (Germany)
15th European Conference on
Thermophysical Properties September 5 - 9, 1999 Würzburg (Germany)
ICTAC 2000 August 14 - 18, 2000 Copenhagen (Denmark)
TA customer courses and seminars (Switzerland)
Information and registration: Helga Judex
Tel.: ++41 806 72 65, Fax: ++41 806 72 40, E-Mail: .helga.judex@mt.com
ADSC Seminar November 10, 1998 Greifensee (Switzerland)
Kalibrier- und Validier Seminar November 11, 1998 Greifensee (Switzerland)
Kinetik Seminar November 12, 1998 Greifensee (Switzerland)
TMA (German) November 17, 1998 Greifensee (Switzerland)
TGA (German) November 18, 1998 Greifensee (Switzerland)
DSC Basic (German) November 19, 1998 Greifensee (Switzerland)
DSC Advanced (German) November 20, 1998 Greifensee (Switzerland)
TMA (English) November 23, 1998 Greifensee (Switzerland)
TGA (English) November 24, 1998 Greifensee (Switzerland)
DSC Basic (English) November 25, 1998 Greifensee (Switzerland)
DSC Advanced (English) November 26, 1998 Greifensee (Switzerland)
TA customer courses and seminars (Region Central)
For further information please contact Mettler-Toledo Giessen, Germany
Tel. +49 641 507 431 (Bianca Bochem) or +49 641 507 404 (Ina Wolf)
DSC Workshop October 15 - 16, 1998 Giessen (Germany)
TG Workshop November 4 - 5, 1998 Giessen (Germany)
TA-Seminar October 14, 1998 Croatia/Slovenia
TA-Seminar October 16, 1998 Czech Republic
TA customer courses and seminars (Netherlands)
For further information please contact Mettler-Toledo Tiel
Tel. 0344-63 83 63 (Hay Berden)
Info Day Juni 18, 1998 Arnhem/Oosterbeek Hotel
West-End
TA customer courses and seminars (Italy)
For further information please contact Mettler-Toledo Italy
Tel. (02) 356 29 73
TA Seminar September 22 - 23, 1998 Novate Milanese
USER COM June 98 19
TA customer courses and seminars (USA/CANADA)
Please contact your local instrument specialist or Jon Foreman
Tel.: 1 800 METTLER (638-8537) extension 4687 or Fax 1 614 438-4958
Info Days October/November 1998
TA customer courses and seminars (UK)
September 1998. Short seminar series “Quality management and system certification in the pharmaceutical
industry”. Venues to be arranged, Customised on-site training courses
Contact Rod Bottom for more information Tel. 0116 234 50 25, Fax 0116 234 50 25
TA customer courses and seminars (Belgium)
STARe User forum 21.10.98 Lot
Contact Pat Hoogeras or Philippe Larbanois for more information Tel. 02 334 02 11,
Fax 02 334 02 80
If you have any other questions concerning other meetings, products or application please contact your local
METTLER TOLEDO dealer.
Internet: http:/www.mt.com
Editorial office
Mettler-Toledo GmbH, Analytical
Sonnenbergstrasse 74
CH-8603 Schwerzenbach, Switzerland
E-Mail: urs.joerimann@mt.com
Tel.: ++41 1 806 73 87, Fax: ++41 1 806 72 60
Dr. J. de Buhr, Dr. R. Riesen, J. Widmann, U. Jörimann
Layout and Production
Layout and Production: Promotion & Documentation Schwerzenbach G. Unterwegner
ME-51709835
You might also like
- Phase Equilibrium in Mixtures: International Series of Monographs in Chemical EngineeringFrom EverandPhase Equilibrium in Mixtures: International Series of Monographs in Chemical EngineeringNo ratings yet
- Measuring Specific Heat CapacityDocument5 pagesMeasuring Specific Heat CapacityhernanNo ratings yet
- USERCom TA 2Document8 pagesUSERCom TA 2rubenssanNo ratings yet
- User Com: Information for METTLER TOLEDO thermal analysis systems usersDocument8 pagesUser Com: Information for METTLER TOLEDO thermal analysis systems usersJosé FigueroaNo ratings yet
- DSCDocument37 pagesDSCshailesh2006_1982100% (3)
- Minimum Number of SamplesDocument5 pagesMinimum Number of SamplesEdwin KohNo ratings yet
- ASTM C1702Document9 pagesASTM C1702adolfo camayoNo ratings yet
- UserCom 11Document28 pagesUserCom 11maestramariNo ratings yet
- Thermal Behaviour of Materials in Interrupted Phase ChangeDocument10 pagesThermal Behaviour of Materials in Interrupted Phase ChangeConstance HoshijimaNo ratings yet
- Interpretation of DSC Curves in Polymer Analysis 2000 - ToledoDocument0 pagesInterpretation of DSC Curves in Polymer Analysis 2000 - ToledoyrecoverNo ratings yet
- UserCom TA 4Document20 pagesUserCom TA 4rubenssanNo ratings yet
- ASTM D5865-2010 - 1250-Coal GCV SopDocument3 pagesASTM D5865-2010 - 1250-Coal GCV SopGirijaNo ratings yet
- Specific Heat Is Another Physical Property of MatterDocument8 pagesSpecific Heat Is Another Physical Property of MatterMarie-Joi PauletteNo ratings yet
- Silicon Temperature Sensing With PrecisionDocument3 pagesSilicon Temperature Sensing With Precisionmesbah mNo ratings yet
- Ancsin J Mendez Lango FP Reproducibility & PRTDocument24 pagesAncsin J Mendez Lango FP Reproducibility & PRTemendez56No ratings yet
- Lecture 5Document11 pagesLecture 5Rahmeh EL saaiedehNo ratings yet
- Measurement of Heat of Hydration of Hydraulic Cementitious Materials Using Isothermal Conduction CalorimetryDocument9 pagesMeasurement of Heat of Hydration of Hydraulic Cementitious Materials Using Isothermal Conduction Calorimetrycarlos salinasNo ratings yet
- Temperature Calibration Depths of ImmersionDocument7 pagesTemperature Calibration Depths of ImmersionDiego Bragga TerrazyNo ratings yet
- Standard Test Method For Measurement of Heat of Hydration of Hydraulic CementitiousDocument9 pagesStandard Test Method For Measurement of Heat of Hydration of Hydraulic CementitiousQuoc Tri PhungNo ratings yet
- Specific Heat of Liquids and Solids: Standard Test Method ForDocument6 pagesSpecific Heat of Liquids and Solids: Standard Test Method ForJean-Patrice DeliaNo ratings yet
- ASTM C1702-2009a - 2500Document3 pagesASTM C1702-2009a - 2500L roachle100% (1)
- Impact of GC Parameters On The Separation - 5 Column TemperatureDocument6 pagesImpact of GC Parameters On The Separation - 5 Column TemperatureastroguilleNo ratings yet
- Temperature Calibration Depths of ImmersionDocument7 pagesTemperature Calibration Depths of ImmersionNicole FloydNo ratings yet
- Javed AssignmentDocument26 pagesJaved AssignmentMustafaNo ratings yet
- DSC Analysis Explained in 40 CharactersDocument38 pagesDSC Analysis Explained in 40 CharactersSukhwant SinghNo ratings yet
- T.Y.B.Sc. 4.1.3 DTA Analysis PPT TYDocument32 pagesT.Y.B.Sc. 4.1.3 DTA Analysis PPT TYmohamed arifNo ratings yet
- Heat of Hydration - ASTM C1702Document8 pagesHeat of Hydration - ASTM C1702WayfrelNo ratings yet
- Ancsin J Mendez Lango NRC CENAM Comparison Metrologia 1996Document7 pagesAncsin J Mendez Lango NRC CENAM Comparison Metrologia 1996emendez56No ratings yet
- Calorimetry: Physikalisch-Chemisches Praktikum I Calorimetry - 2016Document19 pagesCalorimetry: Physikalisch-Chemisches Praktikum I Calorimetry - 2016Agu15kelvinNo ratings yet
- E 457 - 08 (2015)Document6 pagesE 457 - 08 (2015)ruben carcamoNo ratings yet
- EE431 Lab 3: Measuring The Temperature Coefficient of A ResistorDocument3 pagesEE431 Lab 3: Measuring The Temperature Coefficient of A Resistorbilalhaider55No ratings yet
- ASTM E711 - 87 Gross Calorific CalorimeterDocument8 pagesASTM E711 - 87 Gross Calorific Calorimeteremannuelly100% (2)
- The Contactor: Temperature Measurement Discrepancies in The Claus Thermal ReactorDocument2 pagesThe Contactor: Temperature Measurement Discrepancies in The Claus Thermal ReactorMujeeb AhmadNo ratings yet
- Enthalpy of Combustion via Calorimetry ExperimentDocument8 pagesEnthalpy of Combustion via Calorimetry ExperimentJohnPholTalabanNo ratings yet
- Exercicios de TemperaturaDocument9 pagesExercicios de TemperaturaMirellaNo ratings yet
- Calibration of Calorimeters and Thermal Analyzers.: S3 Project Raffort Théo, Bouvier Téo, Starosta Yvann, MCPC ADocument8 pagesCalibration of Calorimeters and Thermal Analyzers.: S3 Project Raffort Théo, Bouvier Téo, Starosta Yvann, MCPC AstarostaNo ratings yet
- DSC WikanDocument23 pagesDSC WikanAhmad Wildan ZakawaliNo ratings yet
- D5865 GCVDocument14 pagesD5865 GCVArunkumar ChandaranNo ratings yet
- Hot Spot Temperature Analysis of Transformer Using FEM On COMSOLDocument7 pagesHot Spot Temperature Analysis of Transformer Using FEM On COMSOLEditor IJTSRDNo ratings yet
- Nara1991 Magneto-Resistance of A Highly Stable Industrial-Grade Platinum Resistance Thermometer Between 20 and 240 K.Document5 pagesNara1991 Magneto-Resistance of A Highly Stable Industrial-Grade Platinum Resistance Thermometer Between 20 and 240 K.Alexandre HenriquesNo ratings yet
- Mini Project ReportDocument14 pagesMini Project ReportfatinNo ratings yet
- Temperature MeasurementDocument57 pagesTemperature MeasurementATUL ASWARNo ratings yet
- Study of Temperature Sensors: Pt100Document19 pagesStudy of Temperature Sensors: Pt100dark*nightNo ratings yet
- Self Calibrating Infrared Thermometer FoDocument6 pagesSelf Calibrating Infrared Thermometer Fomarcelo.lacknerNo ratings yet
- IJREI - Analysis of Calorimetric Measurement Bismuth and Tin SystemDocument5 pagesIJREI - Analysis of Calorimetric Measurement Bismuth and Tin SystemIjrei JournalNo ratings yet
- QI Heater Tube Cleaning and VerificationDocument10 pagesQI Heater Tube Cleaning and VerificationOmid LarkiNo ratings yet
- Differential Scanning Calorimetry: Presented To: Dr. Mohsin Ali RazaDocument23 pagesDifferential Scanning Calorimetry: Presented To: Dr. Mohsin Ali RazaBesma HamdiNo ratings yet
- Applying ISO Guide To Uncertainty of Temperature MeasurementDocument28 pagesApplying ISO Guide To Uncertainty of Temperature Measurementmross0355100% (1)
- D7896 14Document6 pagesD7896 14quoctrangbk02No ratings yet
- Module - 1Document229 pagesModule - 1Sourav PoddarNo ratings yet
- High Temperature NMR GuideDocument5 pagesHigh Temperature NMR GuideAurelia BucurNo ratings yet
- Assignment 1 & 2Document17 pagesAssignment 1 & 2Iyyan Paramanandam67% (3)
- Differential Scanning CalorimetryDocument7 pagesDifferential Scanning CalorimetryGintoki SakataNo ratings yet
- Astm D 2015 - 00Document9 pagesAstm D 2015 - 00alexander guerraNo ratings yet
- Chapter 1. Thermal Analysis: Dr. Nguyen Tuan LoiDocument36 pagesChapter 1. Thermal Analysis: Dr. Nguyen Tuan LoiNhung Đặng100% (1)
- TemperatureDocument8 pagesTemperaturetawananyashahokNo ratings yet
- Thermo Gravimetricanalysistga 190602083546Document33 pagesThermo Gravimetricanalysistga 190602083546Kevin Gilberto S�nchez Ch�vezNo ratings yet
- CALO-3_SPECDocument1 pageCALO-3_SPECjmatallanaNo ratings yet
- Astm D 5468 - 02Document8 pagesAstm D 5468 - 02nerissaloveNo ratings yet
- UserCom TA 9Document24 pagesUserCom TA 9rubenssanNo ratings yet
- UserCom TA 4Document20 pagesUserCom TA 4rubenssanNo ratings yet
- User Com: Investigating Unknown SamplesDocument12 pagesUser Com: Investigating Unknown SamplesrubenssanNo ratings yet
- UserCom TA 5Document16 pagesUserCom TA 5rubenssanNo ratings yet
- UserCom TA 6Document24 pagesUserCom TA 6rubenssanNo ratings yet
- UserCom TA 4Document12 pagesUserCom TA 4rubenssanNo ratings yet
- A Method To Improve The Characteristics of EPDM Rubber Based Eco-Composites With Electron BeamDocument18 pagesA Method To Improve The Characteristics of EPDM Rubber Based Eco-Composites With Electron BeamrubenssanNo ratings yet
- FT IRAnalysisofMaterialsDocument50 pagesFT IRAnalysisofMaterialsrubenssanNo ratings yet
- Press Kit Apollo 11Document256 pagesPress Kit Apollo 11rubenssanNo ratings yet
- IBM SPSS GPL Reference Guide PDFDocument363 pagesIBM SPSS GPL Reference Guide PDFrubenssanNo ratings yet
- Interpreting TGA CurvesDocument20 pagesInterpreting TGA CurvesIoana MoldovanNo ratings yet
- Lifetime prediction for polymer materials using OIT measurements by DSC methodDocument3 pagesLifetime prediction for polymer materials using OIT measurements by DSC methodrubenssanNo ratings yet
- Interlaboratory Comparison of Hydroxyapatite Crystallinity Ratio Measurement According To ISO 13779-3Document10 pagesInterlaboratory Comparison of Hydroxyapatite Crystallinity Ratio Measurement According To ISO 13779-3rubenssanNo ratings yet
- pvmrw13 ps5 Qlab Fowler PDFDocument12 pagespvmrw13 ps5 Qlab Fowler PDFrubenssanNo ratings yet
- C-Therm TCi Operator ManualDocument138 pagesC-Therm TCi Operator ManualrubenssanNo ratings yet
- Methods For Estimating Intrinsic ViscositDocument4 pagesMethods For Estimating Intrinsic ViscositrubenssanNo ratings yet
- Good Weighing Practice in Pharmaceutical IndustryDocument57 pagesGood Weighing Practice in Pharmaceutical IndustrySathish Vemula100% (2)
- Physical Characterization: Surface Area and Porosity: WhitepaperDocument7 pagesPhysical Characterization: Surface Area and Porosity: WhitepaperrubenssanNo ratings yet
- Quick Guide Gplv3 CroppedDocument6 pagesQuick Guide Gplv3 CroppedLobo LopezNo ratings yet
- PVC Fact Book CompleteDocument101 pagesPVC Fact Book CompleterubenssanNo ratings yet
- Sop Tga PeresinDocument5 pagesSop Tga PeresinrubenssanNo ratings yet
- Aging Engines, Avionics, Subsystems and HelicoptersDocument122 pagesAging Engines, Avionics, Subsystems and HelicoptersrubenssanNo ratings yet
- Elastic Plastic MaterialsDocument49 pagesElastic Plastic MaterialsRejita PillaiNo ratings yet
- Complying With ISO 17025 A Practical GuidebookDocument122 pagesComplying With ISO 17025 A Practical Guidebookyes17025100% (8)
- PlasticsDocument56 pagesPlasticsSeow FhanNo ratings yet
- Using The Grubbs and Cochran Tests To Identify OutliersDocument3 pagesUsing The Grubbs and Cochran Tests To Identify OutliersrubenssanNo ratings yet
- Review of Accelerated Ageing Methods and Lifetime Prediction Techniques For Polymeric Materials - Maxwell, Dean Et Al. NPL ReportDocument84 pagesReview of Accelerated Ageing Methods and Lifetime Prediction Techniques For Polymeric Materials - Maxwell, Dean Et Al. NPL ReportDavide DuranteNo ratings yet
- The New ISO 527Document25 pagesThe New ISO 527rubenssan100% (1)
- EPM Assignment 1Document3 pagesEPM Assignment 1arsalan amjadNo ratings yet
- Media OptimisationDocument90 pagesMedia OptimisationMohanan C VarshaNo ratings yet
- Energetic PolymersDocument203 pagesEnergetic Polymerslonelystar19No ratings yet
- Extraction and Uses of Metals: Section 2: Inorganic ChemistryDocument8 pagesExtraction and Uses of Metals: Section 2: Inorganic Chemistryjenn78No ratings yet
- E-159 and E-199-BIOBASE - 25 Freezer BDF-25V368 User Manual 202108Document15 pagesE-159 and E-199-BIOBASE - 25 Freezer BDF-25V368 User Manual 202108Vanessa Margaret Palomino RodriguezNo ratings yet
- Cast Iron Pumps - PGP365 Series - Parker NADocument5 pagesCast Iron Pumps - PGP365 Series - Parker NAgonzalo andres HernandezNo ratings yet
- Rubric Assessment For Laboratory Report: Performance of Water PumpDocument4 pagesRubric Assessment For Laboratory Report: Performance of Water PumpPaulo Angelo LimNo ratings yet
- Material Science 19Document3 pagesMaterial Science 19MAENYA BRUCE OYONDINo ratings yet
- Complete and Simple Solutions For Industrial Process Water and Wastewater TreatmentDocument3 pagesComplete and Simple Solutions For Industrial Process Water and Wastewater TreatmentElena GilNo ratings yet
- Chemistry of The Viscose Process 1Document12 pagesChemistry of The Viscose Process 1biswasblrNo ratings yet
- Ex 8Document3 pagesEx 8Ters MedinaNo ratings yet
- Multiphase ReactorDocument37 pagesMultiphase ReactorMaria Charlene Caraos TapiaNo ratings yet
- Procedures and Observations For Chemical and Physical Changes LabDocument6 pagesProcedures and Observations For Chemical and Physical Changes LabkmwawersNo ratings yet
- Hydrotropic SolubilizationDocument7 pagesHydrotropic SolubilizationDavidGutiérrezNo ratings yet
- Pavement Materials NotesDocument105 pagesPavement Materials NotesProf. Nagarjun Gowda B SNo ratings yet
- Ofet PDFDocument16 pagesOfet PDFFaisal BashirNo ratings yet
- Physics CurrentDocument14 pagesPhysics Currentkumarardash86No ratings yet
- Soluition Charpter 3 - 2 PDFDocument3 pagesSoluition Charpter 3 - 2 PDFBryan de BarrosNo ratings yet
- Standardizing NaOH and HCL SolutionsDocument4 pagesStandardizing NaOH and HCL SolutionsfauziasuedNo ratings yet
- Research PaperDocument5 pagesResearch PaperUttam JhaNo ratings yet
- ChemiluminescenceDocument18 pagesChemiluminescenceza chohan100% (1)
- Astronomy Quiz 1Document6 pagesAstronomy Quiz 1Neil GhoshNo ratings yet
- Tesla 369 theory pdf keyDocument3 pagesTesla 369 theory pdf keyM Yudi SuhendarNo ratings yet
- S1 G2 Mechanically Alloyed MetalsDocument15 pagesS1 G2 Mechanically Alloyed MetalsUmer MuzammilNo ratings yet
- Introduction To Analytical ChemistryDocument4 pagesIntroduction To Analytical ChemistryCHRISTINE JOY RETARDONo ratings yet
- High Temperature Superconducting Cable US6794579Document10 pagesHigh Temperature Superconducting Cable US6794579Leon MutambalaNo ratings yet
- Light Curing DevicesDocument13 pagesLight Curing DevicesAhmed Elhossany100% (1)
- Exlon Hdpe CatalogDocument32 pagesExlon Hdpe Catalogcarlos burgosNo ratings yet
- Water Activity PredictionDocument48 pagesWater Activity PredictionercanefeogluNo ratings yet
- Design of Grit Chamber: Civil Engineering Department S. V. National Institute of Technology, SuratDocument10 pagesDesign of Grit Chamber: Civil Engineering Department S. V. National Institute of Technology, SuratRAVATNo ratings yet