Professional Documents
Culture Documents
Mode of Action Classification: The Key To Insecticide Resistance Management
Uploaded by
Jesus Amilkar Cogollo LizarazoOriginal Description:
Original Title
Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
Mode of Action Classification: The Key To Insecticide Resistance Management
Uploaded by
Jesus Amilkar Cogollo LizarazoCopyright:
Available Formats
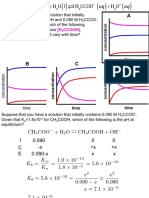
Group 1: Acetylcholinesterase inhibitors (Only major representatives of the groups are shown) Group 16: Inhibitors of chitin Group
Group 16: Inhibitors of chitin Group 17: Moulting disruptor, Dipteran
biosynthesis, type 1, Homopteran
CH 3
CH3 CH(CH3) 2
OCO N S N[(CH 2) 3CH 3] 2 SCH3
OCO N S NCH2CH2 CO2 CH2CH3
O CH3 CH3 NHCO 2 N C N N
C O N (C H 3 )2
O CH3 (C H 3) 3 C
CH3 N S C H 2 C O 2 C H 2 C H 3
CH3 O S H2 N N NH
CH3 CH 3
OCONHCH3 N N C(C H 3 )3
CH 3 NH C O CH3NCO2N C N N
CH 3 N
O CH3 SCH3 Triazamate
CH 3 S C CH N OC ONHCH 3 Benfuracarb Carbosulfan Methomyl S O CH(CH 3 )2 NH 2
CH3 SCH3
CH 3 CH 3 CH 3
Mode of Action Classification
CH3NCO2N C
SCH 3 CH 3 Buprofezin Cyromazine
OCONHCH3 O
CH 3NHC O CH 3
Aldicarb Carbofuran Methiocarb Thiodicarb
CHCH 2 CH3
(CH 3)2 NCOC NOCONHCH 3
SCH 3
The key to Insecticide Resistance Management
Carbaryl Fenobucarb Oxamyl
1A Carbamates Group 18: Ecdysone agonists moulting disruptors
Group 8: Unknown or non specific - fumigants O O O C
O
CO 2 CH 3
C(CH3 ) 3 CH 3 O CH 3
S
O Cl C(CH 3) 3 C(CH 3 ) 3 CH C(C H 3 ) 3 C H O O OH O
3 C H3C H2 3 CH3 CH3
S HN N
S HN N NH N HN N HO
Cl N OP(OCH2 CH3 )2 CH 3
CH 3 O CH 3
O CH 3O
O
(CH3 O) 2PS CHCH2CO 2CH2 CH3 O2N OP(OCH3 )2 O O CH 3 C O OH
O O
O CH 3
CH 3 CH 3 CH3 OC
H
O
Cl Cl O
CO2 CH2 CH3
F S F
O CH 3
S (CH3 O) 2P
O
O H Parathion-methyl
CH3 Br Cl3 C NO 2 Chromafenozide Halofenozide Methoxyfenozide Tebufenozide 18A Diacyl- Azadirachtin
Chlorpyrifos Malathion S O
CH3S P NHCOCH 3 N OP(OCH2CH 3)2 C C
( CH3)3 CSCH2 S P(OCH2CH3) 2 hydrazines 18B
N CH3 CONHCH3
Methyl Sulfuryl
OCH3 (CH3) 2CH
O (E )
Chloropicrin
O O bromide 8A Alkyl halides 8B Chloropicrin fluoride 8C Sulfuryl fluoride
S CH3O PSCH 3 P OCH 2C H 3
Acephate Diazinon Monocrotophos Terbufos
Cl SCH 2 CH 2 CH 3
CH3 NHCOCH2 SP(OCH3) 2 NH 2 Br
Group 19: Octopaminergic Group 20: Coupling site II electron
agonists transport inhibitors (Complex III)
Dimethoate Methamidophos Profenofos
1B Organophosphates Group 9: Unknown or Non specific – selective feeding blockers
N O
O O
CH CF 3 COCH 3 CF3
CH CF 3 CH3
CH 3
C H3 CH
CH 3
O C
CH
CH 3
NH (CH 2 ) 11 CH 3 O N CH 3
F
Group 2: GABA-gated chloride channel antagonists 3N a
F
F
Al
F
F
CH 3
N
N
CONHCH 2 CN
CH 3
CH3
N CH N CH N CH 3
CH 3 NH
N N C
CH
O
CH 2
O N O
CH
CH 3
CH CF 3
F N N
N O
CH 3 CH 2 SO CN H
Amitraz Hydramethylnon Acequinocyl Fluacrypyrim
Fluacrypyrim
Cl Cl
Cl Cl Cl Cl
Cryolite
9A Cryolite Pymetrozine Flonicamid 20A 20B 20C
Cl
Cl
Cl
Cl
Cl
O
SO Cl Cl
H 2N N
N
F 3C N
N CN
9B Pymetrozine 9C Flonicamid
Cl Cl Cl Cl O
Cl Cl O S
Cl Cl Cl NH 2
Cl Cl CF 3
CF 3
gamma-HCH
Chlordane Endosulfan (Lindane) Ethiprole Fipronil
Group 10: Unknown or Non specific – Mite Growth Inhibitors Group 21: Coupling site I electron transport inhibitors
2A Cyclodiene Organochlorines 2B Phenylpyrazoles (Fiproles) CH 3
Cl
S
O
O F
9C: Flonicamid
O C H 2 CH 3
C(CH 3)3
N CH2 CH 2
CH 3 CH 2
Cl
C
O
H
O O
C CH 2
N N Cl
N
N NH O CH3 N N NH CH 2 O
N C N CH 3
NH C(CH 3 )3 N CH 3 C(CH 3)3
CH 3CH 2 Cl
Group 3: Sodium channel modulators (Only major representatives of the groups are shown)
Cl
N N CH3
F
O
O
CH 2
CH 2 (CH2 )2OCH 2 CH 3
C H 3O
OCH 3
H
O
CH 3 N CH 3
N
O H
CH 3
CH 2 C
H Clofentezine 10A Clofentezine Hexythiazox 10A Hexythiazox Etoxazole 10B Etoxazole Fenazaquin H C O Pyrimidifen N
N
C(CH 3 )3
Tebufenpyrad CH 3 CH 2 Cl
O
O
Rotenone
CH 3 H C O C H N N C
CN O R1 O O (CH 3) 3C CH2 S O N CH 3
O O
C CH H NH CH 2
Cl CH3 CN C C O
R CH 3 CH 3 CH 2 C Cl CH 3
C CH CO 2CH O Cl H H O C(CH 3 ) 3
Cl
CH3
CH(CH 3) 2
F3C
(S) (Z)-(1R)-cis -
R = -CH 3 (chrysanthemates) or - CO 2CH 3 ( pyrethr ates)
R 1 = - CH=CH 2 ( pyr ethr in) or - CH 3 ( cinerin) or -CH 2CH 3 ( jasmol in)
Fenpyroximate Pyridaben Tolfenpyrad
21 METI acaricides
& Rotenone
(Z)-(1R)-cis- C CH CH 3 C O CN
F3C CH 3 2
Cl C O
C CH C H3 CO 2C H 2 H H H
Pyrethrins
Cl H
C H3
H
Cypermethrin Cl
C CH
CH3
CO 2CH
CN
O Esfenvalerate
Cl
H
CH 3
CH 3 H
(Pyrethrum)
Group 11: Microbial disruptors of insect mid-gut membranes - includes transgenic crops
CN
expressing Bacillus thuringiensis toxins. B.t. subspecies shown
H C H3 H Cl CH3 C CH CO 2
Cl CH 3 CH 3 C O
F3C
C CH CO 2C H 2 H
C H3 (S) (1R)-cis -
F3C Cl (R) (Z)-(1S)-cis -
C CH CH 3 C O
CN CH 3 O CH OCH 3
Cl
H
2
C O Zeta-
(Z )-(1S)-cis - H
CH 3CH 2 O
Lambda-
Group 23: Inhibitors of
H CCl 3
Bifenthrin
Cl
H
CH 3
CH 3 H
cypermethrin
O
cyhalothrin Group 22: Voltage dependent
lipid synthesis
C CH C O2 CN
Cl CH 3
H
(R) (1S)-cis -
C O
CH 3 C
CH 3
CH 2 OCH 2
F3C
(Z)-(1S)-cis-
F F Methoxychlor sodium channel blockers
Alpha- Cl
C CH CH 3 CO CH
H
2 2
H
CH 3
B.t. israelensis Bacillus.sphaericus B.t. aizawai B.t. kurstaki B.t. tenebrionis
Br F F
CN
Cl
C CH
CH 3
CO 2CH O
cypermethrin Br
C CH CH 3 CO 2
C
CN
O
Etofenprox CH 3
Cl O
Cl
H H H H CH 3
H F F Cl CH Cl
CH 3 CH 3 Cl
O
F
F 3C
C CH
CH 3
CO 2CH 2 CH 3
CCl3
11B2
Cl CO 2 CH 3 Cl O 23 Tetronic
(Z)-(1R)-cis-
F F
11A1 11A2 11B1 11C O O
O
CH 3 CH 3 C
O
C(CH 3 ) 3
CH 2
Cyfluthrin Deltamethrin Tefluthrin DDT N N
OCF 3
CH 3 C
acid
3 Pyrethroids O
N
CO 2 CH 3
CH 3 CH CH
2 3
CH 3
O
O
derivatives
Indoxacarb Spirodiclofen Spiromesifen
Group 12: Inhibitors of oxidative phosphorylation, disruptors of ATP formation (inhibitors
Group 4: Nicotinic acetylcholine receptor agonists/antagonists of ATP synthase)
CH 3
CH 3
C N
CN
CH 3 NH NO 2 N
N NO 2
C(CH 3)3
Group 24: Mitochondrial complex IV Group 25: Neuronal inhibitors
H H C C O
CH 2 N
CH 3
O N N
CH 3
CH2 N H
CH2 CH3
N
CH 2
C H(CH 3) 2
Sn CH 3 CH 3
Cl
electron transport inhibitors (unknown mode of action)
S O
O NH CSNH C(CH 3 )3 N C C H2 Sn O Sn CH 2 C Cl SO2 Cl
N N N CN N CH 3 CH 3 OSO2 CH 2 C CH
NO 2 S Cl
Cl NO 2 Cl N C H(CH 3) 2 N 3 3
H NO 2 N Cl
N N Fenbutatin
CH 3 N Cl N Thiamethoxam
Acetamiprid S Dinotefuran N Nitenpyram CH 2 Diafenthiuron Azocyclotin oxide Propargite Tetradifon
NH Cl CH 2 N N H Sn N HNH C OOC H(C H 3 )2
N
CH 2 OH PH 3 CN- PH 3 OCH 3
N
Cl
12B Organotin 12C 12C
Clothianidin Imidacloprid Thiacloprid
4A Neonicotinoids 12A Diafenthiuron Cyhexatin
miticides Propargite Tetradifon Phosphides
24A Cyanide
24B Phosphine
24C Bifenazate
N CH 3
SO 2 S CH 2
H 2 NCOS CH2 S CH 3
CH 2 SSO 3Na Group 13: Uncouplers of oxidative phos- Group 28: Ryanodine
N
CH N(CH 3 )2 CH N(CH 3 ) 2 .HCl S
CH 3
N CH
Group 14: Unallocated Group 26:Aconitase Group 27: Synergists
H SO 2 S CH 2 H 2 NCOS CH2 (CH 3)2N
S CH 2 SSO 3Na phorylation via disruption of H proton gradient receptor modulator
Cartap
inhibitors
Thiosultap-
Nicotine Bensultap hydrochloride Thiocyclam sodium
OH
O O
CN CH 3
4C Bensultap 4C Cartap
S
4B Nicotine 4C Nereistoxin analogues Br
Cl
O2N
o O
I HN
O
S (CH 2 )3 CH 3 O
CF 3 N F CH 3(CH 2)3 OCH 2CH 2OCH 2CH 2OCH 2 P
CH2 OCH 2CH 3 NO 2 O
CH 3 (C H 2) 3S HN
S(CH 2 )3 CH 3
o- CH 3(CH 2)2 O
C F3
Group 5: Nicotinic acetylcholine
F
Chlorfenapyr DNOC CF3
Piperonyl
Group 6: Chloride channel activators Fluoroacetate butoxide DEF Flubendiamide
receptor agonists (not 4)
Spinosad
27A P450-dependent 27B Esterase
HO
CH 3
OCH 3
O O
OCH 3
CH 3 CH 3 O
CH 3 O
O
CH 3
O
O
CH 3
O
H
R
O
CH 3
CH 3
C H3 H
O
O R
CH3
Group 15: Inhibitors of chitin biosynthesis, type 0, Lepidopteran monooxygenase inhibitors inhibitors
CH 3
( CH 3)2 N O O O
H CH 3
NH2
OCH 3 H
OCH 3 CH 3 O CH 3 CH 3
CH 3 O O
CH 3
O O
C H3
CH 3 O CH 3 O
H
CO 2
O O
6
O CH 3 HO H
H R
HH
CH 3
OH H M ilbem ycin A3: R = -CH 3
O O O O
O OH H O CH 3
M ilbem ycin A4: R = -CH 2CH3
(i) R = -CH 2 CH 3 (avermectin B1 a) H
CH 3CH 2 O B1a R = CH 3CH2 - OH
HH H O CH3
5 Spinosyns Avermectins
O CH 3 B1 b R = CH 3-
R (ii) R = -CH 3 (avermectin B1b ) H H OH
OH F
Cl
Emamectin
Group NS: Miscellaneous non
spi no syn A, R = H-
CONHC ONH O F
Abamectin Milbemectin
benzoate & Milbemycins Group UN: Compounds with unknown mode of action
spi no syn D, R = CH 3 - Cl
F N
Cl C F3 C F 3O N HC ON HC O
specific (multi-site) inhibitors
CF 3CHFCF2O NHCONHCO
F
Cl Cl
F
O
F Cl Cl Fluazuron Cl F Lufenuron Cl F O
C NH
C
Triflumuron
CONHCONH O C F3 C F3 O N HCON HCO C F3 CHFCF 2O NH F
Group 7: Juvenile hormone mimics F Cl
N
F F Cl
F
UNA N S
O
UNC Cl
Cl
O O
O
- + - - 3+ O -
O
O 3 O O
N
OC H 3
Chlorfluazuron C N O
CH 2 NH CONH CO
F
Flufenoxuron Cl F
Noviflumuron CH 3 N S Cl O Cl CF 3
Na2 B4 O7 10H2 O -
O
Sb
O
- -
O
Sb
O-
.2K + .3H O
2
CF3O CHFCF2O NHCO NHCO
(CH 3 )2 C (CH 2) 3
CH 3 C O 2C H2 C CH Cl O O
H C C CH CH2 H
F O
C H3 C C H
CH 3 C C H C NOCH 2CH 3
CH3
CH CH2 CH
CH3
C C
H C H3
CO 2 CH 2 C H 3 CH 3 C H2 C H2
CH C H2 H
H C C N O O O C OCH3 Chinomethionat OH Pyridalyl Borax Tartar emetic
CH3 CH 2 C H2 CH 2 C C C H CH 2 CH 3 C O 2 C H(CH 3) 2 O O C H 2C H 2N H C O 2C H 2C H 3 CH C H 2 F Cl F
H H CH 3
CH 3 O
Flucycloxuron Cl F
Novaluron CH 3O Cl
Cl C Cl
Cl NHCONHCO CHF 2CF 2O NHCO NHCO F NH CO NHC O CC l 3
Hydroprene Kinoprene Methoprene Fenoxycarb
F
Cl F
Cl F F UNB UND NSA NSB
Pyriproxyfen Benzoximate Dicofol
Diflubenzuron Hexaflumuron Teflubenzuron
15 Benzoylureas
7A Juvenile hormone analogues 7B Fenoxycarb 7C Pyriproxyfen
More information on the Insecticide Resistance Action Committee and the Mode of Action Classification is available from: www.irac-online.org or enquiries@irac-online.org
Structures are reproduced from the Pesticide Manual with permission from the British Crop Protection Council Details presented are accurate to the best of our knowledge at the time of publication but IRAC or its member companies cannot accept responsibility for how this information is used or interpreted Poster Version 1, October 2005. Based on the Mode of Action Classification - Version 5.1
You might also like
- Best of Violin Classics: 12 Famous Concert Pieces for Violin and PianoFrom EverandBest of Violin Classics: 12 Famous Concert Pieces for Violin and PianoRating: 1 out of 5 stars1/5 (1)
- Classics meet Jazz: 10 jazz fantasies on classical themes for flute and pianoFrom EverandClassics meet Jazz: 10 jazz fantasies on classical themes for flute and pianoRating: 3.5 out of 5 stars3.5/5 (14)
- Songs Without Words: Selection for piano lessonsFrom EverandSongs Without Words: Selection for piano lessonsRating: 5 out of 5 stars5/5 (1)
- Benzodiazepine Equivalents DassaDocument2 pagesBenzodiazepine Equivalents DassaPsikiater PanturaNo ratings yet
- Instant Assessments for Data Tracking, Grade 1: MathFrom EverandInstant Assessments for Data Tracking, Grade 1: MathRating: 4 out of 5 stars4/5 (1)
- Sistema Eleltrico (LMA) 320cDocument2 pagesSistema Eleltrico (LMA) 320cAlex silvaNo ratings yet
- Instant Assessments for Data Tracking, Grade 2: MathFrom EverandInstant Assessments for Data Tracking, Grade 2: MathNo ratings yet
- AMus Specimen Paper From 2017 PDFDocument11 pagesAMus Specimen Paper From 2017 PDFViolin TtpcNo ratings yet
- Tony Mcmanus Donal Og The Lea RigDocument7 pagesTony Mcmanus Donal Og The Lea RigBillybobwhoretownNo ratings yet
- 倪海夏 人纪 简体针灸教程Document242 pages倪海夏 人纪 简体针灸教程胡佳鑫No ratings yet
- DX490LC 3 HydrDocument1 pageDX490LC 3 HydrvitaliyNo ratings yet
- Tugas Mors Kelompok 5 Analisis ABC Indeks KritisDocument11 pagesTugas Mors Kelompok 5 Analisis ABC Indeks Kritishastomo prabowoNo ratings yet
- Pka de FarmacosDocument6 pagesPka de FarmacosIsidro Santiago Sanchez83% (12)
- Insecticide Resistance Action Committee: (IRAC) 和杀虫剂作用机制分类信息Document1 pageInsecticide Resistance Action Committee: (IRAC) 和杀虫剂作用机制分类信息Catherine TangNo ratings yet
- Chapter 14Document42 pagesChapter 14NayLinNo ratings yet
- Acids and Bases Review Key-1Document6 pagesAcids and Bases Review Key-1Anna BoyajyanNo ratings yet
- Sigma Metabolic Pathways-1Document1 pageSigma Metabolic Pathways-1Marianna Hipólito RochaNo ratings yet
- Mapa MetabólicoDocument1 pageMapa MetabólicoSuelenVernekMarquesNo ratings yet
- Stereochemistry Chiral Molecules QuizDocument3 pagesStereochemistry Chiral Molecules QuizSean McDivittNo ratings yet
- Frac Moa Poster 2021Document1 pageFrac Moa Poster 2021Raul Ernesto Meza CabreraNo ratings yet
- Frac Moa Poster 2022Document1 pageFrac Moa Poster 2022Fernanda Amaral FariaNo ratings yet
- AF S1 SW-1 MaxDocument1 pageAF S1 SW-1 MaxTosikur RahmanNo ratings yet
- 1 - GA - COLUMN PLAN - Rev 0Document1 page1 - GA - COLUMN PLAN - Rev 0Thuận Thảo Công tyNo ratings yet
- Pozioniranje 1-1Document1 pagePozioniranje 1-1Milan MilosavljevicNo ratings yet
- Equilibrium Expressions and CalculatingDocument7 pagesEquilibrium Expressions and CalculatingAmal JaberNo ratings yet
- Fri ModelDocument1 pageFri ModelAhmed MoharramNo ratings yet
- Redmi Note 8 (LLDM516) Schematic DiagramDocument29 pagesRedmi Note 8 (LLDM516) Schematic Diagramakash0% (1)
- Mapa MetabólicoDocument1 pageMapa MetabóliconelsonfqNo ratings yet
- DM - DQ1 DB - DQ1 DA - DQ1: Daq2 Daq5Document2 pagesDM - DQ1 DB - DQ1 DA - DQ1: Daq2 Daq5Huỳnh Văn ĐôngNo ratings yet
- CANCER Epigenetic ModificationsDocument1 pageCANCER Epigenetic ModificationsgustavopsoNo ratings yet
- Hps 1402Document1 pageHps 1402Fonoaudióloga Gisele RezendeNo ratings yet
- Dynacord DC300E ManualDocument6 pagesDynacord DC300E ManualMalapetaka50% (2)
- Poster de Las Vias Metabolicas.Document2 pagesPoster de Las Vias Metabolicas.Moises Rosales100% (1)
- OPUS SOP - Overall SchematicDocument1 pageOPUS SOP - Overall SchematicValentin Andrenoiu0% (1)
- Clasificación Mecanismos de Acción de Insecticidas - 2024Document1 pageClasificación Mecanismos de Acción de Insecticidas - 2024Pedro RodríguezNo ratings yet
- 13.5.3. Plano Diseño de Muro de Contención Ii Etapa - Bosques de La HaciendaDocument1 page13.5.3. Plano Diseño de Muro de Contención Ii Etapa - Bosques de La HaciendaLina DiazNo ratings yet
- Gbh-Pr-001-Swimming Pool LayoutDocument1 pageGbh-Pr-001-Swimming Pool LayoutvigneshviNo ratings yet
- Epigenetic ModificationsDocument1 pageEpigenetic ModificationsFerny PintoNo ratings yet
- Frac Moa Poster 2020v2Document1 pageFrac Moa Poster 2020v2NGUYỄN HỮU THÀNHNo ratings yet
- Captura de Tela 2023-03-21 À(s) 7.09.54 PMDocument1 pageCaptura de Tela 2023-03-21 À(s) 7.09.54 PMGroff DiegoNo ratings yet
- Gbh-Pr-004-Pump Room Mechanical LayoutDocument1 pageGbh-Pr-004-Pump Room Mechanical LayoutvigneshviNo ratings yet
- 09 Ddc-P2c3-Ecv01-Aln-Dr-C3e-50603-06-ADocument1 page09 Ddc-P2c3-Ecv01-Aln-Dr-C3e-50603-06-AvetrismanikkamNo ratings yet
- 9 3 0 0 V e C T o R Configuration D A T E: 0 5 - 0 5 - 2 0 0 0 C 0 0 0 5 2 0 0 0Document1 page9 3 0 0 V e C T o R Configuration D A T E: 0 5 - 0 5 - 2 0 0 0 C 0 0 0 5 2 0 0 0Pero MivanovNo ratings yet
- C 2000 PDFDocument1 pageC 2000 PDFPero MivanovNo ratings yet
- Oxidation Reactions 3Document3 pagesOxidation Reactions 3rushikesh gaikarNo ratings yet
- Mama Structure-Structure 2Document1 pageMama Structure-Structure 2Saugat ThapaNo ratings yet
- Afs Gigabyte Ga-H110m-S2h-Ddr3-1.0 (PDF Boardview)Document2 pagesAfs Gigabyte Ga-H110m-S2h-Ddr3-1.0 (PDF Boardview)Евгений КуценкоNo ratings yet
- Gbh-pr-005-Pump Room Mechanical Section DetailsDocument1 pageGbh-pr-005-Pump Room Mechanical Section DetailsvigneshviNo ratings yet
- Carrascosa - Jaime Texidor Dalmau - Set of ClarinetsDocument19 pagesCarrascosa - Jaime Texidor Dalmau - Set of ClarinetsFausti BravoNo ratings yet
- Edge Slab Detail - A: Combined Footing Plan - Cf1Document1 pageEdge Slab Detail - A: Combined Footing Plan - Cf1khem thapaNo ratings yet
- Bach bwv244Document7 pagesBach bwv244Der MagnetNo ratings yet
- Dynamic 20000 H 2ohmDocument9 pagesDynamic 20000 H 2ohmEspedito Alves SilvaNo ratings yet
- Dynamic 12000 H 2 OhmDocument8 pagesDynamic 12000 H 2 OhmEspedito Alves SilvaNo ratings yet
- Placa Mae LGA1151 v2Document2 pagesPlaca Mae LGA1151 v2paranozjpcNo ratings yet
- Dynamic 12000 H 2 OhmDocument8 pagesDynamic 12000 H 2 OhmMarcos SilvaNo ratings yet
- Gigabyte GA-H61M-S1 r3.0 BoardviewDocument2 pagesGigabyte GA-H61M-S1 r3.0 BoardviewANH TUANNo ratings yet
- GA-H61M-S1 Rev.3.0Document2 pagesGA-H61M-S1 Rev.3.0Đức Hòa Điện Thoại - Máy TínhNo ratings yet
- GA-H61M-S1 Rev.3.0Document2 pagesGA-H61M-S1 Rev.3.0Đức Hòa Điện Thoại - Máy TínhNo ratings yet
- Gigabyte GA-H61M-S1 r3.0 BoardviewDocument2 pagesGigabyte GA-H61M-S1 r3.0 Boardviewsỹ QuốcNo ratings yet
- Gigabyte GA-H61M-S1 r3.0 BoardviewDocument2 pagesGigabyte GA-H61M-S1 r3.0 Boardviewsỹ QuốcNo ratings yet
- H61M-S1 Rev.3.0 BVDocument2 pagesH61M-S1 Rev.3.0 BVĐức Hòa Điện Thoại - Máy TínhNo ratings yet
- White Shade: The Real-World Primer for the Black Professional WomanFrom EverandWhite Shade: The Real-World Primer for the Black Professional WomanNo ratings yet
- Formulary For Cardiology and Hypertension:: Brand Name Generic Name Strength CompanyDocument3 pagesFormulary For Cardiology and Hypertension:: Brand Name Generic Name Strength CompanyDr-Muhammad Junaid Hassan SharifNo ratings yet
- Antibiotik Dan Triamcinolon IntravitrealDocument17 pagesAntibiotik Dan Triamcinolon IntravitrealMeironiWaimirNo ratings yet
- Antimikroba Anti Jamur Antiparasit Antibiotik Antiviral Antimico-Bacterium Antimikosis Antihelmintik Antiamuba AntimalariaDocument22 pagesAntimikroba Anti Jamur Antiparasit Antibiotik Antiviral Antimico-Bacterium Antimikosis Antihelmintik Antiamuba AntimalariaHaris GaulNo ratings yet
- Antibiotics FamiliesDocument5 pagesAntibiotics FamiliesTiffany SamanthaNo ratings yet
- Stock 20.06.22Document25 pagesStock 20.06.22dian elokNo ratings yet
- Daftar ObatDocument1 pageDaftar ObatPramukadua DepokNo ratings yet
- Pure Antibiotic PowderDocument2 pagesPure Antibiotic PowderTitan BiotechNo ratings yet
- Table 10 Oral Antihypertensive DrugsDocument3 pagesTable 10 Oral Antihypertensive DrugsSanti ParambangNo ratings yet
- ExistenciasST 24-07-2017Document6 pagesExistenciasST 24-07-2017Lupita CVNo ratings yet
- KomperacijaDocument65 pagesKomperacijaGledam TeNo ratings yet
- Fake Dummy Data ClaimsDocument616 pagesFake Dummy Data ClaimsAnonymous ErWdFOTFNo ratings yet
- Beta LactamDocument18 pagesBeta LactamCesar Saba0% (1)
- Deuxieme MagasinDocument5 pagesDeuxieme MagasinYacouba DembeleNo ratings yet
- Tabela Venda 2014Document12 pagesTabela Venda 2014GearthipNo ratings yet
- Synthesis Urea Pusri Iv: Urea Reactor DC-101 GB-101 A/B GB-102Document5 pagesSynthesis Urea Pusri Iv: Urea Reactor DC-101 GB-101 A/B GB-102anjarNo ratings yet
- Obat-Obat AntiboitikDocument5 pagesObat-Obat AntiboitikAbdul ChristopherNo ratings yet
- Price List Cv. Untung Kumoro: End of ReportDocument144 pagesPrice List Cv. Untung Kumoro: End of ReportdedirahadiNo ratings yet
- Stokbarangglobal 14 MaretDocument114 pagesStokbarangglobal 14 MaretwuriNo ratings yet
- Golongan Obat KieDocument4 pagesGolongan Obat KienurwandahafsariNo ratings yet
- ASCIA HP Penicillin Allergy Guide 2016Document1 pageASCIA HP Penicillin Allergy Guide 2016kkkssbbNo ratings yet
- PharmacologyDocument3 pagesPharmacologyWaseem RazaNo ratings yet
- PT Quantum LabsDocument4 pagesPT Quantum LabsCipuut PutriNo ratings yet
- Formularium Fix 2Document406 pagesFormularium Fix 2Ricky HadiyanzahNo ratings yet
- Wikijournalclub:List of Landmark Papers/Cardiology: HypertensionDocument5 pagesWikijournalclub:List of Landmark Papers/Cardiology: Hypertensionrvar839No ratings yet
- Rekapitulasi Laporan Narkotika: NO Nama Satuan Stok Awal Pemasukan PBFDocument11 pagesRekapitulasi Laporan Narkotika: NO Nama Satuan Stok Awal Pemasukan PBFBang23 ManikNo ratings yet
- Copy Pedia DrugsDocument3 pagesCopy Pedia DrugsMonica LeeNo ratings yet