Professional Documents
Culture Documents
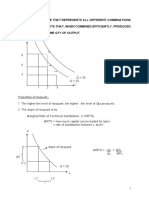
Relation Between KC and KP If KC KP
Relation Between KC and KP If KC KP
Uploaded by
Abdul Akber0 ratings0% found this document useful (0 votes)
8 views1 pageKc represents the equilibrium constant for a chemical reaction in terms of concentrations, while Kp represents the equilibrium constant in terms of partial pressures. If Kc equals Kp, the reaction is independent of concentration and pressure. If Kc is greater than Kp, the reaction favors higher pressures. If Kc is less than Kp, the reaction favors higher concentrations.
Original Description:
Original Title
Ksp.docx
Copyright
© © All Rights Reserved
Available Formats
DOCX, PDF, TXT or read online from Scribd
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentKc represents the equilibrium constant for a chemical reaction in terms of concentrations, while Kp represents the equilibrium constant in terms of partial pressures. If Kc equals Kp, the reaction is independent of concentration and pressure. If Kc is greater than Kp, the reaction favors higher pressures. If Kc is less than Kp, the reaction favors higher concentrations.
Copyright:
© All Rights Reserved
Available Formats
Download as DOCX, PDF, TXT or read online from Scribd
0 ratings0% found this document useful (0 votes)
8 views1 pageRelation Between KC and KP If KC KP
Relation Between KC and KP If KC KP
Uploaded by
Abdul AkberKc represents the equilibrium constant for a chemical reaction in terms of concentrations, while Kp represents the equilibrium constant in terms of partial pressures. If Kc equals Kp, the reaction is independent of concentration and pressure. If Kc is greater than Kp, the reaction favors higher pressures. If Kc is less than Kp, the reaction favors higher concentrations.
Copyright:
© All Rights Reserved
Available Formats
Download as DOCX, PDF, TXT or read online from Scribd
You are on page 1of 1
Relation Between Kc and Kp
If Kc =Kp
If Kc > Kp
If Kc < Kp
You might also like
- SubhashreeDocument15 pagesSubhashreearijit mannaNo ratings yet
- Topic 7 TutorialDocument26 pagesTopic 7 TutorialElsa MahardikaNo ratings yet
- 4.chemical Equilibrium PDFDocument39 pages4.chemical Equilibrium PDFP. E. I. AcademicsNo ratings yet
- ch15 PDFDocument29 pagesch15 PDFHafidz RafiqiNo ratings yet
- Chemical Equilibrium: Condensed VersionDocument19 pagesChemical Equilibrium: Condensed VersionAllen SiaNo ratings yet
- Isoquan Dan IsocostDocument20 pagesIsoquan Dan Isocostkezia aureliaNo ratings yet
- Chem EquiDocument8 pagesChem EquiJeremy CNo ratings yet
- 5th Lesson Comparison TestsDocument15 pages5th Lesson Comparison Testsyirago8621No ratings yet
- The Nature of The Dirac Equation: I T C I X M CDocument4 pagesThe Nature of The Dirac Equation: I T C I X M CjsnvrghseNo ratings yet
- 5-Chemical EquilibriumDocument3 pages5-Chemical EquilibriumVaibhav bhardwajNo ratings yet
- Resources For Students AssessmentDocument1 pageResources For Students Assessment306345407No ratings yet
- ModelCondensation - & IRS TechniquesDocument24 pagesModelCondensation - & IRS Techniquesjuan carlos molano toroNo ratings yet
- Series 1Document5 pagesSeries 1Jickson JoyNo ratings yet
- CS30 Exercise Mod7Document1 pageCS30 Exercise Mod7Race CariNo ratings yet
- Chemical EquilibriumDocument66 pagesChemical EquilibriumalinNo ratings yet
- Applications of First Order DEDocument31 pagesApplications of First Order DEstxtchNo ratings yet
- Fundamentals of NanoelectronicsDocument11 pagesFundamentals of NanoelectronicsSachin Pathak100% (1)
- 2 Chemical Equilibrium - FDocument94 pages2 Chemical Equilibrium - FSelena Dela CruzNo ratings yet
- Isoquant & IsocostDocument5 pagesIsoquant & IsocostFathimahNo ratings yet
- 2WDocument8 pages2WSuneel ChinchanikarNo ratings yet
- Geometric Inequalities: What'S MoreDocument5 pagesGeometric Inequalities: What'S MoreJeanette Bonifacio CorpuzNo ratings yet
- Chemistry A Chemical Equilibrium KineticsDocument56 pagesChemistry A Chemical Equilibrium Kineticsokumuenock000No ratings yet
- Math Ch12Document6 pagesMath Ch12MbambaNo ratings yet
- 7th Congruent TrianglesDocument23 pages7th Congruent TrianglesRajeswari SareddyNo ratings yet
- Millman Halkias - Integrated ElectronicsDocument14 pagesMillman Halkias - Integrated ElectronicsSushruti Richaa KashyapNo ratings yet
- Standard G.SRT.5: ObjectiveDocument15 pagesStandard G.SRT.5: Objectiveth6297cs9kNo ratings yet
- 3 Outliers in Rough K-Means ClusteringDocument6 pages3 Outliers in Rough K-Means ClusteringAnwar ShahNo ratings yet
- EC203 - Problem Set 6 - SolutionsDocument9 pagesEC203 - Problem Set 6 - SolutionshayalabiNo ratings yet
- CHE 402 Omar's Notes (211) 2Document93 pagesCHE 402 Omar's Notes (211) 2mohammedNo ratings yet
- Estimate K ParameterDocument5 pagesEstimate K ParameterKittipun KhamprasoetNo ratings yet
- CHM 111 Notes - 2021 - 2022Document19 pagesCHM 111 Notes - 2021 - 2022j9927091No ratings yet
- Physics DPP 1Document1 pagePhysics DPP 1Crazy iitphysicsNo ratings yet
- Introduction To Reactor Design, 3K4 Tutorial 4/assignment 3ADocument6 pagesIntroduction To Reactor Design, 3K4 Tutorial 4/assignment 3AazatklcNo ratings yet
- Chapter 9 Test 1 Review Outline and ChartsDocument7 pagesChapter 9 Test 1 Review Outline and ChartssatwithalexNo ratings yet
- Math 6110 Pset 1Document6 pagesMath 6110 Pset 1Miguel Antonio Leonardo SepulvedaNo ratings yet
- Bar-Spring System - Structural StabilityDocument10 pagesBar-Spring System - Structural StabilityAnonymous Sl60bL50% (2)
- TrianglesDocument42 pagesTrianglesKavita YadavNo ratings yet
- Che 407 TolDocument4 pagesChe 407 Toldavidolalere7No ratings yet
- pOWER sYSTEM NOTEDocument20 pagespOWER sYSTEM NOTEnishuNo ratings yet
- Grade-10 Mathematics Chapter06 Triangles-1Document9 pagesGrade-10 Mathematics Chapter06 Triangles-1shubham.rNo ratings yet
- Quantum Computing Lecture 14Document4 pagesQuantum Computing Lecture 14JanuszPawlaczNo ratings yet
- 5 - Ce6101-Cam Clay Model-03102020Document30 pages5 - Ce6101-Cam Clay Model-03102020rihongkeeNo ratings yet
- EQUILIBRIUMDocument11 pagesEQUILIBRIUMSumit PatilNo ratings yet
- A Short Course On Mesh Quality: 16 International Meshing Roundtable Seattle, Wa October 14, 2007Document63 pagesA Short Course On Mesh Quality: 16 International Meshing Roundtable Seattle, Wa October 14, 2007huilemoteur2020No ratings yet
- Electricity & Magnetism Lecture 1: Coulomb's LawDocument16 pagesElectricity & Magnetism Lecture 1: Coulomb's LawKen LimoNo ratings yet
- CHEM103 Week 7 Equilibrium Lecture NotesDocument19 pagesCHEM103 Week 7 Equilibrium Lecture NotesOmar MatarNo ratings yet
- Density of StatesDocument2 pagesDensity of StatesVuong100% (1)
- 6.cross Regulation in Flyback Convertrers - SolutionsDocument6 pages6.cross Regulation in Flyback Convertrers - SolutionsMityaNo ratings yet
- Chapter 7 EquilibriumDocument3 pagesChapter 7 EquilibriumRonnie0209No ratings yet
- Optimisation of Measurement Error in Partial Discharge TestingDocument6 pagesOptimisation of Measurement Error in Partial Discharge TestingPeter Jhoan Pulido NuñezNo ratings yet
- May 6 Compressible-Flow-Homework Solutions: U U C T T H H C T T S S C T T RLN P PDocument6 pagesMay 6 Compressible-Flow-Homework Solutions: U U C T T H H C T T S S C T T RLN P PlmjNo ratings yet
- Chapter 1. Angle Chasing: RedpigDocument12 pagesChapter 1. Angle Chasing: RedpigBu BuNo ratings yet
- Title Bout Flow Chart v3.0Document1 pageTitle Bout Flow Chart v3.0Chris van MelsNo ratings yet
- 18 March Shift II PhysicsDocument5 pages18 March Shift II PhysicsHarshul SingalNo ratings yet
- Chemical and Ionic Equilibrium - Short Notes - Yakeen NEET 2024Document4 pagesChemical and Ionic Equilibrium - Short Notes - Yakeen NEET 2024Habibi AmjidNo ratings yet
- Quiz 2 Mod 2 Ce131p-2Document6 pagesQuiz 2 Mod 2 Ce131p-2anonymousoreviewerNo ratings yet
- Conversion and Reactor SizingDocument19 pagesConversion and Reactor SizingJiana NasirNo ratings yet
- NROSCI 1012 - Lecture 11Document4 pagesNROSCI 1012 - Lecture 11HonzaNo ratings yet
- Unit - I - Electrostatics 12th PhysicsDocument22 pagesUnit - I - Electrostatics 12th PhysicsparveezNo ratings yet
- 17.5 Correlated Process and Observation NoiseDocument4 pages17.5 Correlated Process and Observation NoiseAhmet TOLUCNo ratings yet
- Chapter 9 Question Review PDFDocument12 pagesChapter 9 Question Review PDFAbdul AkberNo ratings yet
- Assignment4 PDFDocument4 pagesAssignment4 PDFAbdul AkberNo ratings yet
- General Mental Ability: Measurement InstrumentsDocument3 pagesGeneral Mental Ability: Measurement InstrumentsAbdul AkberNo ratings yet
- Junaid Saleem (sp20-mscs-0051) Problem StatementDocument3 pagesJunaid Saleem (sp20-mscs-0051) Problem StatementAbdul AkberNo ratings yet
- Valance Bond Theory (VBT)Document1 pageValance Bond Theory (VBT)Abdul AkberNo ratings yet
- The Equilibrium Constant Expression For A Gaseous Equilibrium Is: K (NO) + (H O) / (NH) + (O) From This Write The Equation For The ReactionDocument1 pageThe Equilibrium Constant Expression For A Gaseous Equilibrium Is: K (NO) + (H O) / (NH) + (O) From This Write The Equation For The ReactionAbdul AkberNo ratings yet
- KC&KPDocument1 pageKC&KPAbdul AkberNo ratings yet
- Dipole Moment: Ii) Magnitude of ChargeDocument1 pageDipole Moment: Ii) Magnitude of ChargeAbdul AkberNo ratings yet
- Ionization EnergyDocument1 pageIonization EnergyAbdul AkberNo ratings yet
- Mcqs 1Document2 pagesMcqs 1Abdul AkberNo ratings yet
- Atomic Structure Chapter # 03: List of TopicsDocument6 pagesAtomic Structure Chapter # 03: List of TopicsAbdul AkberNo ratings yet
- Mcqs 1Document2 pagesMcqs 1Abdul AkberNo ratings yet
- N + L Rule/ Wise-Wesser Rule:sDocument1 pageN + L Rule/ Wise-Wesser Rule:sAbdul AkberNo ratings yet
- McqsDocument4 pagesMcqsAbdul AkberNo ratings yet
- McqsDocument4 pagesMcqsAbdul AkberNo ratings yet