Professional Documents
Culture Documents
1.2 Question Database
1.2 Question Database
Uploaded by
123456Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
1.2 Question Database
1.2 Question Database
Uploaded by
123456Copyright:
Available Formats
PRACTICE EXAMINATION QUESTIONS FOR 1.
2 AMOUNT OF SUBSTANCE
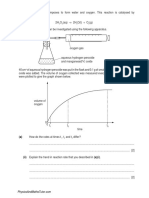
1. Nitroglycerine, C3H5N3O9, is an explosive which, on detonation, decomposes rapidly to form a
large number of gaseous molecules. The equation for this decomposition is given below.
4C3H5N3O9(l) → 12CO2(g) + 10H2O(g) + 6N2(g) + O2(g)
(a) A sample of nitroglycerine was detonated and produced 0.350 g of oxygen gas.
(i) State what is meant by the term one mole of molecules.
...........................................................................................................................
(ii) Calculate the number of moles of oxygen gas produced in this reaction, and hence
deduce the total number of moles of gas formed.
Moles of oxygen gas .........................................................................................
Total moles of gas ............................................................................................
...........................................................................................................................
...........................................................................................................................
(iii) Calculate the number of moles, and the mass, of nitroglycerine detonated.
Moles of nitroglycerine ....................................................................................
...........................................................................................................................
Mass of nitroglycerine ......................................................................................
...........................................................................................................................
...........................................................................................................................
(7)
Mill Hill High School 1
(b) A second sample of nitroglycerine was placed in a strong sealed container and detonated.
–3 3
The volume of this container was 1.00 × 10 m . The resulting decomposition produced
a total of 0.873 mol of gaseous products at a temperature of 1100 K.
State the ideal gas equation and use it to calculate the pressure in the container after
detonation.
–1 –1
(The gas constant R = 8.31 J K mol )
Ideal gas equation .......................................................................................................
Pressure ......................................................................................................................
.....................................................................................................................................
.....................................................................................................................................
.....................................................................................................................................
(4)
(Total 11 marks)
2. Sodium chlorate(V), NaClO3, contains 21.6% by mass of sodium, 33.3% by mass of chlorine
and 45.1% by mass of oxygen.
(a) Use the above data to show that the empirical formula of sodium chlorate(V) is
NaClO3
...........................................................................................................................
...........................................................................................................................
...........................................................................................................................
...........................................................................................................................
...........................................................................................................................
(b) Sodium chlorate(V) may be prepared by passing chlorine into hot aqueous sodium
hydroxide. Balance the equation for this reaction below.
…… Cl2 + …… NaOH → …… NaCl + NaClO3 + 3H2O
(3)
(Total 3 marks)
Mill Hill High School 2
3. Potassium nitrate, KNO3, decomposes on strong heating, forming oxygen and solid Y as
the only products.
(a) A 1.00 g sample of KNO3 (Mr = 101.1) was heated strongly until fully decomposed
into Y.
(i) Calculate the number of moles of KNO3 in the 1.00 g sample.
...........................................................................................................................
...........................................................................................................................
(ii) At 298 K and 100 kPa, the oxygen gas produced in this decomposition occupied a
–4 3
volume of 1.22 × 10 m .
State the ideal gas equation and use it to calculate the number of moles of oxygen
produced in this decomposition.
–1 –1
(The gas constant R = 8.31 J K mol )
Ideal gas equation .............................................................................................
Moles of oxygen ................................................................................................
...........................................................................................................................
...........................................................................................................................
...........................................................................................................................
...........................................................................................................................
(5)
(b) Compound Y contains 45.9% of potassium and 16.5% of nitrogen by mass, the remainder
being oxygen.
(i) State what is meant by the term empirical formula.
...........................................................................................................................
...........................................................................................................................
(ii) Use the data above to calculate the empirical formula of Y.
...........................................................................................................................
...........................................................................................................................
...........................................................................................................................
(4)
(c) Deduce an equation for the decomposition of KNO 3 into Y and oxygen.
.....................................................................................................................................
Mill Hill High School 3
(1)
(Total 10 marks)
4.
Mill Hill High School 4
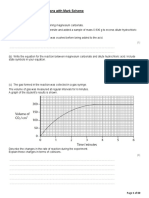
5. The equation for the reaction between magnesium hydroxide and hydrochloric
acid is
shown below.
Mg(OH)2(s) + 2HCl(aq) → MgCl2(aq) + 2H2O(l)
3 –3
Calculate the volume, in cm , of 1.00 mol dm hydrochloric acid required to react
completely with 1.00 g of magnesium hydroxide.
.....................................................................................................................................
.....................................................................................................................................
.....................................................................................................................................
.....................................................................................................................................
.....................................................................................................................................
.....................................................................................................................................
(4)
(Total 4 marks)
5. When aluminium is added to an aqueous solution of copper(II) chloride, CuCl 2, copper
metal and aluminium chloride, AlCl3, are formed. Write an equation to represent this
reaction.
.....................................................................................................................................
(1)
(Total 1 mark)
6. (a) Lead(II) nitrate may be produced by the reaction between nitric acid and lead(II) oxide as
shown by the equation below.
PbO + 2HNO3 ® Pb(NO3)2 + H2O
3 –3
An excess of lead(II) oxide was allowed to react with 175 cm of 1.50 mol dm nitric
acid. Calculate the maximum mass of lead(II) nitrate which could be obtained from this
reaction.
......................................................................................................................................
......................................................................................................................................
......................................................................................................................................
......................................................................................................................................
......................................................................................................................................
(4)
Mill Hill High School 5
(b) An equation representing the thermal decomposition of lead(II) nitrate is shown below.
2Pb(NO3)2(s) → 2PbO(s) + 4NO2(g) + O2(g)
A sample of lead(II) nitrate was heated until the decomposition was complete. At a
temperature of 500 K and a pressure of 100 kPa, the total volume of the gaseous mixture
–4 3
produced was found to be 1.50 × 10 m .
(i) State the ideal gas equation and use it to calculate the total number of moles of gas
produced in this decomposition.
–1 –1
(The gas constant R = 8.31 J K mol )
Ideal gas equation .........................................................................................
Total number of moles of gas ............................................................................
............................................................................................................................
............................................................................................................................
............................................................................................................................
(ii) Deduce the number of moles, and the mass, of NO2 present in this gaseous
mixture. (If you have been unable to calculate the total number of moles of gas in
–3
part (b)(i), you should assume this to be 2.23 × 10 mol. This is not the correct
answer.)
Number of moles of NO2 ..................................................................................
...........................................................................................................................
Mass of NO2 ........................................................................................................
............................................................................................................................
(7)
(Total 11 marks)
7. (a) Ammonia, NH3, reacts with sodium to form sodium amide, NaNH 2, and hydrogen.
Write an equation for the reaction between ammonia and sodium.
........................................................................................…...............................
(1)
Mill Hill High School 6
(b) A salt, X, contains 16.2% by mass of magnesium, 18.9% by mass of nitrogen and 64.9%
by mass of oxygen.
(i) State what is meant by the term empirical formula.
........................................................................................…...............................
........................................................................................…...............................
(ii) Determine the empirical formula of X.
........................................................................................…...............................
........................................................................................…...............................
........................................................................................…...............................
........................................................................................…...............................
(3)
(Total 4 marks)
Mill Hill High School 7
8. (a) Ammonium sulphate reacts with aqueous sodium hydroxide as shown by the equation
below.
(NH4)2SO4 + 2NaOH ® 2NH3 + Na2SO4 + 2H2O
3 –3
A sample of ammonium sulphate was heated with 100 cm of 0.500 mol dm
aqueous sodium hydroxide. To ensure that all the ammonium sulphate reacted, an excess
of sodium hydroxide was used.
Heating was continued until all of the ammonia had been driven off as a gas.
3
The unreacted sodium hydroxide remaining in the solution required 27.3 cm of
–3
0.600 mol dm hydrochloric acid for neutralisation.
3 –3
(i) Calculate the original number of moles of NaOH in 100 cm of 0.500 mol dm
aqueous sodium hydroxide.
........................................................................................…...............................
........................................................................................…...............................
3 –3
(ii) Calculate the number of moles of HCl in 27.3 cm of 0.600 mol dm
hydrochloric acid.
........................................................................................…...............................
........................................................................................…...............................
(iii) Deduce the number of moles of the unreacted NaOH neutralised by the
hydrochloric acid.
........................................................................................…...............................
(iv) Use your answers from parts (a) (i) and (a) (iii) to calculate the number of moles of
NaOH which reacted with the ammonium sulphate.
........................................................................................…...............................
........................................................................................…...............................
(v) Use your answer in part (a) (iv) to calculate the number of moles and the mass of
ammonium sulphate in the sample.
(If you have been unable to obtain an answer to part (a) (iv), you may assume that
the number of moles of NaOH which reacted with ammonium sulphate equals
–2
2.78 × 10 mol. This is not the correct answer.)
Moles of ammonium sulphate ...........................................................................
........................................................................................…...............................
Mass of ammonium sulphate ............................................….............................
........................................................................................…...............................
(7)
Mill Hill High School 8
–4 3
(b) A 0.143g gaseous sample of ammonia occupied a volume of 2.86 × 10 m at a
temperature T and a pressure of 100 kPa.
State the ideal gas equation, calculate the number of moles of ammonia present and
deduce the value of the temperature T.
–1 –1
(The gas constant R = 8.31 J K mol )
Ideal gas equation ......................................................................................................
Moles of ammonia ......................................................................................................
.........................……….............................................................…...............................
Value of T ...................................................................................................................
.........................……….............................................................…...............................
.........................……….............................................................…...............................
.........................……….............................................................…...............................
.........................……….............................................................…...............................
(4)
(Total 11 marks)
9. Compound A is an oxide of sulphur. At 415 K, a gaseous sample of A, of mass 0.304 g,
3
occupied a volume of 127 cm at a pressure of 103 kPa.
State the ideal gas equation and use it to calculate the number of moles of A in the sample, and
hence calculate the relative molecular mass of A.
–1 –1
(The gas constant R = 8.31 J K mol )
Ideal gas equation .................................................................................................................
Calculation ............................................................................................................................
...............................................................................................................................................
...............................................................................................................................................
...............................................................................................................................................
...............................................................................................................................................
(Total 5 marks)
Mill Hill High School 9
10. A hydrocarbon, W, contains 92.3% carbon by mass. The relative molecular mass of W is 78.0
(a) Calculate the empirical formula of W.
.....................................................................................................................
.....................................................................................................................
.....................................................................................................................
.....................................................................................................................
(b) Calculate the molecular formula of W.
.....................................................................................................................
......................................................................................................................
(4)
(Total 4 marks)
11. (a) One isotope of sodium has a relative mass of 23.
Calculate the mass, in grams, of a single atom of this isotope of sodium.
–1
(The Avogadro constant, L, is 6.023 × 1023 mol )
...........................................................................................................................
...........................................................................................................................
...........................................................................................................................
(2)
(Total 2 marks)
12. (a) State what is meant by the term empirical formula.
...........................................................................................................................
...........................................................................................................................
(b) A chromium compound contains 28.4% of sodium and 32.1% of chromium by
mass, the remainder being oxygen.
Calculate the empirical formula of this compound.
...........................................................................................................................
...........................................................................................................................
...........................................................................................................................
...........................................................................................................................
(4)
(Total 4 marks)
Mill Hill High School 10
13. (a) Titanium(IV) chloride reacted with water as shown in the following equation.
TiCl4(l) + 2H2O(l) ® 4HCl(aq) + TiO2(s)
3
The reaction produced 200 cm of a 1.20M solution of hydrochloric acid.
Calculate the number of moles of HCl in the solution and use your answer to
find the original mass of TiCl4
Moles of HCl...............................................................................................................
.....................................................................................................................................
Mass of TiCl4..............................................................................................................
.....................................................................................................................................
.....................................................................................................................................
.....................................................................................................................................
(4)
(b) Calculate the volume of 1.10 M sodium hydroxide solution which would be required to
3
neutralise a 100 cm portion of the 1.20 M solution of hydrochloric acid.
.....................................................................................................................................
.....................................................................................................................................
.....................................................................................................................................
(3)
3
(c) An excess of magnesium metal was added to a 100 cm portion of the 1.20 M solution of
hydrochloric acid. Calculate the volume of hydrogen gas produced at 98 kPa and 20°C.
Mg(s) + 2HCl(aq)® 4 MgCl2(aq) + H2(g)
.....................................................................................................................................
.....................................................................................................................................
.....................................................................................................................................
.....................................................................................................................................
(4)
(Total 11 marks)
Mill Hill High School 11
14.
(a) Calculate the maximum mass of sodium sulphide that can be obtained from 10.0 g
of sulphur.
(2)
3
(b) Calculate the minimum volume of hydrogen, in cm , at 298 K and 101.3 kPa, that
is needed to form 5.00 g of hydrogen sulphide.
(3)
(Total 5 marks)
15. When a sample of liquid, X, of mass 0.406 g was vaporised, the vapour was found to occupy a
–4 3
volume of 2.34 × 10 m at a pressure of 110 kPa and a temperature of 473 K.
(a) Give the name of the equation pV = nRT.
...............................................................................................................................
(1)
(b) Use the equation pV = nRT to calculate the number of moles of X in the sample and
hence deduce the relative molecular mass of X.
–1 –1
(The gas constant R = 8.31 J K mol )
Moles of X ............................................................................................................
...............................................................................................................................
...............................................................................................................................
Relative molecular mass of X ...............................................................................
...............................................................................................................................
(4)
Mill Hill High School 12
(c) Compound X, which contains carbon, hydrogen and oxygen only, has 38.7% carbon and
9.68% hydrogen by mass. Calculate the empirical formula of X.
...............................................................................................................................
...............................................................................................................................
...............................................................................................................................
...............................................................................................................................
(3)
(d) Using your answers to parts (b) and (c) above, deduce the molecular formula of X.
...............................................................................................................................
...............................................................................................................................
(1)
(Total 9 makrs)
–3
16. (a) Calculate the concentration, in mol dm , of the solution formed when 19.6 g of hydrogen
3
chloride, HCl, are dissolved in water and the volume made up to 250 cm .
...............................................................................................................................
...............................................................................................................................
...............................................................................................................................
(3)
Mill Hill High School 13
(b) The carbonate of metal M has the formula M2CO3. The equation for the reaction of this
carbonate with hydrochloric acid is given below.
M2CO3 + 2HCl ® 2MCl + CO2 + H2O
3
A sample of M2CO3, of mass 0.394 g, required the addition of 21.7 cm of a
–3
0.263 mol dm solution of hydrochloric acid for complete reaction.
(i) Calculate the number of moles of hydrochloric acid used.
....................................................................................................................
....................................................................................................................
(ii) Calculate the number of moles of M2CO3 in 0.394 g.
....................................................................................................................
....................................................................................................................
(iii) Calculate the relative molecular mass of M 2CO3
....................................................................................................................
....................................................................................................................
(iv) Deduce the relative atomic mass of M and hence suggest its identity.
Relative atomic mass of M ........................................................................
....................................................................................................................
Identity of M ..............................................................................................
(6)
(Total 9 marks)
17. (a) The mass of one mole of 1H atoms is 1.0078 g and that of one 1H atom is
–24
1.6734 × 10 g.
Use these data to calculate a value for the Avogadro constant accurate to five significant
figures. Show your working.
...............................................................................................................................
...............................................................................................................................
...............................................................................................................................
(2)
(b) How does the number of atoms in one mole of argon compare with the number of
molecules in one mole of ammonia?
...............................................................................................................................
(1)
Mill Hill High School 14
Mill Hill High School 15
3
(c) A sample of ammonia gas occupied a volume of 0.0352 m at 298 K and 98.0 kPa.
Calculate the number of moles of ammonia in the sample.
–1 –1
(The gas constant R = 8.31 J K mol )
...............................................................................................................................
...............................................................................................................................
...............................................................................................................................
...............................................................................................................................
(3)
3
(d) A solution containing 0.732 mol of ammonia was made up to 250 cm in a volumetric
flask by adding water. Calculate the concentration of ammonia in this final solution and
state the appropriate units.
...............................................................................................................................
...............................................................................................................................
...............................................................................................................................
(2)
(e) A different solution of ammonia was reacted with sulphuric acid as shown in the equation
below.
2NH3(aq) + H2SO4(aq) ® (NH4)2SO4(aq)
3 –3
In a titration, 25.0 cm of a 1.24 mol dm solution of sulphuric acid required 30.8 cm3 of
this ammonia solution for complete reaction.
(i) Calculate the concentration of ammonia in this solution.
......................................................................................................................
......................................................................................................................
......................................................................................................................
......................................................................................................................
(ii) Calculate the mass of ammonium sulphate in the solution at the end of this
titration.
......................................................................................................................
......................................................................................................................
......................................................................................................................
......................................................................................................................
(6)
Mill Hill High School 16
Mill Hill High School 17
(f) The reaction of magnesium nitride, Mg3N2, with water produces ammonia and
magnesium hydroxide. Write an equation for this reaction.
................................................................................................................................
(2)
(Total 16 marks)
18. (a) Define the term relative molecular mass.
.....................................................................................................................................
.....................................................................................................................................
(2)
(b) Give the meaning of the term empirical formula.
.....................................................................................................................................
.....................................................................................................................................
(1)
(c) Compound X contains 32.9% by mass of carbon and 1.40% by mass of hydrogen; the
remainder is oxygen.
(i) Calculate the empirical formula of X.
...........................................................................................................................
...........................................................................................................................
...........................................................................................................................
...........................................................................................................................
...........................................................................................................................
(ii) The relative molecular mass of X is 146. Deduce its molecular formula.
...........................................................................................................................
...........................................................................................................................
(4)
Mill Hill High School 18
(d) A 1.0 kg sample of methane was burned in air. It reacted as follows:
CH4 (g) + 2O2 (g) ® CO2 (g) + 2H2O(g)
(i) Calculate the number of moles in 1.0 kg of methane.
...........................................................................................................................
...........................................................................................................................
(ii) Calculate the volume of oxygen gas, measured at 298 K and 100 kPa, which would
be required for the complete combustion of 1.0 kg of methane.
...........................................................................................................................
...........................................................................................................................
...........................................................................................................................
...........................................................................................................................
(6)
(Total 13 marks)
19. Give the meaning of the term mole as used in the phrase 'one mole of molecules'.
...................................................................................................................................
(1)
(Total 1 mark)
20. (a) What is the name given to the number of molecules in one mole of carbon dioxide?
.....................................................................................................................................
(1)
Mill Hill High School 19
(b) (i) State the ideal gas equation.
...........................................................................................................................
(ii) Calculate the volume of 1.00 mol of carbon dioxide gas at 298 K and 100 kPa.
–1 –1
(The gas constant R = 8.31 J mol K )
...........................................................................................................................
...........................................................................................................................
...........................................................................................................................
...........................................................................................................................
(iii) Calculate the mass of carbon dioxide gas at 273 K and 500 kPa contained in a
3
cylinder of volume 0.00500 m .
...........................................................................................................................
...........................................................................................................................
...........................................................................................................................
...........................................................................................................................
...........................................................................................................................
(7)
(c) Hydrogen can be made by the reaction of hydrochloric acid with magnesium according to
the equation
2HCl + Mg ® MgCl2 + H2
3
What mass of hydrogen is formed when 100cm of hydrochloric acid of concentration 5.0
–3
mol dm reacts with an excess of magnesium?
.....................................................................................................................................
.....................................................................................................................................
.....................................................................................................................................
.....................................................................................................................................
.....................................................................................................................................
(3)
Mill Hill High School 20
(d) A compound of iron contains 38.9% by mass of iron and 16.7% by mass of carbon, the
remainder being oxygen.
(i) Determine the empirical formula of the iron compound.
...........................................................................................................................
...........................................................................................................................
...........................................................................................................................
...........................................................................................................................
...........................................................................................................................
(ii) When one mole of this iron compound is heated, it decomposes to give one mole of
iron(II) oxide, FeO, one mole of carbon dioxide and one mole of another gas.
Identify this other gas. (The molecular formula of the iron compound is the same as
its empirical formula.)
...........................................................................................................................
(4)
(Total 15 marks)
21. Ammonium nitrate can be prepared by the reaction between ammonia and nitric acid:
NH3 + HNO3 ® NH4NO3
–3
(a) The concentration of a nitric acid solution is 2.00 mol dm . Calculate the volume of this
solution which would be required to react with exactly 20.0 g of ammonia.
.....................................................................................................................................
.....................................................................................................................................
.....................................................................................................................................
.....................................................................................................................................
(4)
Mill Hill High School 21
(b) A sample of ammonium nitrate decomposed on heating as shown in the equation below.
NH4NO3 ® 2H2O + N2 + ½O2
On cooling the resulting gases to 298 K, the volume of nitrogen and oxygen together was
3
found to be 0.0500 m at a pressure of 95.0 kPa.
(i) State the ideal gas equation and use it to calculate the total number of moles of
–1 –1
nitrogen and oxygen formed. (The gas constant R = 8.31 J mol K )
...........................................................................................................................
...........................................................................................................................
...........................................................................................................................
...........................................................................................................................
...........................................................................................................................
(ii) Using your answer to part (b)(i), deduce the number of moles of ammonium nitrate
decomposed and hence calculate the mass of ammonium nitrate in the sample.
Moles of ammonium nitrate ..............................................................................
Mass of ammonium nitrate ...............................................................................
...........................................................................................................................
(6)
(Total 10 marks)
12 –23
22. The mass of one atom of C is 1.99 × 10 g. Use this information to calculate a value
for the Avogadro constant. Show your working.
.....................................................................................................................................
.....................................................................................................................................
(2)
(Total 2 marks)
Mill Hill High School 22
23. The equation below represents the thermal decomposition of KClO 3.
2KClO3(s) ® 2KCl(s) + 3O2(g)
(a) Calculate the mass of oxygen which could be produced by the complete
decomposition of 1.47 g of KClO3.
(2)
3
(b) Calculate the mass of KClO3 required to produce 1.00 dm (at 20 °C and 101.3
kPa) of oxygen.
(3)
(Total 5 marks)
24. (a) What experimental data are required in order to calculate the empirical formula of a
compound?
.....................................................................................................................................
.....................................................................................................................................
(1)
(b) Give the meaning of the term molecular formula.
.....................................................................................................................................
.....................................................................................................................................
(1)
Mill Hill High School 23
(c) When barium nitrate is heated it decomposes as follows:
Ba(NO3)2(s) ® BaO(s) + 2NO2(g) + ½ O2(g)
(i) Calculate the total volume, measured at 298 K and 100 kPa, of gas which is
produced by decomposing 5.00 g of barium nitrate.
............................................................................................................................
............................................................................................................................
............................................................................................................................
............................................................................................................................
............................................................................................................................
(ii) Calculate the volume of 1.20 M hydrochloric acid which is required to neutralise
exactly the barium oxide formed by decomposition of 5.00 g of barium nitrate.
Barium oxide reacts with hydrochloric acid as follows
BaO(s) + 2HCl(aq) ® BaCl2(aq) + H2O(l)
............................................................................................................................
............................................................................................................................
............................................................................................................................
(7)
(Total 9 marks)
25. The mass spectrum of a compound has a molecular ion peak at m/z = 168.
Elemental analysis shows it to contain 42.9% carbon, 2.4% hydrogen and 16.7% nitrogen by
mass. The remainder is oxygen.
Calculate the empirical and molecular formulae of this compound
(4)
(Total 4 marks)
Mill Hill High School 24
26. Compound X contains only boron and hydrogen. The percentage by mass of boron in X is
81.2%. In the mass spectrum of X the peak at the largest value of m/z occurs at 54.
(a) Use the percentage by mass data to calculate the empirical formula of X.
...........................................................................................................................
...........................................................................................................................
...........................................................................................................................
(b) Deduce the molecular formula of X.
...........................................................................................................................
(4)
(Total 4 marks)
27. (a) Define the term relative molecular mass.
.....................................................................................................................................
.....................................................................................................................................
(2)
12 –23
(b) The mass of one atom of C is 1.993 × 10 g. Use this mass to calculate a value
for the Avogadro constant (L) showing your working.
.....................................................................................................................................
.....................................................................................................................................
(1)
(Total 3 marks)
28. When iodine reacts directly with fluorine, a compound containing 57.2% by mass of
iodine is formed.
(a) Determine the empirical formula of this compound.
...........................................................................................................................
...........................................................................................................................
...........................................................................................................................
(b) The empirical formula of this compound is the same as the molecular formula.
Write a balanced equation for the formation of this compound.
...........................................................................................................................
(4)
(Total 4 marks)
Mill Hill High School 25
29. Compound A (Mr = 215.8) contains 22.24% carbon, 3.71% hydrogen and 74.05%
bromine by mass. Show that the molecular formula of A is C4H8Br2.
(3)
(Total 3 marks)
Mill Hill High School 26
You might also like
- Mole Concept Lesson PlanDocument4 pagesMole Concept Lesson PlanJhezreel John100% (3)
- Chemistry Malaysian Matriculation Full Notes Amp Slides For Semester 1 and 2 PDFDocument1,743 pagesChemistry Malaysian Matriculation Full Notes Amp Slides For Semester 1 and 2 PDFHaarini100% (1)
- Amount of Substance CombinedDocument23 pagesAmount of Substance CombinedJainam MehtaNo ratings yet
- Periodic Table, Group 2 and The Halogens 2 QPDocument14 pagesPeriodic Table, Group 2 and The Halogens 2 QPmalakNo ratings yet
- The Mole & Avogadro Constant 1 QPDocument8 pagesThe Mole & Avogadro Constant 1 QPkateNo ratings yet
- Balanced Equations & Associated Calc's 05 QPDocument8 pagesBalanced Equations & Associated Calc's 05 QPlmao lmaoNo ratings yet
- As Level Chemistry: Topic 2 - Amount of Substance TestDocument10 pagesAs Level Chemistry: Topic 2 - Amount of Substance Testkarima akterNo ratings yet
- Amount of Substance PPQsDocument10 pagesAmount of Substance PPQsLakshyaSoniNo ratings yet
- F321 Module 3 Practice 5Document4 pagesF321 Module 3 Practice 5coughsyrup123No ratings yet
- 9701 s17 QP 42 RemovedDocument18 pages9701 s17 QP 42 RemovedSherise EeNo ratings yet
- Energy Changes, Rate, Reversible, NH3, H2SO4Document5 pagesEnergy Changes, Rate, Reversible, NH3, H2SO4HudaNo ratings yet
- Chemical Energetics 2 QPDocument11 pagesChemical Energetics 2 QPWilliam TsuiNo ratings yet
- 1 Hydrogen Peroxide Decomposes To Form Water and Oxygen. This Reaction Is Catalysed byDocument9 pages1 Hydrogen Peroxide Decomposes To Form Water and Oxygen. This Reaction Is Catalysed byKelvin DenhereNo ratings yet
- Amount of SubstanceDocument13 pagesAmount of Substanceyahvip07No ratings yet
- 2.3.1 Enthalpy ChangesDocument25 pages2.3.1 Enthalpy Changesp01084240882No ratings yet
- Rate of Reaction 2 QP (Tomek)Document9 pagesRate of Reaction 2 QP (Tomek)Tomasz OstrowskiNo ratings yet
- My TestDocument20 pagesMy TestHidayah TeacherNo ratings yet
- F321: Atoms, Bonds and Groups Redox: Plymstock School 1Document7 pagesF321: Atoms, Bonds and Groups Redox: Plymstock School 1Younes AlahmadNo ratings yet
- F321 Module 1 Practice 4Document7 pagesF321 Module 1 Practice 4coughsyrup123No ratings yet
- Reaction Rates - Equilibrium 2 QPDocument18 pagesReaction Rates - Equilibrium 2 QPKeerthy verNo ratings yet
- Born-Haber Cycles 1 QP PDFDocument9 pagesBorn-Haber Cycles 1 QP PDFTalha KhanNo ratings yet
- Topic 4 - Group 7Document9 pagesTopic 4 - Group 7Abirame SivakaranNo ratings yet
- t2 Chem Revision Ex 22Document19 pagest2 Chem Revision Ex 22Nicholas OwNo ratings yet
- t2 Chem Revision Ex 18Document16 pagest2 Chem Revision Ex 18Nicholas OwNo ratings yet
- Metal p4 Ws 4Document10 pagesMetal p4 Ws 4NonnoonoNo ratings yet
- Extra Moles QuestionsDocument6 pagesExtra Moles QuestionschenzNo ratings yet
- Periodicity Past QuestionsDocument17 pagesPeriodicity Past QuestionsMohammad KhanNo ratings yet
- Water and HydrogenDocument7 pagesWater and HydrogenOkumu KevinsNo ratings yet
- WS A Level CHEMDocument57 pagesWS A Level CHEMPreet ShahNo ratings yet
- P6 Chemical Equilibirua Le Chatelier's Principle and KCDocument73 pagesP6 Chemical Equilibirua Le Chatelier's Principle and KC/ “Nu” /No ratings yet
- Reversible ReactionDocument17 pagesReversible ReactionNavyaa SinghNo ratings yet
- 03 0620 42 3RP - InddDocument5 pages03 0620 42 3RP - InddIzzati AnuarNo ratings yet
- Topical Questions For ElectrolysisDocument6 pagesTopical Questions For Electrolysisokguserfucker idontgiveashitNo ratings yet
- 525 1 Eot 3 2022Document17 pages525 1 Eot 3 2022OTTO OLIMANo ratings yet
- Test 1 As Chemistry Unit 2 - KineticsDocument10 pagesTest 1 As Chemistry Unit 2 - KineticsKajana Sivarasa Shenthan100% (1)
- Mixed Topic Revision 1 DiffusionDocument23 pagesMixed Topic Revision 1 DiffusionYaakkwNo ratings yet
- Introducing Energy Changes in ReactionsDocument18 pagesIntroducing Energy Changes in ReactionsFatema KhatunNo ratings yet
- GP 7 AsDocument129 pagesGP 7 Ashussainjunaid210No ratings yet
- Calculations QuestionsDocument14 pagesCalculations Questionsan7li721No ratings yet
- F321 Module 3 Practice 2Document7 pagesF321 Module 3 Practice 2coughsyrup123No ratings yet
- Energetics: Q1. (A) State What Is Meant by The Term Mean Bond EnthalpyDocument95 pagesEnergetics: Q1. (A) State What Is Meant by The Term Mean Bond EnthalpyWeronika JRNo ratings yet
- A-Level Paper 1 pp1Document14 pagesA-Level Paper 1 pp1fibim38794No ratings yet
- Chemical EnergerticsDocument24 pagesChemical EnergerticsAnotidaishe ChakanetsaNo ratings yet
- Topic 9 Past Papers Questions With Mark SchemeDocument60 pagesTopic 9 Past Papers Questions With Mark SchemeQasim PerachaNo ratings yet
- As-Level Paper 2 pp8Document11 pagesAs-Level Paper 2 pp8facts.agency45No ratings yet
- Cambridge IGCSE: Chemistry 0620Document14 pagesCambridge IGCSE: Chemistry 0620PizzaNo ratings yet
- Stoichiometry 4 QPDocument8 pagesStoichiometry 4 QPraghavi luthraNo ratings yet
- Jan 2019Document27 pagesJan 2019Mariyam ManaarathNo ratings yet
- t2 Chem Revision Ex 18 Answer SchemeDocument17 pagest2 Chem Revision Ex 18 Answer SchemeNicholas OwNo ratings yet
- Topic 4 - Group 2 Volume 2Document11 pagesTopic 4 - Group 2 Volume 2Abirame SivakaranNo ratings yet
- t2 Chem Revision Ex 12Document16 pagest2 Chem Revision Ex 12Nicholas OwNo ratings yet
- Mock PaperDocument9 pagesMock PaperjunetaskinNo ratings yet
- Nitroglycerine, C3H5N3O9, Is An Explosive Which, On Detonation, Decomposes RapidlyDocument14 pagesNitroglycerine, C3H5N3O9, Is An Explosive Which, On Detonation, Decomposes Rapidlyapi-25909541No ratings yet
- WS2 Molesgr9Document6 pagesWS2 Molesgr9irabedi9No ratings yet
- Empirical & Molecular Formula 2 QPDocument8 pagesEmpirical & Molecular Formula 2 QPjade.davis0019No ratings yet
- As Level Chemistry: Topic 2 - Amount of Substance Assessed HomeworkDocument15 pagesAs Level Chemistry: Topic 2 - Amount of Substance Assessed Homeworkkarima akterNo ratings yet
- A-Level: ChemistryDocument10 pagesA-Level: ChemistryNusrat HoqueNo ratings yet
- A-Level: ChemistryDocument8 pagesA-Level: ChemistryRoshae SinclairNo ratings yet
- Quiz - 1 - Matter & StoichiometryDocument4 pagesQuiz - 1 - Matter & StoichiometryDaniel Ngenokesho WandyaNo ratings yet
- Group 2 - The Alkaline Earth Metals 1 QPDocument8 pagesGroup 2 - The Alkaline Earth Metals 1 QPWilliam TsuiNo ratings yet
- Advanced Synthesis of Gold and Zirconia Nanoparticles and their CharacterizationFrom EverandAdvanced Synthesis of Gold and Zirconia Nanoparticles and their CharacterizationNo ratings yet
- Test 14 MechanicsDocument16 pagesTest 14 MechanicsKamrul Alam MasumNo ratings yet
- Test 05 MaterialsDocument16 pagesTest 05 MaterialsKamrul Alam MasumNo ratings yet
- Test 04 Further MechanicsDocument18 pagesTest 04 Further MechanicsKamrul Alam Masum100% (1)
- Test 06 MaterialsDocument30 pagesTest 06 MaterialsKamrul Alam MasumNo ratings yet
- Examiner Report Test 04 MechanicsDocument40 pagesExaminer Report Test 04 MechanicsKamrul Alam MasumNo ratings yet
- Spectroscopy & ChromatographyDocument23 pagesSpectroscopy & ChromatographyKamrul Alam MasumNo ratings yet
- Examiner Report of Test 03 MechanicsDocument36 pagesExaminer Report of Test 03 MechanicsKamrul Alam MasumNo ratings yet
- Test 02 Further MechanicsDocument20 pagesTest 02 Further MechanicsKamrul Alam MasumNo ratings yet
- Mark-Scheme of Test 03 MechanicsDocument7 pagesMark-Scheme of Test 03 MechanicsKamrul Alam MasumNo ratings yet
- Test 03 MechanicsDocument22 pagesTest 03 MechanicsKamrul Alam MasumNo ratings yet
- Ms Test 01 MechanicsDocument11 pagesMs Test 01 MechanicsKamrul Alam MasumNo ratings yet
- Test 04 MechanicsDocument26 pagesTest 04 MechanicsKamrul Alam MasumNo ratings yet
- Test 01 Further MechanicsDocument16 pagesTest 01 Further MechanicsKamrul Alam MasumNo ratings yet
- MD. KAMRUL ALAM KHAN, B.SC Honors in Chemistry (SUST), M.SC in Chemistry (SUST), CCNAE (All Through First Class), CELL: 01557704046Document10 pagesMD. KAMRUL ALAM KHAN, B.SC Honors in Chemistry (SUST), M.SC in Chemistry (SUST), CCNAE (All Through First Class), CELL: 01557704046Kamrul Alam MasumNo ratings yet
- Test 06 MechanicsDocument18 pagesTest 06 MechanicsKamrul Alam MasumNo ratings yet
- As Practical 11 - Heating SolidsDocument2 pagesAs Practical 11 - Heating SolidsKamrul Alam MasumNo ratings yet
- Test 05 MechanicsDocument28 pagesTest 05 MechanicsKamrul Alam MasumNo ratings yet
- MD. KAMRUL ALAM KHAN, B.SC Honors in Chemistry (SUST), M.SC in Chemistry (SUST), CCNA (All Through First Class), CELL: 01557704046Document4 pagesMD. KAMRUL ALAM KHAN, B.SC Honors in Chemistry (SUST), M.SC in Chemistry (SUST), CCNA (All Through First Class), CELL: 01557704046Kamrul Alam MasumNo ratings yet
- Astrophysics For Edexcel IgcseDocument16 pagesAstrophysics For Edexcel IgcseKamrul Alam Masum100% (3)
- MCQ HydrocarbonDocument20 pagesMCQ HydrocarbonKamrul Alam Masum100% (1)
- Structure 1.4 - Counting Particles by Mass The MoleDocument92 pagesStructure 1.4 - Counting Particles by Mass The Molemohammedhider71No ratings yet
- Apex Tutorial: 12) Chemistry PathshalaDocument2 pagesApex Tutorial: 12) Chemistry PathshalaratanNo ratings yet
- Required Practical - Investigating Gas LawsDocument29 pagesRequired Practical - Investigating Gas LawsrizwanNo ratings yet
- Chemistry PDFDocument113 pagesChemistry PDFShubham YadavNo ratings yet
- Moles Lesson PlanDocument3 pagesMoles Lesson Planapi-284227012No ratings yet
- Kcse Form 3 Chemistry NotesDocument262 pagesKcse Form 3 Chemistry NotesMichelle Hlungwani100% (1)
- Millikan Oil DropDocument34 pagesMillikan Oil Dropカリイオ ヘクトルNo ratings yet
- 1st Year Chemistry Revision Assignment For Test 1Document9 pages1st Year Chemistry Revision Assignment For Test 1Syed Moeen NaqviNo ratings yet
- NCERT Grade 11 Physics CH 13 Kinetic TheoryDocument14 pagesNCERT Grade 11 Physics CH 13 Kinetic Theory2ly PajingNo ratings yet
- General Chemistry: Atoms and The Atomic TheoryDocument27 pagesGeneral Chemistry: Atoms and The Atomic TheoryOrxan ƏhmədovNo ratings yet
- General Organic and Biological Chemistry 7Th Edition Stoker Solutions Manual Full Chapter PDFDocument18 pagesGeneral Organic and Biological Chemistry 7Th Edition Stoker Solutions Manual Full Chapter PDFallison.young656100% (15)
- Kinetic TheoryDocument54 pagesKinetic Theorykhanan_5No ratings yet
- Chemical Quantities: Section 10.1 The Mole: A Measurement of MatterDocument1 pageChemical Quantities: Section 10.1 The Mole: A Measurement of MatterMm KFZNo ratings yet
- Physics Ideal Gas Law NotesDocument5 pagesPhysics Ideal Gas Law NotesSeblewengelNo ratings yet
- Ch.2 Moles.Document26 pagesCh.2 Moles.basilabdellatiefNo ratings yet
- Namma Kalvi 11th Chemistry Unit 1 and 8 Sura Guide emDocument82 pagesNamma Kalvi 11th Chemistry Unit 1 and 8 Sura Guide emMonisha Kumar100% (1)
- 1 Moles and StoichiometryDocument37 pages1 Moles and StoichiometryArvin LiangdyNo ratings yet
- Lesson 2 - Moles and Molar Masses & Determination of Chemical FormulasDocument15 pagesLesson 2 - Moles and Molar Masses & Determination of Chemical FormulasMelvyn DarauayNo ratings yet
- MolesDocument21 pagesMolesAndre BirchNo ratings yet
- Plus One Chemistry Notes Chapter 1 Some Basic Concepts of Chemistry - A Plus TopperDocument20 pagesPlus One Chemistry Notes Chapter 1 Some Basic Concepts of Chemistry - A Plus Topperpsuresh_reddyNo ratings yet
- Solution Manual For Engineering Fluid Me PDFDocument61 pagesSolution Manual For Engineering Fluid Me PDFOmar Ahmed AbuLeilaNo ratings yet
- Teodor Ognean 1809.0446v1Document8 pagesTeodor Ognean 1809.0446v1arg0nautNo ratings yet
- General Organic and Biological Chemistry 6Th Edition Stoker Test Bank Full Chapter PDFDocument38 pagesGeneral Organic and Biological Chemistry 6Th Edition Stoker Test Bank Full Chapter PDFyhenryhnorc5100% (14)
- Mole Concept ModuleDocument57 pagesMole Concept ModuleManashNo ratings yet
- Quizizz 2 KimiaDocument5 pagesQuizizz 2 KimiaDaffa SayyidNo ratings yet
- Chapter 3 - Stoichiometry 20-2-2016 PDFDocument63 pagesChapter 3 - Stoichiometry 20-2-2016 PDFSyukuri JaafarNo ratings yet
- StoichiometryDocument26 pagesStoichiometryClaus LawrenceNo ratings yet
- 10th Science - I Notes of Lessson PDFDocument104 pages10th Science - I Notes of Lessson PDFTajiriMollelNo ratings yet