Professional Documents
Culture Documents
BriggsHaldane PDF
BriggsHaldane PDF
Uploaded by
AlinOriginal Title
Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
BriggsHaldane PDF
BriggsHaldane PDF
Uploaded by
AlinCopyright:
Available Formats
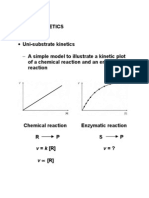
The Quasi-Steady State Approach to enzyme kinetics
(Briggs-Haldane)
kf1-> kf2->
S+E<---->SE---->P+E
<-kb1 <-kb2
The differential equations for a saturable reversible
enzyme conversion process are given as
d [ S ]/ dt=−kf1∗[ S ][ E ]kb1∗[ SE ]
d [ SE ]/ dt=kf1∗[ S ][ E ]−kf2kb1∗[ SE ]kb2∗[ P ][ E ].
There are two conservation equations"
[S TOT ]=[ S ][SE ][ P ]
[ E TOT ]=[ E ][ SE ] .
The usual assumptions given in texts are that kb2=0, and
kf1,kb1 >> kf2 so that [SE] and [S] are in equilibrium
(Michaelis-Menten) or that [SE] is in a quasi-steady state
(Briggs-Haldane). The result in either case is that the
rate of change of the substrate enzyme complex, d[SE]/dt,
is very small (assumed zero) and we can set
kf1∗[ S ]∗[ E ]−kf2kb1∗[ SE ]=0.
Solve for [E] to get
[ E ]=kf2kb1∗[ SE ]/kf1∗[S ]
We can write
[ E ]=kf2kb1/ kf1∗[SE ]/[ S ]
Km=kf2kb1/ kf1.
Therefore,
[ E ]=Km [ SE ]/[ S ].
Substitute this expression into the second conservation
equation yielding
[ E TOT ]=Km [ SE ]/[ S ][ SE ].
Solving this equation for [SE] yields
[ SE ]=[ E TOT ][ S ]/ Km[ S ].
Substituting the expression for [E] into the equation for
d[S]/dt yields
d [ S ]/ dt=−kf1∗[ S ][ E ]kb1∗[ SE ]
d [ S ]/ dt=−kf1∗[ S ]∗Km∗[ SE ]/[ S ]kb1∗[ SE ]
to give
d [ S ]/ dt=−kf1∗Kmkb1∗[ SE ]=−kf2∗[ SE ].
Substituting for [SE] gives
d [ S ]/ dt=−kf2∗[ E TOT ][ S ]/ Km[ S ].
Letting
kf2∗[ E TOT ]=Vmax
allows us to write the familiar Michaelis-Menten (Briggs-
Haldane) form,
d [ S ]/ dt=−Vmax [ S ]/ Km[ S ].
In 1925, G. E. Briggs and J.B.S. Haldane derived a new interpretation of the enzyme
kinetics law described by Victor Henri in 1903, different from the 1913 Michaelis
Menten equation. Leonor Michaelis and Maud Menten assumed that enzyme (catalyst)
and substrate (reactant) are in fast equilibrium with their complex, which then dissociates
to yield product and free enzyme. The BriggsHaldane equation was of the same
algebraic form, but their derivation is based on the quasi steady state approximation, that
is the concentration(s) of intermediate complex(es) do(es) not change. As a result, the
microscopic meaning of the "Michaelis Constant" (Km) is different. Although commonly
referring it as MichaelisMenten kinetics, most of the current models actually use the
BriggsHaldane derivation.
You might also like
- Foot Atomic Solutions by Zhao, C.Document32 pagesFoot Atomic Solutions by Zhao, C.Jacob Francis94% (16)
- Sanyo Denki TapeReader Model 2702 Instruction ManualDocument18 pagesSanyo Denki TapeReader Model 2702 Instruction ManualcoronaqcNo ratings yet
- 4Document15 pages4phamdienthongNo ratings yet
- WordPress SEO ChecklistDocument7 pagesWordPress SEO ChecklistTLC for Coaches100% (2)
- Lecture 10: Enzyme Kinetics: Reading: Chapter 6 Pp. 202-213Document21 pagesLecture 10: Enzyme Kinetics: Reading: Chapter 6 Pp. 202-213SriArthiNo ratings yet
- Michaelis-Menten Kinetics and Briggs-Haldane Kinetics - ChemwikiDocument15 pagesMichaelis-Menten Kinetics and Briggs-Haldane Kinetics - ChemwikiEmmanuel Alexander PilapilNo ratings yet
- Consecutive Reactions, Steady State Approximation: and Enzyme Catalysed ReactionsDocument19 pagesConsecutive Reactions, Steady State Approximation: and Enzyme Catalysed ReactionsPreeti YadavNo ratings yet
- EnzymeDocument39 pagesEnzymeBhaski MadhappanNo ratings yet
- MIT Lecture 6 5.07 Biochemistry LectureDocument16 pagesMIT Lecture 6 5.07 Biochemistry LectureakiridoNo ratings yet
- EnzymeKinetics PDFDocument21 pagesEnzymeKinetics PDFaathiraNo ratings yet
- LSM1101 Enzyme2Document40 pagesLSM1101 Enzyme2givena2ndchanceNo ratings yet
- MRQ - #3 Enzyme Kinetics - 2015Document44 pagesMRQ - #3 Enzyme Kinetics - 2015yanuar setiawanNo ratings yet
- QSSA SegelDocument6 pagesQSSA SegelInga EskitziNo ratings yet
- Lection 5 (Eng) PDFDocument9 pagesLection 5 (Eng) PDFa320neoNo ratings yet
- Enzyme KineticsDocument18 pagesEnzyme KineticsRoyal MindNo ratings yet
- Michaelis Menten Equation SummaryDocument7 pagesMichaelis Menten Equation SummaryBüşra GültekinNo ratings yet
- 3 Enzyme KineticsDocument22 pages3 Enzyme KineticsLee Ping Shin100% (1)
- Enzyme KineticsDocument65 pagesEnzyme KineticsSulabh JainNo ratings yet
- 2.1 Michaelis Menten EquationDocument8 pages2.1 Michaelis Menten EquationKishoreNo ratings yet
- Abramson Restricted Combinatioons and CompositionsDocument14 pagesAbramson Restricted Combinatioons and CompositionsCeiba GrooveNo ratings yet
- Enzyme KineticsDocument2 pagesEnzyme KineticsYilun DuNo ratings yet
- MIT5 - 111F14 - Lecture34 CompressedDocument6 pagesMIT5 - 111F14 - Lecture34 CompressedBishal kafleNo ratings yet
- Lecture 14-Estimation of MM Kinetic ParametersDocument17 pagesLecture 14-Estimation of MM Kinetic ParametersAmogha G C 1SI19CH002No ratings yet
- Viscoelasticity 05 LaplaceDocument7 pagesViscoelasticity 05 LaplacealirezakhadirNo ratings yet
- Kjdfks FDocument11 pagesKjdfks FTee BeeNo ratings yet
- Phy Pharm AssignmentDocument4 pagesPhy Pharm AssignmentNazura AliasNo ratings yet
- Binding KineticsDocument8 pagesBinding Kineticskyliever100% (1)
- Enzyme KineticsDocument26 pagesEnzyme KineticsLyra LasangreNo ratings yet
- PSoln 2Document4 pagesPSoln 2anon020202No ratings yet
- Enzymekinetics 160123162523Document36 pagesEnzymekinetics 160123162523Rxjviie BodaNo ratings yet
- Invlap, KDocument10 pagesInvlap, KMohamed AdelNo ratings yet
- Lecture 14: Enzyme Kinetics: Biochemistry I Fall Term, 2004Document6 pagesLecture 14: Enzyme Kinetics: Biochemistry I Fall Term, 2004Louis FortunatoNo ratings yet
- Enzymes: 1. Catalysis, Co-Enzymes, Kinetics 2. Catalytic Mechanisms, Enzyme RegulationDocument40 pagesEnzymes: 1. Catalysis, Co-Enzymes, Kinetics 2. Catalytic Mechanisms, Enzyme Regulationbarrj2No ratings yet
- Enzyme M&LDocument6 pagesEnzyme M&LNethra Sasikumar100% (1)
- Metabolic Biochemistry: BIBC 102Document38 pagesMetabolic Biochemistry: BIBC 102FlowerNo ratings yet
- Enzyme KineticsDocument26 pagesEnzyme KineticsLyra LasangreNo ratings yet
- EnzymeKinetics by P.C. Misra Professor, Department of Biochemistry Lucknow University, Lucknow-226 007Document21 pagesEnzymeKinetics by P.C. Misra Professor, Department of Biochemistry Lucknow University, Lucknow-226 007Dr. SHIVA AITHALNo ratings yet
- Chapter 1 Single-Enzyme Kinetics: Molecular Cell BiologyDocument7 pagesChapter 1 Single-Enzyme Kinetics: Molecular Cell BiologySparrowGospleGilbertNo ratings yet
- Lecture Notes Enzyme 2 Enzyme Kinetics WebDocument29 pagesLecture Notes Enzyme 2 Enzyme Kinetics WebAldren RebaLdeNo ratings yet
- 2010 Low Temperature Plasma Modeling & SimulationDocument100 pages2010 Low Temperature Plasma Modeling & SimulationpresledovatelNo ratings yet
- Units ConservationDocument5 pagesUnits ConservationMohamed AamerNo ratings yet
- Lecture 9 Introductory Kinetics PDFDocument78 pagesLecture 9 Introductory Kinetics PDFskrim240No ratings yet
- Chapter 5 (Part 2) : Enzyme KineticsDocument20 pagesChapter 5 (Part 2) : Enzyme KineticsTrisuciati SyahwardiniNo ratings yet
- However, in the absence of B, v (k (t') ) = v (k) time-independent and T g (k) = g (k) + (E (k) (-∂f/∂E) v (k) - (- e - μ - T (E (k) - μ) /T)Document7 pagesHowever, in the absence of B, v (k (t') ) = v (k) time-independent and T g (k) = g (k) + (E (k) (-∂f/∂E) v (k) - (- e - μ - T (E (k) - μ) /T)Krishna B ma21m015No ratings yet
- The Michaelis - Menten Equation: Enzyme: Substrate Dissociation Constant. at EquilibriumDocument6 pagesThe Michaelis - Menten Equation: Enzyme: Substrate Dissociation Constant. at EquilibriumSoham TrivediNo ratings yet
- Biocatalysis Chen 5751: OutlineDocument14 pagesBiocatalysis Chen 5751: OutlineHussein SalihNo ratings yet
- Chapter 2Document18 pagesChapter 2Lucy BrownNo ratings yet
- Analysis of The Michaelis-Menten Mechanism in An Immobilised Enzyme ReactorDocument12 pagesAnalysis of The Michaelis-Menten Mechanism in An Immobilised Enzyme ReactorMarín HernándezNo ratings yet
- Thermofluids Data BookDocument30 pagesThermofluids Data BookAjmal SalamNo ratings yet
- On The Numerical Computation of Enzyme Kinetic Parameters: Biomath CommunicationsDocument24 pagesOn The Numerical Computation of Enzyme Kinetic Parameters: Biomath CommunicationsNatalia OchoaNo ratings yet
- Minkowski-Space Correlators in Ads/Cft Correspondence: Recipe and ApplicationsDocument28 pagesMinkowski-Space Correlators in Ads/Cft Correspondence: Recipe and ApplicationsdamtsonNo ratings yet
- Ch12 1 Transition Metals Reaction MechanismsDocument22 pagesCh12 1 Transition Metals Reaction MechanismsGuru P MNo ratings yet
- PHZ6426: Fall 2013 Problem Set # 6: Electron Transport. Phonons. Due Monday, 12/02 at The Time of The Class Instructor: D. L. Maslov Maslov@phys - Ufl.edu 392-0513 Rm. 2114 Office Hours: TR 3 pm-4 PMDocument9 pagesPHZ6426: Fall 2013 Problem Set # 6: Electron Transport. Phonons. Due Monday, 12/02 at The Time of The Class Instructor: D. L. Maslov Maslov@phys - Ufl.edu 392-0513 Rm. 2114 Office Hours: TR 3 pm-4 PMMkt SchrbrNo ratings yet
- Hall MobilityDocument27 pagesHall MobilityTushar Dhabal DasNo ratings yet
- Additional Electrochemistry Slides HardacreDocument18 pagesAdditional Electrochemistry Slides Hardacretongshuzhang882No ratings yet
- Enzyme KineticsDocument4 pagesEnzyme KineticsvnNo ratings yet
- Deriving The Michaelis MENTEN EQUTIONDocument5 pagesDeriving The Michaelis MENTEN EQUTIONManish GaurNo ratings yet
- Solution For Chapter 22Document5 pagesSolution For Chapter 22Sveti JeronimNo ratings yet
- 5.60 Thermodynamics & Kinetics: Mit OpencoursewareDocument6 pages5.60 Thermodynamics & Kinetics: Mit OpencoursewarecaptainhassNo ratings yet
- Non-Relativistic Scattering of A Spin-1/2 Particle Off A Self-Dual MonopoleDocument8 pagesNon-Relativistic Scattering of A Spin-1/2 Particle Off A Self-Dual MonopoleBayer MitrovicNo ratings yet
- Logical progression of twelve double binary tables of physical-mathematical elements correlated with scientific-philosophical as well as metaphysical key concepts evidencing the dually four-dimensional basic structure of the universeFrom EverandLogical progression of twelve double binary tables of physical-mathematical elements correlated with scientific-philosophical as well as metaphysical key concepts evidencing the dually four-dimensional basic structure of the universeNo ratings yet
- ITA - FSC.Cosmoprof.2015 Final2 PDFDocument108 pagesITA - FSC.Cosmoprof.2015 Final2 PDFSri Dwi AryaniNo ratings yet
- Indonesia Cosmetic Products - GBGDocument4 pagesIndonesia Cosmetic Products - GBGSri Dwi AryaniNo ratings yet
- Personal Selling and Sales ManagementDocument26 pagesPersonal Selling and Sales ManagementSri Dwi AryaniNo ratings yet
- Phenomenological Model of #Ltration Processes: 1. Cake Formation and ExpressionDocument17 pagesPhenomenological Model of #Ltration Processes: 1. Cake Formation and ExpressionSri Dwi AryaniNo ratings yet
- Charge Cycle: See AlsoDocument3 pagesCharge Cycle: See AlsoSri Dwi AryaniNo ratings yet
- P130 Questions Ch08Document5 pagesP130 Questions Ch08Sri Dwi AryaniNo ratings yet
- Definition and Purposes of ExplanationDocument1 pageDefinition and Purposes of ExplanationSri Dwi AryaniNo ratings yet
- SBARDocument1 pageSBARJeffrey CollinsNo ratings yet
- WaterfloodingDocument29 pagesWaterfloodingAdel Al-Enazi100% (1)
- σcosθ ρgd σ = surface tension in N/m θ = wetting angle, 0 for water d = diameter of tube in m ρ = density of fluidDocument1 pageσcosθ ρgd σ = surface tension in N/m θ = wetting angle, 0 for water d = diameter of tube in m ρ = density of fluidJustine Ejay MoscosaNo ratings yet
- Accomplishment Clubs ProjectDocument3 pagesAccomplishment Clubs Projectshai24No ratings yet
- IELTS Listening Part 1Document17 pagesIELTS Listening Part 1GiangNo ratings yet
- User Guide IRCTCDocument23 pagesUser Guide IRCTCManoj KumarNo ratings yet
- Powercfg Command-Line OptionsDocument15 pagesPowercfg Command-Line OptionsNiko PaNo ratings yet
- Solomon y Corbit - 1978 - An Opponent-Process Theory of MotivationDocument14 pagesSolomon y Corbit - 1978 - An Opponent-Process Theory of Motivationmario_garcés_4No ratings yet
- The Weak Interaction: Project PHYSNET Physics Bldg. Michigan State University East Lansing, MIDocument8 pagesThe Weak Interaction: Project PHYSNET Physics Bldg. Michigan State University East Lansing, MIEpic WinNo ratings yet
- The 14 Tablets of Lord EnkiDocument10 pagesThe 14 Tablets of Lord EnkiChirita GheorgheNo ratings yet
- Sample WLHP BichronousDocument7 pagesSample WLHP BichronousJoanna Joy MercadoNo ratings yet
- TOK Essay - "Context Is All"Document3 pagesTOK Essay - "Context Is All"VivarisNo ratings yet
- Ab PLC EpksDocument82 pagesAb PLC EpksBouazzaNo ratings yet
- Thesis Synopsis For A Design SchoolDocument5 pagesThesis Synopsis For A Design SchoolAll about NBJNo ratings yet
- 5 HotsDocument16 pages5 HotsJeffrey Ramos100% (1)
- Almighty Military Format Revised by Welbeck (Male)Document11 pagesAlmighty Military Format Revised by Welbeck (Male)jadenroy442No ratings yet
- Fiber Cable Preparation, Splicing, and Termination InstructionsDocument8 pagesFiber Cable Preparation, Splicing, and Termination InstructionsHunaAdjaganNo ratings yet
- Cadence Layout TutorialDocument12 pagesCadence Layout TutorialpchengoesNo ratings yet
- Lesson Plan in Practical Research 1Document4 pagesLesson Plan in Practical Research 1Dominica Recoy Pontica - DiololaNo ratings yet
- Mechanisms in Modern Engineering V2 Pt2Document448 pagesMechanisms in Modern Engineering V2 Pt2drpisson100% (1)
- C Guide ExamDocument724 pagesC Guide ExamAlan MirandaNo ratings yet
- Hypothesis Tests About The Mean and ProportionDocument109 pagesHypothesis Tests About The Mean and Proportion03435013877100% (1)
- Summary Education System in SpainDocument6 pagesSummary Education System in Spainsoniadedios100% (1)
- Competitor Comparison Table - v2Document2 pagesCompetitor Comparison Table - v2Cherry EstradaNo ratings yet
- Marrin - CIA Kent School and University-2000Document3 pagesMarrin - CIA Kent School and University-2000Stephen MarrinNo ratings yet
- Killzone Rules v3Document25 pagesKillzone Rules v3dracomaxisNo ratings yet
- Research On Web BrowsersDocument60 pagesResearch On Web BrowserstravpreneurNo ratings yet
- MultiFlexETH UserManualDocument218 pagesMultiFlexETH UserManualrocketmenchNo ratings yet