Professional Documents
Culture Documents
Mol Asetosal Mol Naoh Mol Asetosal (V X N) : Rumus
Mol Asetosal Mol Naoh Mol Asetosal (V X N) : Rumus
Uploaded by
Tian Nugraha0 ratings0% found this document useful (0 votes)
1 views1 pageThis document provides formulas for calculating the amount of acetic acid in a sample. The amount of acetic acid moles equals the amount of NaOH moles used in the reaction. The mass of acetic acid can be calculated by multiplying the moles of acetic acid by its molar mass. The concentration in grams per milliliter is the mass of acetic acid divided by the total volume of the sample.
Original Description:
notes
Original Title
rumus
Copyright
© © All Rights Reserved
Available Formats
DOCX, PDF, TXT or read online from Scribd
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentThis document provides formulas for calculating the amount of acetic acid in a sample. The amount of acetic acid moles equals the amount of NaOH moles used in the reaction. The mass of acetic acid can be calculated by multiplying the moles of acetic acid by its molar mass. The concentration in grams per milliliter is the mass of acetic acid divided by the total volume of the sample.
Copyright:
© All Rights Reserved
Available Formats
Download as DOCX, PDF, TXT or read online from Scribd
0 ratings0% found this document useful (0 votes)
1 views1 pageMol Asetosal Mol Naoh Mol Asetosal (V X N) : Rumus
Mol Asetosal Mol Naoh Mol Asetosal (V X N) : Rumus
Uploaded by
Tian NugrahaThis document provides formulas for calculating the amount of acetic acid in a sample. The amount of acetic acid moles equals the amount of NaOH moles used in the reaction. The mass of acetic acid can be calculated by multiplying the moles of acetic acid by its molar mass. The concentration in grams per milliliter is the mass of acetic acid divided by the total volume of the sample.
Copyright:
© All Rights Reserved
Available Formats
Download as DOCX, PDF, TXT or read online from Scribd
You are on page 1of 1
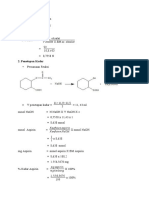
Rumus
Mol asetosal = mol NaOH mol asetosal = (V x N)NaOH
massa asetosal = mol asetosal x BEasetosal = (V x N)NaOH x BEasetosal
( )
Kadar (gr/mL) =
You might also like
- Lab Report CHM 256Document9 pagesLab Report CHM 256mhd sssyamilNo ratings yet
- Section 11: Calculations Involving Chemicals in SolutionDocument15 pagesSection 11: Calculations Involving Chemicals in SolutionTravel UnlimitedNo ratings yet
- What Type of Reaction? HCL + Naoh H O + Nacl: Neutralization Acid Base Salt Water Salt Cation AnionDocument9 pagesWhat Type of Reaction? HCL + Naoh H O + Nacl: Neutralization Acid Base Salt Water Salt Cation AnionelenaNo ratings yet
- A × M × V B × M ×V B × M ×V A×v: As .Oksalat As .Oksalat Naoh Naoh Naoh Naoh As - OksalatDocument4 pagesA × M × V B × M ×V B × M ×V A×v: As .Oksalat As .Oksalat Naoh Naoh Naoh Naoh As - OksalatElseria Togatorop 071No ratings yet
- Model Answer Lesson 5 Chapter 2Document12 pagesModel Answer Lesson 5 Chapter 2alaamza2020No ratings yet
- CHY 47.1 Sample Calculation Factors Affecting Buffer Capacity 1st Sem SY 2021-2022Document7 pagesCHY 47.1 Sample Calculation Factors Affecting Buffer Capacity 1st Sem SY 2021-2022Kathryne May JinonNo ratings yet
- CH Cooc H: AppendiksDocument5 pagesCH Cooc H: AppendiksKatherine PutriNo ratings yet
- Andat Acara 4 Kimfis KelompokDocument5 pagesAndat Acara 4 Kimfis Kelompokizani maulanaNo ratings yet
- D. Data Hasil Praktikum 1. Pembuatan Larutan Standar HCL 0,1 MDocument3 pagesD. Data Hasil Praktikum 1. Pembuatan Larutan Standar HCL 0,1 MGilbert Rizal FawwaziNo ratings yet
- Perhitungan HidrolisisDocument16 pagesPerhitungan HidrolisisUtari Ika CahyaniNo ratings yet
- Lembar PerhitunganDocument12 pagesLembar PerhitunganArie SetieawanNo ratings yet
- Hasil Praktikum Diketahui:: BE N NDocument3 pagesHasil Praktikum Diketahui:: BE N NsekarNo ratings yet
- Assay of AspirinDocument10 pagesAssay of Aspirindavin otooleNo ratings yet
- Phenolphthaleinalkalinity (MG L As Caco 3) Volume of HCL Used, ML X N HCL X 50,000 Volume of Sample Used, ML X 10 0Document1 pagePhenolphthaleinalkalinity (MG L As Caco 3) Volume of HCL Used, ML X N HCL X 50,000 Volume of Sample Used, ML X 10 0DCRUZNo ratings yet
- Lembar Perhitungan HMJ 2 GatauDocument6 pagesLembar Perhitungan HMJ 2 GatauRendy Renaldy FirdausNo ratings yet
- AIM-To Determine The Strength of The Given Sodium Hydroxide Solution. Provided Is M/20 Oxalic Acid Solution TheoryDocument2 pagesAIM-To Determine The Strength of The Given Sodium Hydroxide Solution. Provided Is M/20 Oxalic Acid Solution TheoryBhavya DalalNo ratings yet
- Lembar Perhitungan Ester p0Document10 pagesLembar Perhitungan Ester p0Zahra AlifiaNo ratings yet
- Lemper RevisedDocument7 pagesLemper RevisedTitin Setya MuktiNo ratings yet
- Data Pengamatan: MG Naoh Be Naoh X Vasam Oksalat MG NaohDocument2 pagesData Pengamatan: MG Naoh Be Naoh X Vasam Oksalat MG Naohriska mulyasNo ratings yet
- Lembar Perhitungan HMJ 2Document6 pagesLembar Perhitungan HMJ 2Rendy Renaldy FirdausNo ratings yet
- Hitungan Titrasi Asam BasaDocument1 pageHitungan Titrasi Asam BasaNur Laili FitrianiNo ratings yet
- Penentuan Bilangan PeroksidaDocument1 pagePenentuan Bilangan Peroksidasyawal12345No ratings yet
- Data Dan Perhitungan P11Document5 pagesData Dan Perhitungan P11Shinta SetyowatiNo ratings yet
- Grafik Hubungan Antara Viskositas Terhadap PH: 0.8675 0.868 F (X) - 0.0004702419x + 0.8685250968 R 0.9037957466Document2 pagesGrafik Hubungan Antara Viskositas Terhadap PH: 0.8675 0.868 F (X) - 0.0004702419x + 0.8685250968 R 0.9037957466ine rahayuNo ratings yet
- Analisis Data Isoterm (4-6)Document10 pagesAnalisis Data Isoterm (4-6)Ahmad FandiNo ratings yet
- Part BC ComputationDocument6 pagesPart BC ComputationReylhen BansilNo ratings yet
- Tugas KimfarDocument7 pagesTugas KimfarNisa AyuNo ratings yet
- Solutions PDFDocument7 pagesSolutions PDFMaria AngelinNo ratings yet
- Concentration Units: Percent and Molarity. We Will Consider Molarity Here and Mass Percent Later. MolarityDocument7 pagesConcentration Units: Percent and Molarity. We Will Consider Molarity Here and Mass Percent Later. MolarityEdgar GarciaNo ratings yet
- Concentration Units: Percent and Molarity. We Will Consider Molarity Here and Mass Percent Later. MolarityDocument7 pagesConcentration Units: Percent and Molarity. We Will Consider Molarity Here and Mass Percent Later. MolarityEdgar GarciaNo ratings yet
- MolarityDocument6 pagesMolarityRenzNo ratings yet
- Lampiran PerhitunganDocument9 pagesLampiran PerhitunganErinNo ratings yet
- Stoikiometri: Mol Pereaksiberlebihan Mol Pereksiterbatas Mol Pereaksi TerbatasDocument5 pagesStoikiometri: Mol Pereaksiberlebihan Mol Pereksiterbatas Mol Pereaksi TerbatasJhon Chalvin GintingNo ratings yet
- Calculation A. Preparation of The Hydrochloric Acid (HCL) SolutionDocument4 pagesCalculation A. Preparation of The Hydrochloric Acid (HCL) SolutionCtNo ratings yet
- Polymer Reaction Engineering Labexp##1Document4 pagesPolymer Reaction Engineering Labexp##1Rana AnasNo ratings yet
- Latihan Soal TitrasiDocument15 pagesLatihan Soal TitrasifirstcaNo ratings yet
- Class 12 Chap-1 Chemistry NotesDocument20 pagesClass 12 Chap-1 Chemistry NotesPranothi PrashanthNo ratings yet
- Environmental Science and Engineering: Assignment No. 8: Solid Waste ManagementDocument3 pagesEnvironmental Science and Engineering: Assignment No. 8: Solid Waste ManagementLucienne Iriana0% (1)
- Experiment 3: Neutralization Capacity of Commercial Antacid TabletDocument6 pagesExperiment 3: Neutralization Capacity of Commercial Antacid TabletNur Aliya Ikmal Hisham100% (1)
- Adsorption of Oxalic AcidDocument2 pagesAdsorption of Oxalic AcidDebadyuti SarkarNo ratings yet
- Exp 01Document2 pagesExp 01nayeemislammeghNo ratings yet
- Batch ReactorDocument5 pagesBatch ReactorSayyeda Neha FatimaNo ratings yet
- I. Perhitungan 6.1 Pembuatan Larutan Etilasetat 0,02 M: Mol Gram GramDocument7 pagesI. Perhitungan 6.1 Pembuatan Larutan Etilasetat 0,02 M: Mol Gram GramMade KrisnaNo ratings yet
- Calculos AAIDocument3 pagesCalculos AAIPatty RubyNo ratings yet
- Massa Jenis) × (Volume) Berat Molekul) Jumlah Mol A Jumlah Mol (A +B) Jumlah Mol B Jumlah Mol (A +B)Document4 pagesMassa Jenis) × (Volume) Berat Molekul) Jumlah Mol A Jumlah Mol (A +B) Jumlah Mol B Jumlah Mol (A +B)Ivan FadhillahNo ratings yet
- Analytical Chem EXP 1 OCEDocument12 pagesAnalytical Chem EXP 1 OCEWilliam ChiengNo ratings yet
- Pembakuan: V Naoh X BM As - OksalatDocument2 pagesPembakuan: V Naoh X BM As - OksalatFirman ImanudinNo ratings yet
- Distillation AbsorptionDocument245 pagesDistillation AbsorptionDikshant NautiyalNo ratings yet
- Lampiran: Gram BM 1000 ML Gram 40 1000 2000Document10 pagesLampiran: Gram BM 1000 ML Gram 40 1000 2000hafizatmasasmitaNo ratings yet
- Pchem10e Solutions ch05Document39 pagesPchem10e Solutions ch05nohnarimNo ratings yet
- Lembar Perhitungan HMJDocument6 pagesLembar Perhitungan HMJRendy Renaldy FirdausNo ratings yet
- 2 Physical Chemistry Laboratory - 07 - 09 - 21 PDFDocument18 pages2 Physical Chemistry Laboratory - 07 - 09 - 21 PDFAakash BanerjeeNo ratings yet
- Analisis 4Document9 pagesAnalisis 4Nhur RahmahNo ratings yet
- Lecture - 3 NormalityDocument7 pagesLecture - 3 NormalityGeorgeNo ratings yet
- Titration ProblemsDocument8 pagesTitration ProblemsAngela KocevskaNo ratings yet
- Acid-Base Titration With Lactic AcidDocument8 pagesAcid-Base Titration With Lactic AcidsinapovanadiaNo ratings yet
- Lampiran B Contoh Perhitungan: B.1 Pembuatan Larutan Yang Digunakan B.1.1 Pembuatan Larutan CH COOH 0,5 M 150 MLDocument3 pagesLampiran B Contoh Perhitungan: B.1 Pembuatan Larutan Yang Digunakan B.1.1 Pembuatan Larutan CH COOH 0,5 M 150 MLfatimah fahira siregarNo ratings yet
- Lembar Perhitungan EsterDocument7 pagesLembar Perhitungan EsterpanjigomisNo ratings yet
- Analytical 2Document8 pagesAnalytical 2Victor MbowuraNo ratings yet