Professional Documents
Culture Documents
Chemical Kinetics Formulas
Chemical Kinetics Formulas
Uploaded by
vedant tomar0 ratings0% found this document useful (0 votes)
8 views2 pagesThis document discusses the kinetics of zero-order and first-order chemical reactions. For zero-order reactions, the rate is constant and the integrated rate law gives the concentration as a linear function of time. The half-life of a zero-order reaction is independent of concentration. For first-order reactions, the rate is proportional to concentration and the integrated rate law gives the natural log of the concentration as a linear function of time. The half-life of a first-order reaction is the same regardless of initial concentration.
Original Description:
epic formulas hand made
Original Title
chemical kinetics formulas
Copyright
© © All Rights Reserved
Available Formats
DOCX, PDF, TXT or read online from Scribd
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentThis document discusses the kinetics of zero-order and first-order chemical reactions. For zero-order reactions, the rate is constant and the integrated rate law gives the concentration as a linear function of time. The half-life of a zero-order reaction is independent of concentration. For first-order reactions, the rate is proportional to concentration and the integrated rate law gives the natural log of the concentration as a linear function of time. The half-life of a first-order reaction is the same regardless of initial concentration.
Copyright:
© All Rights Reserved
Available Formats
Download as DOCX, PDF, TXT or read online from Scribd
0 ratings0% found this document useful (0 votes)
8 views2 pagesChemical Kinetics Formulas
Chemical Kinetics Formulas
Uploaded by
vedant tomarThis document discusses the kinetics of zero-order and first-order chemical reactions. For zero-order reactions, the rate is constant and the integrated rate law gives the concentration as a linear function of time. The half-life of a zero-order reaction is independent of concentration. For first-order reactions, the rate is proportional to concentration and the integrated rate law gives the natural log of the concentration as a linear function of time. The half-life of a first-order reaction is the same regardless of initial concentration.
Copyright:
© All Rights Reserved
Available Formats
Download as DOCX, PDF, TXT or read online from Scribd
You are on page 1of 2
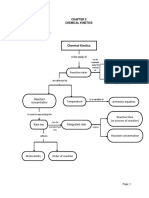
For zeroth order reaction
Integrated law of rate
Half life for zeroth order
Total time of zero order reaction
For 1st order reaction
Integrated law of rate
or
Time equation
Half life of the reaction
For 1st order reaction
You might also like
- CFE - 10 - Chemical Kinetics Part 2Document19 pagesCFE - 10 - Chemical Kinetics Part 2Christian C. SuaseNo ratings yet
- Chemical Kinetics GibbsDocument35 pagesChemical Kinetics GibbsTHE CREATORSTERNo ratings yet
- Chemical Kinetics - NotesDocument6 pagesChemical Kinetics - Notesn611704No ratings yet
- CBSE Class 12 Chemistry Notes: Chemical Kinetics: HomepageDocument14 pagesCBSE Class 12 Chemistry Notes: Chemical Kinetics: HomepageBHAVYA BNo ratings yet
- Module 2 Kinetics of Materials Reading MaterialsDocument14 pagesModule 2 Kinetics of Materials Reading MaterialsmayNo ratings yet
- ChemistryDocument12 pagesChemistryAbdullahSohailNo ratings yet
- Chemical Kinetics: Quick RevisionDocument20 pagesChemical Kinetics: Quick Revisionfakeheartattack8kNo ratings yet
- Chemistry Notes For Class 12 Chapter 4 Chemical KineticsDocument11 pagesChemistry Notes For Class 12 Chapter 4 Chemical Kineticspala100% (1)
- Long Exam 1: Overview of Chemical Reaction EngineeringDocument2 pagesLong Exam 1: Overview of Chemical Reaction EngineeringKurt BiduaNo ratings yet
- 4 - The Rates of Chemical Reactions - ADocument42 pages4 - The Rates of Chemical Reactions - AÇetin UzişNo ratings yet
- Lesson - 7 Chemical Kinetics: Average RateDocument4 pagesLesson - 7 Chemical Kinetics: Average RateAnnamalai BoomikaNo ratings yet
- Chemical Kinetics: Recap of Early ClassesDocument72 pagesChemical Kinetics: Recap of Early ClassesRaashiNo ratings yet
- Chemical Kinetics PDFDocument42 pagesChemical Kinetics PDFrockingrazzNo ratings yet
- Chemistry Notes For Class 12 Chapter 4 Chemical KineticsDocument11 pagesChemistry Notes For Class 12 Chapter 4 Chemical KineticsAyush singh PrinceNo ratings yet
- L-Chemical KineticsDocument96 pagesL-Chemical Kineticssalma khanNo ratings yet
- Chapter - 4: Chemical KineticsDocument21 pagesChapter - 4: Chemical KineticsJeffrey ImmanuelNo ratings yet
- Rates of ReactionDocument33 pagesRates of ReactionMd. Mujahid HasanNo ratings yet
- Integral Rate Law, Half-LifeDocument10 pagesIntegral Rate Law, Half-LifeaminNo ratings yet
- First Law of ThermodynamicsDocument2 pagesFirst Law of ThermodynamicsPrajwal PatilNo ratings yet
- Physics Project Report Class 10Document28 pagesPhysics Project Report Class 10Deepak SinghNo ratings yet
- Athirah Multazimah Binti Ali Z - Tee0817 4s1-5s1/09-10Document5 pagesAthirah Multazimah Binti Ali Z - Tee0817 4s1-5s1/09-10athir_ahNo ratings yet
- Chemical Kinetics 2Document39 pagesChemical Kinetics 2Md. Hasanur RahmanNo ratings yet
- I Odin Ization of AcetoneDocument15 pagesI Odin Ization of AcetoneanfatallaNo ratings yet
- A. The Title: Reaction Rate B. Date of The Experiment Begin: Tuesday / April, 4Document37 pagesA. The Title: Reaction Rate B. Date of The Experiment Begin: Tuesday / April, 4NurvitaEkaWulanNo ratings yet
- Chemical Kinetics (Lecture Note)Document12 pagesChemical Kinetics (Lecture Note)Mayookh elambulasseryNo ratings yet
- Year 13 IAL Chemistry: Lessons Sunday, Monday, Tuesday Wednesday, ThursdayDocument41 pagesYear 13 IAL Chemistry: Lessons Sunday, Monday, Tuesday Wednesday, ThursdayjohnNo ratings yet
- Kinetics Mastery AnswersDocument5 pagesKinetics Mastery AnswersAnonymous vRpzQ2BLNo ratings yet
- Chapter 9Document18 pagesChapter 9JeromeNo ratings yet
- Chemical KineticsDocument1 pageChemical KineticsSachinNo ratings yet
- WastedDocument9 pagesWastedMark Francis DaculanNo ratings yet
- Ap ChemDocument6 pagesAp ChemholaholaholacarlottaNo ratings yet
- Determination of Rate of ReactionDocument1 pageDetermination of Rate of ReactiondarshinyyvijayanNo ratings yet
- Chemical KineticsDocument27 pagesChemical KineticsRoshen RonyNo ratings yet
- Rate Equation: Zeroth-Order ReactionsDocument16 pagesRate Equation: Zeroth-Order ReactionsBastab DeyNo ratings yet
- KineticsDocument2 pagesKineticsSharad GuptaNo ratings yet
- Block 2BBCCT 107Document44 pagesBlock 2BBCCT 107robinNo ratings yet
- Teknik Reaksi Kimia I: by HaryantoDocument23 pagesTeknik Reaksi Kimia I: by HaryantoHarymsl MslNo ratings yet
- Chemical Reaction Engineering: Cap Iii: Rate Laws and StoichiometryDocument53 pagesChemical Reaction Engineering: Cap Iii: Rate Laws and StoichiometryMarthaAlbaGuevaraNo ratings yet
- Kinetics NotesDocument4 pagesKinetics NotesDharaneesh S.k.No ratings yet
- Zero Order: Integral MethodDocument2 pagesZero Order: Integral MethodmuhammadismailNo ratings yet
- Required Practical Revision SheetsDocument13 pagesRequired Practical Revision SheetsgsapkaiteNo ratings yet
- CHM 241Document24 pagesCHM 241Adams DeborahNo ratings yet
- CHem InalsDocument3 pagesCHem InalsAbigail OconNo ratings yet
- CBRE Module 1 Part 1Document21 pagesCBRE Module 1 Part 1Ronima Rajive100% (1)
- Module 4b - Control SystemsDocument31 pagesModule 4b - Control SystemsSyed Muhammad AliNo ratings yet
- FactorDocument2 pagesFactorJitendra KumarNo ratings yet
- CH CRE Sample Career Avenues PDFDocument13 pagesCH CRE Sample Career Avenues PDFPravin AgalaveNo ratings yet
- Lech 10994Document28 pagesLech 10994Deadly OopsNo ratings yet
- Chemical Kinetics - ClearDocument18 pagesChemical Kinetics - ClearDeviNo ratings yet
- Lec 9 & 10Document10 pagesLec 9 & 10asim zeshanNo ratings yet
- Chemical KineticsDocument40 pagesChemical KineticsHirdesh Sehgal100% (3)
- Kinetics Handout 3510Document2 pagesKinetics Handout 3510anon_870764932No ratings yet
- One Stop For Colleges Education Career: Minglebox Engineering PrepDocument14 pagesOne Stop For Colleges Education Career: Minglebox Engineering PrepVigneshwar DhamodharanNo ratings yet
- XII - CHEMICAL KINETICS - Module 2Document5 pagesXII - CHEMICAL KINETICS - Module 2Rahul Joseph ThomasNo ratings yet
- Chemical KineticsDocument34 pagesChemical Kineticskingrustam950No ratings yet
- Chemical Kinetics - QUESTION BANK XII CHEMDocument15 pagesChemical Kinetics - QUESTION BANK XII CHEMswastik aroraNo ratings yet
- Topic 1Document41 pagesTopic 1ainmnrhNo ratings yet
- Wave OpticsDocument2 pagesWave Opticsvedant tomarNo ratings yet
- PE NotesDocument82 pagesPE Notesvedant tomarNo ratings yet
- Electromagnetic WavesDocument2 pagesElectromagnetic Wavesvedant tomarNo ratings yet
- Dual Nature of Radiation and MatterDocument2 pagesDual Nature of Radiation and Mattervedant tomarNo ratings yet
- Alternating CurrentDocument5 pagesAlternating Currentvedant tomarNo ratings yet