Professional Documents
Culture Documents
Test For Functional Gps - Aldehydes & Carboxylic Acids
Test For Functional Gps - Aldehydes & Carboxylic Acids
Uploaded by
gilchrist0 ratings0% found this document useful (0 votes)
4 views1 pageOriginal Title
Test for Functional Gps- Aldehydes & Carboxylic Acids
Copyright
© © All Rights Reserved
Available Formats
PDF, TXT or read online from Scribd
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
© All Rights Reserved
Available Formats
Download as PDF, TXT or read online from Scribd
0 ratings0% found this document useful (0 votes)
4 views1 pageTest For Functional Gps - Aldehydes & Carboxylic Acids
Test For Functional Gps - Aldehydes & Carboxylic Acids
Uploaded by
gilchristCopyright:
© All Rights Reserved
Available Formats
Download as PDF, TXT or read online from Scribd
You are on page 1of 1
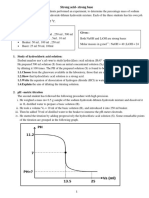
Distinguishing Tests for Functional Groups
1) Tests for Aldehydes and Carboxylic acids
EXPERIMENT OBSERVATION INFERENCE
1) To the given liquids Brisk effervescence is Test tube A contains
A and B, add a few observed in test tube A, but carboxylic acid.
drops of NaHCO3 there is no change in B.
solution.
2) Mix equal There is no change in test Test tube B contains
quantities of tube A, while reddish- aldehyde.
Fehling’s solution brown precipitate is formed
A and B. Add this in test tube B.
mixture to the given
liquids A and B and
warm in a water
bath.
RESULT: Test tube A contains carboxylic acid while test tube B contains aldehyde.
You might also like
- Test For Functional Gps - Aldehydes & KetonesDocument1 pageTest For Functional Gps - Aldehydes & KetonesgilchristNo ratings yet
- Test For Functional Gps - Phenol & AlcoholDocument1 pageTest For Functional Gps - Phenol & AlcoholgilchristNo ratings yet
- Kelm 205Document12 pagesKelm 205Soumik MukhopadhyayNo ratings yet
- Experiment Journal 4 - Group 2 - Chemistry 40Document7 pagesExperiment Journal 4 - Group 2 - Chemistry 40Daniella ChandraNo ratings yet
- Monique Tran - 27 Titration Curves - SDocument8 pagesMonique Tran - 27 Titration Curves - Smoni tranNo ratings yet
- 9dtddfAcids-Bases-and-Salt-2-2vghjjccvjjxs HjyffgDocument6 pages9dtddfAcids-Bases-and-Salt-2-2vghjjccvjjxs HjyffgKumari KNo ratings yet
- Chem Assignment NotesDocument5 pagesChem Assignment Notessnalo mdludluNo ratings yet
- Asm 10061Document1 pageAsm 10061Manvi GuptaNo ratings yet
- Chemistry Released URT Items 2018Document6 pagesChemistry Released URT Items 2018Bassant Hossam17No ratings yet
- Buffers, 2024 (Print)Document12 pagesBuffers, 2024 (Print)lh1721No ratings yet
- Implementation For Exp # 27 - Qualitative Analysis of Inorganic SaltsDocument6 pagesImplementation For Exp # 27 - Qualitative Analysis of Inorganic Saltsezethomas6No ratings yet
- Experiment 7: Acid, Bases and Salts: Page 1 of 4Document4 pagesExperiment 7: Acid, Bases and Salts: Page 1 of 4danicagalvan0% (1)
- TB 82pHandpOHofstrongacidandbase 61edf8eca0b257.61edf8edc066f0.51892966Document6 pagesTB 82pHandpOHofstrongacidandbase 61edf8eca0b257.61edf8edc066f0.51892966任思诗No ratings yet
- Worksheet 2: Acid and BasesDocument2 pagesWorksheet 2: Acid and BasesRayNo ratings yet
- Laboratory Report No. 1Document4 pagesLaboratory Report No. 1YvonneNo ratings yet
- Hydrolysis of Salt and The Action of Buffer SolutionDocument5 pagesHydrolysis of Salt and The Action of Buffer SolutionFaisal MumtazNo ratings yet
- Laboratory: (Buffer Solution Experiment Using Red Cabbage As Indicator)Document25 pagesLaboratory: (Buffer Solution Experiment Using Red Cabbage As Indicator)Reylsea MayNo ratings yet
- Experiment Manual of CITRATE-NA2HPO4 BUFFERDocument5 pagesExperiment Manual of CITRATE-NA2HPO4 BUFFERnindi silvianaNo ratings yet
- Buffers and Redox Laboratory 6 v2Document12 pagesBuffers and Redox Laboratory 6 v2skyeandoNo ratings yet
- Investigation of Strong Acid and Strong BaseDocument8 pagesInvestigation of Strong Acid and Strong BasePamNo ratings yet
- Extra Ex1 Strong Acid and Strong BaseDocument2 pagesExtra Ex1 Strong Acid and Strong BaseSara KhalifehNo ratings yet
- Effects of Buffer On PHDocument5 pagesEffects of Buffer On PHAnge OuedraogoNo ratings yet
- MODULE 2 - Group 4Document6 pagesMODULE 2 - Group 4Anne Therese / Annie KanaanNo ratings yet
- Lab chm301 Carboxylic AcidDocument7 pagesLab chm301 Carboxylic AcidbbbbNo ratings yet
- CHM 104 PQ Answer 1Document6 pagesCHM 104 PQ Answer 1bdbtghzk5jNo ratings yet
- CPB 30103 Biochemical Engineering UniKL MICET Experiment 1: Preparation of Buffer Solution Full Lab ReportDocument10 pagesCPB 30103 Biochemical Engineering UniKL MICET Experiment 1: Preparation of Buffer Solution Full Lab ReportSiti Hajar Mohamed0% (1)
- Experiment 1 CHM 420Document6 pagesExperiment 1 CHM 420bellaamin100% (1)
- Final Lab Report 2Document5 pagesFinal Lab Report 2api-340388320No ratings yet
- Experiment 2 Standardization of Sodium Hydroxide With HCLDocument2 pagesExperiment 2 Standardization of Sodium Hydroxide With HCLvafaashkNo ratings yet
- ACFrOgCaBCFxgI1puepNIEiilX MI5sqz9qE64xv2eIt73WI8E4L3T4L ZrdEd2CUynymN MwnUuRCIkz2i48bB4Ko4 TH1yDkOSR4rYahi3J9wwOvjOc n6bg54A0mDaLu56SWYLCzdGJu1CwkDocument4 pagesACFrOgCaBCFxgI1puepNIEiilX MI5sqz9qE64xv2eIt73WI8E4L3T4L ZrdEd2CUynymN MwnUuRCIkz2i48bB4Ko4 TH1yDkOSR4rYahi3J9wwOvjOc n6bg54A0mDaLu56SWYLCzdGJu1CwkLanaya Puti Anma Adriansyah LanayaNo ratings yet
- Chem LabDocument20 pagesChem LabKate Mae GeronimoNo ratings yet
- TB 85titration 61edfa70ef6ee4.61edfa733da046.15966159Document33 pagesTB 85titration 61edfa70ef6ee4.61edfa733da046.15966159任思诗No ratings yet
- Experiment 6 Acid and Bases CHM 420Document9 pagesExperiment 6 Acid and Bases CHM 420najwa nabila100% (1)
- Applications of Aqueous EquilibriaDocument24 pagesApplications of Aqueous EquilibriaEuler MendozaNo ratings yet
- Written ReportDocument4 pagesWritten ReportCarla Solina Olivares GuicoNo ratings yet
- Buffer Preparation and PH Measurement Using The Electrometric Method and Colorimetric MethodDocument2 pagesBuffer Preparation and PH Measurement Using The Electrometric Method and Colorimetric MethodArndrei CunananNo ratings yet
- Organic Lab 6Document19 pagesOrganic Lab 6badirmhammadNo ratings yet
- AP Chemistry Study Guide: Chapter 14: Acids and Bases and Chapter 15, 16.1 and 21.3: Aqueous and Acid-Base EquilibriaDocument8 pagesAP Chemistry Study Guide: Chapter 14: Acids and Bases and Chapter 15, 16.1 and 21.3: Aqueous and Acid-Base Equilibrialorraine_cuaNo ratings yet
- 4 TitrationDocument14 pages4 TitrationTing Choon YinNo ratings yet
- Preparation of BuffersDocument4 pagesPreparation of Bufferskleyr100% (1)
- I. Preliminary Examination: Grade: Xi/Xii (Chemistry Practicals)Document3 pagesI. Preliminary Examination: Grade: Xi/Xii (Chemistry Practicals)Vanisha SinghNo ratings yet
- Soal Titrasi Asam Basa1 (Annisa Rahmah 17176001)Document3 pagesSoal Titrasi Asam Basa1 (Annisa Rahmah 17176001)annisa rahmahNo ratings yet
- Experiment Number 7Document6 pagesExperiment Number 7api-457258209No ratings yet
- Carboxylic Acids and Their DerivativesDocument5 pagesCarboxylic Acids and Their DerivativesCrissalyn AyentoNo ratings yet
- SadasdsadsadDocument2 pagesSadasdsadsadArndrei CunananNo ratings yet
- Regine T. Diaz, Lordjel Kin M. Eleda: Figure 1. PH MeterDocument2 pagesRegine T. Diaz, Lordjel Kin M. Eleda: Figure 1. PH MeterArndrei CunananNo ratings yet
- Chem (Ii) 5Document12 pagesChem (Ii) 5Nurul Hasanah100% (1)
- PH and Buffers ReportDocument7 pagesPH and Buffers ReportMirandaNo ratings yet
- Fehling's Solution Is A Chemical Reagent Used To Differentiate BetweenDocument2 pagesFehling's Solution Is A Chemical Reagent Used To Differentiate BetweenSankar AdhikariNo ratings yet
- Wis Ap Chem Lab 17 Buffer Solutions ArcherDocument7 pagesWis Ap Chem Lab 17 Buffer Solutions Archerapi-201479236No ratings yet
- Acid Base Titrations PDFDocument34 pagesAcid Base Titrations PDFsherryNo ratings yet
- Experiment 5: Common Ion Effect: Prof. Kreza LigayaDocument16 pagesExperiment 5: Common Ion Effect: Prof. Kreza LigayaIna ChiuNo ratings yet
- Experiment-Reducing SugarsDocument2 pagesExperiment-Reducing SugarsRyanNo ratings yet
- Ammonium Carbonate and Lead AcetateDocument8 pagesAmmonium Carbonate and Lead AcetatedallenainaNo ratings yet
- Experiment 9 Formal ReportDocument5 pagesExperiment 9 Formal ReportTrishaNo ratings yet
- Acids Bases and SaltsDocument3 pagesAcids Bases and SaltsGauravNo ratings yet
- Acids Bases and Salts: A Solution Turns Red Litmus Blue Its PH Is Likely To Be A) 1 B) 4 C) 5 D) 10Document6 pagesAcids Bases and Salts: A Solution Turns Red Litmus Blue Its PH Is Likely To Be A) 1 B) 4 C) 5 D) 10is-hakNo ratings yet
- Ch-2: Acids, Bases and Salts Question BankDocument2 pagesCh-2: Acids, Bases and Salts Question Bankvrat0% (2)
- Functional GroupsDocument5 pagesFunctional GroupsDhairya KumarNo ratings yet
- Relations and Functions Board Exam QuestionsDocument4 pagesRelations and Functions Board Exam QuestionsgilchristNo ratings yet
- Inverse Trig FnsDocument3 pagesInverse Trig FnsgilchristNo ratings yet
- 12 Half Yerly Examination - Marking SchemeDocument13 pages12 Half Yerly Examination - Marking SchemegilchristNo ratings yet
- VivaDocument4 pagesVivagilchristNo ratings yet
- Practical ExamDocument21 pagesPractical ExamgilchristNo ratings yet