Professional Documents
Culture Documents
Lembar Perhitungan
Uploaded by
diniOriginal Description:
Original Title
Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
Lembar Perhitungan
Uploaded by
diniCopyright:
Available Formats
P3
LEMBAR PERHITUNGAN REAGEN
Menghitung Densitas Reagen
Asam Asetat
m picno kosong
= 22,257 gr
V picno
= 24,136 ml
m picno + Asam Asetat
= 47,648 gr
asam asetat
= 1,05199
Etanol
m picno kosong
= 22,257 gr
V picno
= 24,136 ml
m picno + Etanol
= 42,710 gr
Etanol
=
= 0,847
H2SO4
m picno kosong
= 22,257 gr
V picno
= 24,136 ml
m picno + H2SO4
H2SO4
= 66,329 gr
=
= 1,826
Menghitung Kebutuhan NaOH
NaOH 0,35 N
Basis
= 500 ml
N NaOH
x eq x kadar
P3
m NaOH
m NaOH
= 7,14 gram
Menghitung kebutuhan katalis H2SO4 0,25 N
N H2SO4
x ek x kadar
= 3, 19 gram
volume
volume
volume
= 1,75 ml
Menghitung kebutuhan Etanol dan Asam Asetat (Variabel 1)
V etanol + V asam asetat
= 250 ml V HCl
= 250 ml - 1,75 ml
= 248,25 ml
Mol asam asetat : mol etanol = 1 : 2,3
2,3
2,3
(0,0395 ml) Vasetat
= (0,01747 ml) Vetanol
Vasetat
= 0,44228 Vetanol
Vasetat + Vetanol
= 248,25 ml
0,44228 Vetanol + Vetanol
= 248,25ml
P3
1,44228 Vetanol
= 248,25 ml
Vetanol
= 172,1 ml
= 248,25 ml 172,1 ml
Vasetat
Vasetat
= 76,15 ml
Menghitung kebutuhan Etanol dan Asam Asetat (Variabel 2)
Mol asam asetat : mol etanol = 1 : 2,8
2,8
2,8
(0,04807 ml) Vasetat
= (0,01747 ml) Vetanol
Vasetat
= 0,36343 Vetanol
Vasetat + Vetanol
= 248,25 ml
0,36343 Vetanol + Vetanol
= 248,25ml
1,36343 Vetanol
= 248,25 ml
Vetanol
Vasetat
Vasetat
= 182,1 ml
= 248,25 ml 182,1 ml
= 66,15 ml
P3
LEMBAR PERHITUNGAN
Perhitungan Konversi Variabel 1
CH3COOH + C2H5OH CH3COOC2H5 + H2O
A
Gram asam asetat
= v kadar
= 1,05199
76,15 ml 0.98
= 78,447 gram
CAO
=
= 5,2254 mol/L
Gram etanol
= v kadar
= 0,847
172,1 ml 0.96
= 139,938 gram
CBO
=
=
= 12,15 mol/L
CA
=
=
= 0.07V 0,25
Xa
= 1-
P3
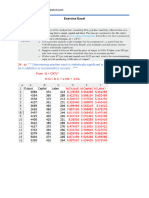
t(menit)
Volume NaOH
Ca= 0.07V-
Xa
(ml)
0,25
67
4,44
0,15
11
47,1
3,047
0,417
22
46,6
3,012
0,423
33
38,3
2,431
0,535
44
35,1
2,207
0,578
CH3COOH + C2H5OH CH3COOC2H5 + H2O
A
Stoikiometri
CA
= CAO (1-XA)
= CAO CAO.XA
= 5,2254 (1-XA)
CB
= CBO CAO.XA
= 12,15 5,2254 XA
CC
= CAO.XA
= 5,2254 XA
CD
= CAO.XA
= 5,2254 XA
= 2,325
P3
=
= k1(
= k1 (
= k1 (
= k1 (
= k1 .
= k1
= k1 .
.
(
)
/
/
/
Pada saat kesetimbangan
K=
Konstanta Kesetimbangan
K=
=
= k1
= k1
= 0,453
(
/
/
Mencari nilai
=
=
=
+
*
P3
Rumus ABC
= (XA + 3,35)(XA - 0,579)
-0,16
=
+
-0,579A + 3,35B
=1
=0
-0,579A
3,35B
=1
A = -0,254
B = 0,254
Sehingga persamaan menjadi
-0,16*
-0,16*
+ = k1 . t
-+
k1 . t
k1 . t
0,04 [
0,04[
0,04 [ ln (
]
-
] = k1 . t
) = k1 . t
k1. t
P3
0,04 [ ln (
) = k1 . t
0,04ln *
t(x)
+ = k1 . t y = mx
Xa
X2
Xy
0,15
0,0137
11
0,417
0,055
0,605
121
22
0,423
0,057
1,254
484
33
0,535
0,11
3,63
1089
44
0,578
0,26
11,44
1936
K=
k2 =
= 4,3.10-3 mol/menit
=
= 9,5.10-3mol/menit
Perhitungan Konversi Variabel 2
CH3COOH + C2H5OH CH3COOC2H5 + H2O
A
Gram asam asetat
= v kadar
= 1,05199
66,15 ml 0.98
= 68,197 gram
P3
CAO
=
= 4,543 mol/L
Gram etanol
= v kadar
= 0,847
182,1 ml 0.96
= 148,07 gram
CBO
=
=
= 12,856 mol/L
CA
=
=
= 0.07V 0,25
Xa
= 1-
t(menit)
Volume NaOH
Ca= 0.07V-0,25
Xa
(ml)
56,2
3,684
0,189
11
45,7
2,949
0,35
22
40,9
2,55
0,44
P3
33
36,0
2,27
0,5
44
30,8
1,906
0,580
CH3COOH + C2H5OH CH3COOC2H5 + H2O
A
Stoikiometri
CA
= CAO (1-XA)
= CAO CAO.XA
= 4,543 (1-XA)
= CBO CAO.XA
CB
= 12,856 4,543 XA
CC
= CAO.XA
= 4,543.XA
CD
= CAO.XA
= 4,543.XA
= 2,83
= k1 (
= k1 (
= k1 (
= k1 (
= k1 .
)
/
P3
= k1
= k1 .
/
/
Pada saat kesetimbangan
K=
Konstanta Kesetimbangan
K=
=
= k1
= k1
= 0,356
(
/
/
Mencari nilai
*
+
*
Rumus ABC
= (XA + 2,7)(XA 0,58)
-0,121
=
=1
P3
=0
-0,58A
2,7B
=1
A = -0,305
B = 0,305
Sehingga persamaan menjadi
-0,121*
+ = k1 . t
-0,121*
-+
k1 . t
k1 . t
0,037 [
0,037[
] = k1 . t
0,037 [ ln (
) = k1 . t
0,037 [ ln (
) = k1 . t
0,037ln *
t(x)
k1. t
+ = k1 . t y = mx
Xa
X2
Xy
0,189
0,017
11
0,35
0,039
0,43
121
22
0,44
0,058
1,276
484
33
0,50
0,08
2,64
1089
P3
44
0,58
K=
k2 =
= 2,88.10-4 mol/menit
=
= 8,1.10-4 mol/menit
1936
You might also like
- Instructor's Manual to Accompany CALCULUS WITH ANALYTIC GEOMETRYFrom EverandInstructor's Manual to Accompany CALCULUS WITH ANALYTIC GEOMETRYNo ratings yet
- Solution Manual for an Introduction to Equilibrium ThermodynamicsFrom EverandSolution Manual for an Introduction to Equilibrium ThermodynamicsNo ratings yet
- Concrete Column Design Based On ACI 318-11: Input Data & Design SummaryDocument3 pagesConcrete Column Design Based On ACI 318-11: Input Data & Design Summaryjaimeleoxxx0% (1)
- ReservoirEngineering Solution ManualDocument92 pagesReservoirEngineering Solution ManualHeba100% (3)
- Trigonometric Ratios to Transformations (Trigonometry) Mathematics E-Book For Public ExamsFrom EverandTrigonometric Ratios to Transformations (Trigonometry) Mathematics E-Book For Public ExamsRating: 5 out of 5 stars5/5 (1)
- Gas Condensate ReservoirDocument33 pagesGas Condensate ReservoirMohammad Iqbal Mahamad Amir100% (3)
- Fundamentals of Momentum Heat and Mass Transfer 6th Edition Welty Solutions ManualDocument34 pagesFundamentals of Momentum Heat and Mass Transfer 6th Edition Welty Solutions ManualGregoryShafferdwrbo89% (19)
- Applied Homework 1#Document7 pagesApplied Homework 1#Roba SaidNo ratings yet
- Uts TermodinamikaDocument13 pagesUts TermodinamikaMuhammad Bachtiar100% (2)
- Mathcad - Fundatii IzolateDocument32 pagesMathcad - Fundatii IzolateMaris NicuNo ratings yet
- Lembar Perhitungan HMJDocument7 pagesLembar Perhitungan HMJAhmad AsfahaniNo ratings yet
- Lembar Perhitungan EsterDocument7 pagesLembar Perhitungan EsterpanjigomisNo ratings yet
- Lembar Perhitungan ReagenDocument10 pagesLembar Perhitungan ReagenwakwakNo ratings yet
- Lembar Perhitungan PatiDocument4 pagesLembar Perhitungan PatiestiononugrohoNo ratings yet
- CH 17Document54 pagesCH 17firebot4No ratings yet
- Chapter11 ADocument33 pagesChapter11 ANic BlandoNo ratings yet
- Perhitungan Volume Molar ParsialDocument3 pagesPerhitungan Volume Molar ParsialAntonius Rendi SupriantoNo ratings yet
- Columnas EsquinerasDocument6 pagesColumnas EsquinerasJomayraMayteVillaltaNo ratings yet
- Antoine Equation ConstantsDocument5 pagesAntoine Equation ConstantsMika PelagioNo ratings yet
- Exercise ExcelDocument7 pagesExercise Excelhelennguyen242004No ratings yet
- To: From: Date: Subject: CC:: ECH 158ADocument4 pagesTo: From: Date: Subject: CC:: ECH 158ATrường Tùng LýNo ratings yet
- ECH 158A Ex2Document4 pagesECH 158A Ex2Trường Tùng LýNo ratings yet
- Lembar Perhitungan 1. Perhitungan Reagen: Asam AsetatDocument8 pagesLembar Perhitungan 1. Perhitungan Reagen: Asam AsetatRizky Ardias DarmawanNo ratings yet
- Chapter5 ADocument21 pagesChapter5 ANic BlandoNo ratings yet
- Chapter4 ADocument47 pagesChapter4 ANic BlandoNo ratings yet
- Data Pengamatan Dan Perhitungan ARISDocument6 pagesData Pengamatan Dan Perhitungan ARISAris RiantoNo ratings yet
- Exercise Solutions EX8.1: V V IR IV V VDocument8 pagesExercise Solutions EX8.1: V V IR IV V Vapi-3741518No ratings yet
- MTPPT4 - Design of Isolated FootingDocument19 pagesMTPPT4 - Design of Isolated FootingMineski Prince GarmaNo ratings yet
- ENPE 523 Assignment - 2 SolutionDocument14 pagesENPE 523 Assignment - 2 Solutiontiger8878100% (1)
- Topo - Calculos # 3Document10 pagesTopo - Calculos # 3RNo ratings yet
- Calculos Analitica Y DiscusionDocument7 pagesCalculos Analitica Y DiscusionKEVIN ADRIAN HERNANDEZ BOCANEGRANo ratings yet
- Simple Mixtures: Answers To Discussion QuestionsDocument18 pagesSimple Mixtures: Answers To Discussion QuestionsLudimila Araújo LodiNo ratings yet
- Lemper RevisedDocument7 pagesLemper RevisedTitin Setya MuktiNo ratings yet
- Caracteristici Geometrice BruteDocument33 pagesCaracteristici Geometrice BruteTudose AndreiNo ratings yet
- ØtinputDocument2 pagesØtinputRizky RamadhanNo ratings yet
- Hasil Pengamatan Data Nilai Absorbansi Tabel 1. Nilai Absorbansi PhabsiabsiiDocument6 pagesHasil Pengamatan Data Nilai Absorbansi Tabel 1. Nilai Absorbansi PhabsiabsiiAnisaNo ratings yet
- Ch15p PDFDocument11 pagesCh15p PDFLuis AntonioNo ratings yet
- MA2177 Ex6 SolDocument3 pagesMA2177 Ex6 Solkyle cheungNo ratings yet
- 1252 EquationSheetDocument2 pages1252 EquationSheetAllie GleditschNo ratings yet
- Lampiran CompleteDocument23 pagesLampiran CompleteEllsworthNo ratings yet
- Perhitungan P6 YuliDocument6 pagesPerhitungan P6 YuliyuliraraNo ratings yet
- Tugas Menggunakan Data Sondir PT Panca Tunggal PersadaDocument9 pagesTugas Menggunakan Data Sondir PT Panca Tunggal PersadaBoy Rizky SilalahiNo ratings yet
- For Water: .. .. ..: C KJ Kmol - K 2.413 KJ KG - K KJ KG - KDocument10 pagesFor Water: .. .. ..: C KJ Kmol - K 2.413 KJ KG - K KJ KG - KanmarNo ratings yet
- Che 254 (Midsem)Document6 pagesChe 254 (Midsem)obumaradonaNo ratings yet
- Energy Balance Around ReactorDocument9 pagesEnergy Balance Around ReactorRishi RajNo ratings yet
- Perhitungan Volume Molar ParsialDocument3 pagesPerhitungan Volume Molar ParsialAntonius Rendi SupriantoNo ratings yet
- Semana4 RM CpuDocument8 pagesSemana4 RM CpuJENRRY JAMER RODRIGO VITONNo ratings yet
- Vessel DesignDocument15 pagesVessel DesignSurya Kanta SenNo ratings yet
- Distillation: Abdulsalam Sufyaan Hamed Mustafa Jumaah Abbas Zahraa ShehabDocument17 pagesDistillation: Abdulsalam Sufyaan Hamed Mustafa Jumaah Abbas Zahraa ShehabAbdulsalam SNo ratings yet
- Energy Balance FINAL SUBMITDocument34 pagesEnergy Balance FINAL SUBMITRelaxing MusicNo ratings yet
- Student Ski Rad 2Document27 pagesStudent Ski Rad 2dino_051No ratings yet
- ElektroDocument3 pagesElektroaprilia kurnia putriNo ratings yet
- Lembar Perhitungan Variabel 1: Asam Asetat A ADocument10 pagesLembar Perhitungan Variabel 1: Asam Asetat A AMuhammad FarhanNo ratings yet
- Lembar Perhitungan ReagenDocument16 pagesLembar Perhitungan ReagenZoe SinulinggaNo ratings yet
- 10 T Blender Crack AnalysismodDocument8 pages10 T Blender Crack AnalysismodJayesh JayanNo ratings yet
- Let The Required Sales Quantity Be X: Quantitative MethodsDocument8 pagesLet The Required Sales Quantity Be X: Quantitative MethodsUmer RehmanNo ratings yet
- Laboratory Exercises in Astronomy: Solutions and AnswersFrom EverandLaboratory Exercises in Astronomy: Solutions and AnswersNo ratings yet
- Ten-Decimal Tables of the Logarithms of Complex Numbers and for the Transformation from Cartesian to Polar Coordinates: Volume 33 in Mathematical Tables SeriesFrom EverandTen-Decimal Tables of the Logarithms of Complex Numbers and for the Transformation from Cartesian to Polar Coordinates: Volume 33 in Mathematical Tables SeriesNo ratings yet