Professional Documents
Culture Documents
Data Sheet - Quiz 1
Uploaded by
Dylan HamOriginal Description:
Original Title
Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
Data Sheet - Quiz 1
Uploaded by
Dylan HamCopyright:
Available Formats
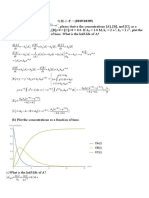
Data Sheet
∆𝐺 = ∆𝐻 − 𝑇∆𝑆
𝑙𝑛[𝐴] = −𝑎𝑘𝑡 + 𝑙𝑛[𝐴0 ]
1 1
akt
A Ao
PV=nRT
Tb Kbim; T f K f im
aA + bB → cC + dD
H o cH of C d H of D ;
aH of A bH of B
𝑃 ∆𝐻𝑣𝑎𝑝 1 1
𝑙𝑛 ( 2) = − (𝑇 − 𝑇 )
𝑃 1 𝑅 2 1
𝑦 2 = 2𝑘𝑝 𝑡
K H o 1 1
ln 2
K1 R T2 T1
∆𝐸 = 𝑞 − 𝑤
𝑑 = √6𝐷𝑡
𝑉
𝑤 = ∫𝑉 2 𝑃𝑑𝑉
1
H E ( PV ) ;
0 C 273 K
R = 8.314 J K-1 mol-1
1 atm. = 101,350 Pa
Atomic masses H = 1 g/mol, O = 16 g/mol, C = 12 g/mol, N = 14 g/mol
You might also like
- Solution Manual for an Introduction to Equilibrium ThermodynamicsFrom EverandSolution Manual for an Introduction to Equilibrium ThermodynamicsNo ratings yet
- Tugas - 1 - PSPK Firstiando YudaDocument10 pagesTugas - 1 - PSPK Firstiando YudaBrayonoFlo100% (1)
- PCTechAuthority May2018 PDFDocument116 pagesPCTechAuthority May2018 PDFDylan HamNo ratings yet
- Kerson Huang - Introduction To Statistical PhysicsDocument440 pagesKerson Huang - Introduction To Statistical PhysicsMaher Al oboodyNo ratings yet
- 100 Years of Bowral BricksDocument143 pages100 Years of Bowral BricksDylan Ham100% (1)
- Chemistry Cheat Sheet-1Document1 pageChemistry Cheat Sheet-1r3birthvalNo ratings yet
- Exam3 EquationsheetDocument1 pageExam3 EquationsheetNguyễn Minh AnhNo ratings yet
- General Chemistry II Equation SheetDocument2 pagesGeneral Chemistry II Equation Sheetadilb24032000No ratings yet
- Chemistry 101/3 Formula SheetDocument5 pagesChemistry 101/3 Formula SheetMathew WebsterNo ratings yet
- Second Semester Stage 2Document99 pagesSecond Semester Stage 2Watch DogsNo ratings yet
- 5.60 Thermodynamics & Kinetics: Mit OpencoursewareDocument7 pages5.60 Thermodynamics & Kinetics: Mit OpencoursewarecaptainhassNo ratings yet
- Elektronika KesetimbanganDocument28 pagesElektronika KesetimbanganPutri Nur AngelinaNo ratings yet
- Lecture 38Document5 pagesLecture 38Hagere EthiopiaNo ratings yet
- Chemical Equilibrium: 2.1. Some DefinitionsDocument24 pagesChemical Equilibrium: 2.1. Some DefinitionsNguyễn Quốc HưngNo ratings yet
- Boardgamedisplay14671 140524165124 Phpapp01Document1 pageBoardgamedisplay14671 140524165124 Phpapp01KitkatNo ratings yet
- Chapter 1 Solutions DetailedDocument30 pagesChapter 1 Solutions DetailedYeonjae JeongNo ratings yet
- Chap. 8 Reaction Kinetics: (1) Definition of Reaction RateDocument6 pagesChap. 8 Reaction Kinetics: (1) Definition of Reaction RateArasuNo ratings yet
- Derivation of Force and Torque On Elliptic Molecules Due To EES PotentialDocument3 pagesDerivation of Force and Torque On Elliptic Molecules Due To EES PotentialabdoNo ratings yet
- Information, Constants, Formulae Information: Theoretical PartDocument10 pagesInformation, Constants, Formulae Information: Theoretical PartATHAYYA FORTUNANo ratings yet
- Final Exam Equations, Constants and TablesDocument4 pagesFinal Exam Equations, Constants and Tablessammy alanNo ratings yet
- University of Technology, JamaicaDocument7 pagesUniversity of Technology, JamaicaAthinaNo ratings yet
- Mass Transfer Theories: T y y T y yDocument5 pagesMass Transfer Theories: T y y T y ynermeen ahmedNo ratings yet
- Jee Chem 1 Eng 26 03Document4 pagesJee Chem 1 Eng 26 03vikasgnsharma100% (1)
- C Sol Ch-06 EquilibriumDocument10 pagesC Sol Ch-06 Equilibriummysoftinfo.incNo ratings yet
- HYDRO 1 - Module 2.4 Bernoulli's Energy TheoremDocument8 pagesHYDRO 1 - Module 2.4 Bernoulli's Energy TheoremPhilip SabadiNo ratings yet
- Chapter 9. Kinetics: Rates of Chemical ReactionsDocument11 pagesChapter 9. Kinetics: Rates of Chemical ReactionsAtiya SultanaNo ratings yet
- Balances Reactores12Document4 pagesBalances Reactores12Yoe CavanaNo ratings yet
- Lecture 9: Multiple Reactions in SeriesDocument13 pagesLecture 9: Multiple Reactions in SeriesmlermantovNo ratings yet
- General Chemistry Lecturer-2Document34 pagesGeneral Chemistry Lecturer-2Bảo Long Trần LêNo ratings yet
- Equilibri Quimic HW12 SolDocument8 pagesEquilibri Quimic HW12 SolmarzinusNo ratings yet
- 9-Chapter 8-Chemical Reaction Equilibria-27March Online Class-STDNDocument15 pages9-Chapter 8-Chemical Reaction Equilibria-27March Online Class-STDNMahamed HusseinNo ratings yet
- CM150-2 Exercise 3 (MODULE 3)Document6 pagesCM150-2 Exercise 3 (MODULE 3)owl lawletNo ratings yet
- Lec 5chemical EquilibriumDocument25 pagesLec 5chemical Equilibriuma.alsayed6805No ratings yet
- Lecture Guide 4 Conduction Through Plain Spherical WallDocument5 pagesLecture Guide 4 Conduction Through Plain Spherical WallCllyan ReyesNo ratings yet
- Useful Formulas For ChemistryDocument1 pageUseful Formulas For ChemistryJemarey de RamaNo ratings yet
- 4.6 Real solution: activity of solute and solvent: γ γ By R.L. The activity factorDocument18 pages4.6 Real solution: activity of solute and solvent: γ γ By R.L. The activity factordhika mtNo ratings yet
- Notes 3 (Kinetic Examples) 12Document12 pagesNotes 3 (Kinetic Examples) 12Sharon FonsecaNo ratings yet
- Widrow-Hoff Learning: (LMS Algorithm)Document26 pagesWidrow-Hoff Learning: (LMS Algorithm)Alee LópezNo ratings yet
- Widrow-Hoff Learning: (LMS Algorithm)Document26 pagesWidrow-Hoff Learning: (LMS Algorithm)Alee LópezNo ratings yet
- Chemical Equilibrium Part III-1 PDFDocument6 pagesChemical Equilibrium Part III-1 PDFEstivenson Vasquez CNo ratings yet
- Temperature Dependence: Change in G - (Change in T) X SDocument5 pagesTemperature Dependence: Change in G - (Change in T) X SShahday BayanNo ratings yet
- Ioniz PDFDocument3 pagesIoniz PDFSyed Raza Ali RazaNo ratings yet
- 507 Lecture 22Document21 pages507 Lecture 22Ali AzanNo ratings yet
- Equilibrium Electrochemistry: Physical Chemistry Division Departement of ChemistryDocument28 pagesEquilibrium Electrochemistry: Physical Chemistry Division Departement of ChemistryPutri Nur AngelinaNo ratings yet
- 108 Quiz1Document1 page108 Quiz1沈智恩No ratings yet
- Kinetics Probset (LE3)Document1 pageKinetics Probset (LE3)Jewls HatudNo ratings yet
- Feb2020-Chuong4-Toc Do Va Co Che Phan Ung Hoa HocDocument36 pagesFeb2020-Chuong4-Toc Do Va Co Che Phan Ung Hoa HocHồng NgọcNo ratings yet
- Formula Sheet NumericalDocument3 pagesFormula Sheet NumericalZain BhinderNo ratings yet
- OriginalDocument4 pagesOriginalyigaf49105No ratings yet
- Chapter 2Document50 pagesChapter 2NgânNo ratings yet
- Chapter 3Document51 pagesChapter 3Ngọc Hà NguyễnNo ratings yet
- Chemical KineticsDocument20 pagesChemical Kineticsvijaylakshmi0727No ratings yet
- Jee-Neet - D30-Nov-2022 AnswerDocument13 pagesJee-Neet - D30-Nov-2022 AnswerDhruvNo ratings yet
- CHEM-UA 652: Thermodynamics and Kinetics: Notes For Lecture 23Document5 pagesCHEM-UA 652: Thermodynamics and Kinetics: Notes For Lecture 23Nur Faiizah AfNo ratings yet
- Combustion of Methanol-Air MixtureDocument3 pagesCombustion of Methanol-Air MixturemrpcuNo ratings yet
- HN OH: Multiple Choice QuestionsDocument5 pagesHN OH: Multiple Choice QuestionsMahesh Kumar KumawatNo ratings yet
- Chemical Kinetics Short NotesDocument4 pagesChemical Kinetics Short NotesSanket Patil100% (1)
- Chapter 6Document8 pagesChapter 6StefanPerendijaNo ratings yet
- Steady StateDocument30 pagesSteady Statesus023No ratings yet
- 5.60 Thermodynamics & Kinetics: Mit OpencoursewareDocument7 pages5.60 Thermodynamics & Kinetics: Mit OpencoursewarecaptainhassNo ratings yet
- Lecture 13Document19 pagesLecture 13hema maliniNo ratings yet
- Critical Evaluation of Some Equilibrium Constants Involving Organophosphorus ExtractantsFrom EverandCritical Evaluation of Some Equilibrium Constants Involving Organophosphorus ExtractantsNo ratings yet
- DCP 2014 Revision 24 Part 3 Development Within Residential Zones Adopted 9 March 20202Document55 pagesDCP 2014 Revision 24 Part 3 Development Within Residential Zones Adopted 9 March 20202Dylan HamNo ratings yet
- Chapter2 - Thermodynamics - Why Things ChangeDocument25 pagesChapter2 - Thermodynamics - Why Things ChangeDylan HamNo ratings yet