Professional Documents
Culture Documents
Gas Laws Ws PDF
Uploaded by
Ivy JoyceOriginal Title
Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
Gas Laws Ws PDF
Uploaded by
Ivy JoyceCopyright:
Available Formats
CHEMISTRY GAS LAW’S WORKSHEET
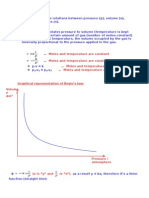
Boyle’s Law Charles’ Law Guy-Lassac's Law Combined Gas Law
For a given mass of gas The volume of a fixed The pressure of a gas is Combines Boyle’s,
at constant temperature, mass of gas is directly directly proportional to Charles’, and the
the volume of a gas proportional to its the Kelvin temperature if Temperature-Pressure
varies inversely with Kelvin temperature if the volume is kept relationship into one
pressure the pressure is kept constant. equation. Each of these
constant. laws can be derived from
this law.
V P PV
PV = k =k =k =k
T T T
V1 T2 = V2T1 P1T2 = P2T1 V1P1T2 = V2P2T1
P1V1 = P2V2 V1 V2 P1 P2 P1V1 P2 V2
= = =
T1 T2 T1 T2 T1 T2
Dalton’s Law Ideal Gas Law Graham’s Law
At constant volume and The Ideal Gas Law relates the The rate of effusion/diffusion of
temperature, the total pressure pressure, temperature, volume, two gases (A and B) are inversely
exerted by a mixture of gases is and mass of a gas through the proportional to the square roots
equal to the sum of the pressures gas constant “R”. of their formula masses. [It can
exerted by each gas, be a ratio of molecular speeds,
effusion /diffusion times, distance
traveled by molecules, or amount
of gas effused]
Rate A molar mass B
Ptotal = P1 + P2 + P3 + ...Pn PV = nRT =
Rate B molar mass A
Abbreviations Standard Conditions
atm = atmosphere 0°C = 273 K
mm Hg = millimeters of mercury 1.00 atm = 760.0 mm Hg = 76 cm Hg =101.325
torr = another name for mm Hg kPa = 101, 325 Pa = 29.9 in Hg
Pa = Pascal kPa = kilopascal
K = Kelvin
°C = degrees Celsius
Conversions Gas Law’s Equation Symbols
K = °C + 273 Subscript (1) = old condition or initial condition
o o
F = 1.8C + 32 Subscript (2) = new condition or final condition
Fo − 32 Temperature must be in Kelvins
Co = n = number of moles = grams/Molar mass
1.8 R = 8.31 L-kPa/ mol-K = 0.0821 L-atm/mol-K =
3
1 cm (cubic centimeter) = 1 mL (milliliter) 62.4 L-Torr/mol-K
1 dm3 (cubic decimeter) = 1 L (liter) = 1000 mL You must have a common set of units in the
problem
CHEMISTRY GAS LAW’S WORKSHEET
1. Convert the following
temperatures to K.
a) 104 C
b) -3 C
2. Convert the following
temperatures to C.
a) 67 K
b) 1671 K
3. A sample of nitrogen gas
has a volume of 478 cm3 and
a pressure of 104.1 kPa. What
volume would the gas occupy
at 88.2 kPa if the temperature
remains constant?
4. 8.98 dm3 of hydrogen gas
is collected at 38.8 °C. Find
the volume the gas will occupy
at -39.9 °C if the pressure
remains constant.
5. A sample of gas has a
volume of 215 cm3 at 23.5 °C
and 84.6 kPa. What volume
will the gas occupy at STP?
CHEMISTRY GAS LAW’S WORKSHEET
6. At a certain temperature,
molecules of methane gas,
CH4 have an average velocity
of 0.098 m/s. What is the
average velocity of carbon
dioxide molecules at this same
temperature?
7. Find the relative rate of
diffusion for the gases
chlorine, Cl2 and ethane,
C2H6.
8. 495 cm3 of oxygen gas and
877 cm3 of nitrogen gas, both
at 25.0 °C and 114.7 kPa, are
injected into an evacuated
536 cm3 flask. Find the total
pressure in the flask, assuming
the temperature remains
constant.
9. A sample of gas is
transferred from a 75 mL
vessel to a 500.0 mL vessel. If
the initial pressure of the gas is
145 atm and if the temperature
is held constant, what is the
pressure of the gas sample in
the 500.0 mL vessel?
10. A sample of gas occupies a
volume of 450.0 mL at 740
mm Hg and 16°C. Determine
the volume of this sample at
760 mm Hg and 37°C.
CHEMISTRY GAS LAW’S WORKSHEET
11. One mole of H2S gas
escapes from a container by
effusion in 77 seconds. How
long would it take one mole of
NH3 gas to escape from the
same container?
12. Convert a pressure of
0.0248 mm Hg to the
equivalent pressure in pascals
(Pa).
13. Air in a closed cylinder is
heated from 25°C to 36°C. If
the initial pressure is 3.80 atm,
what is the final pressure?
14. A bubble of helium gas has
a volume of 0.650 mL near the
bottom of a large aquarium
where the pressure is 1.54 atm
and the temperature is 12°C.
Determine the bubble’s volume
upon rising near the top where
the pressure is 1.01 atm and
16°C.
15. At what temperature
Celsius will 19.4 g of
molecular oxygen, O2, exert a
pressure of 1820 mm Hg in a
5.12 L cylinder?
CHEMISTRY GAS LAW’S WORKSHEET
16. A sample of nitrogen gas,
N2, is collected in a100 mL
container at a pressure of 688
mm Hg and a temperature of
565 °C. How many grams of
nitrogen gas are present in this
sample?
17. What is the pressure in
mm of Hg , of a gas mixture
that contains 1g of H2, and 8.0
g of Ar in a 3.0 L container at
27°C.
18. To what temperature must
32.0 ft3 of a gas at 2°C be
heated for it to occupy
1.00 x 102 ft3 at the same
pressure?
19. What is the pressure in atm
exerted by 2.48 moles of a gas
in a 250.0 mL container at
58°C?
20. Determine the molar mass
of a gas that has a density of
2.18 g/L at 66°C and 720 mm
Hg.
(Hint: the number of moles of
a substance is its
mass/molecular mass
and density is mass/volume.)
CHEMISTRY GAS LAW’S WORKSHEET
Key
1 a) 377 K
b) 270 K
2 a) -206 C
b) 1398 C
3. 564 cm3
4. 6.71 dm3
5. 165 cm3
6. 0.059 m/s
7. rate Cl2 : C2H6 = 0.650
8. 294 kPa
9. 21.8 atm
10.. 470 mL
11. 54 sec
12. 3.31 Pa
13. 3.94 atm
14. 1.00 mL
15. - 27°C
16. 0.0368 g
17. 4332 mm Hg
18. 586°C
19. 270 atm
20. 64 g/mole
You might also like
- CHM 101 Lecture Note-Gas LawsDocument11 pagesCHM 101 Lecture Note-Gas LawsMichael DanielsNo ratings yet
- Gas Laws Packet 2 ANSWERSDocument5 pagesGas Laws Packet 2 ANSWERSJoseph Turner100% (1)
- G484 Module 3 4.3.4 Ideal GasesDocument10 pagesG484 Module 3 4.3.4 Ideal GasesIgnatius AgustaNo ratings yet
- Worksheet 3 (Partial Pressures)Document2 pagesWorksheet 3 (Partial Pressures)Jose Ruben SortoNo ratings yet
- Mgo LabDocument3 pagesMgo Labapi-336093393No ratings yet
- Physical Chemistry 1 Prob SetDocument8 pagesPhysical Chemistry 1 Prob SetArrianne Jaye MataNo ratings yet
- Empirical and Molecular Formulae WorksheetDocument3 pagesEmpirical and Molecular Formulae WorksheetJohnclyde Ferry100% (1)
- Relationship Between The Number of Moles of A Gas and Its VolumeDocument15 pagesRelationship Between The Number of Moles of A Gas and Its VolumeFidree AzizNo ratings yet
- Worksheet 3.3Document2 pagesWorksheet 3.3khodijahaminNo ratings yet
- Chemical EquilibriumDocument11 pagesChemical EquilibriumYuaNo ratings yet
- Worksheet 6 Colligative PropertiesDocument7 pagesWorksheet 6 Colligative Propertiesani illuriNo ratings yet
- 13 Partial Pressures of GasesDocument6 pages13 Partial Pressures of GasesTanisha Marie100% (1)
- Practice estimating to find the best answer.: Δt 5 x rate = -Δ (B) 5 x 0.0243 M/s = - Δ (B) -0.12125~ - 0.122 M/sDocument10 pagesPractice estimating to find the best answer.: Δt 5 x rate = -Δ (B) 5 x 0.0243 M/s = - Δ (B) -0.12125~ - 0.122 M/sjeffrey XiaoNo ratings yet
- AP Chemistry - Hess's Law LabDocument3 pagesAP Chemistry - Hess's Law LabJonathan Chen83% (12)
- Chemistry Chapter 3: Nuclear Chemistry: The Nuclide of AtomDocument6 pagesChemistry Chapter 3: Nuclear Chemistry: The Nuclide of AtomBren Jousef BayhonNo ratings yet
- C1 - Basic Concepts of Chemistry - Solutions (v18) - HD - CLDocument20 pagesC1 - Basic Concepts of Chemistry - Solutions (v18) - HD - CLAashish DubeyNo ratings yet
- Stoichiometric ProblemsDocument2 pagesStoichiometric ProblemsJep Balisi PayusanNo ratings yet
- PW-049a-Limiting Reagents-01Document5 pagesPW-049a-Limiting Reagents-01JoHan Xyth RizaldoNo ratings yet
- 05 Gases Without AnswersDocument17 pages05 Gases Without Answersapi-287405319No ratings yet
- Thermo LabDocument3 pagesThermo LabMaggie GilliganNo ratings yet
- Algebraic Method To Balance Chemical EquationDocument3 pagesAlgebraic Method To Balance Chemical EquationBruce WalkerNo ratings yet
- CHE 160 Semester Review Zumdahl CH 5-7Document20 pagesCHE 160 Semester Review Zumdahl CH 5-7Kinal PatelNo ratings yet
- Unit 1 Part BDocument247 pagesUnit 1 Part BhairtNo ratings yet
- Characteristics of Chemical EquilibriumDocument43 pagesCharacteristics of Chemical Equilibriumpimpin1No ratings yet
- Gas Laws:: P V K VDocument18 pagesGas Laws:: P V K VFarah Zu'biNo ratings yet
- Gases & The Kinetic-Molecular TheoryDocument20 pagesGases & The Kinetic-Molecular TheoryAshley Marie ChildersNo ratings yet
- Chemistry 12: Solutions Manual Part ADocument44 pagesChemistry 12: Solutions Manual Part ADerrick JamesNo ratings yet
- Combined Gas Law WorksheetDocument1 pageCombined Gas Law Worksheetapi-483662721No ratings yet
- Section 3 EnergeticsDocument47 pagesSection 3 Energeticsapi-3734333No ratings yet
- Faraday's Law WorksheetDocument4 pagesFaraday's Law WorksheetBrianna MalcolmNo ratings yet
- Washburn, E.W. - International Critical Tables of Numerical Data, Physics, Chemistry and Technology (1926 - 1930 - 20, Knovel)Document1,055 pagesWashburn, E.W. - International Critical Tables of Numerical Data, Physics, Chemistry and Technology (1926 - 1930 - 20, Knovel)Guillermo Aquino100% (1)
- Combined Gas Law WS1Document2 pagesCombined Gas Law WS1Fe Pakias GullodNo ratings yet
- Apch3.1 Problems-Equilibrium AnsDocument5 pagesApch3.1 Problems-Equilibrium AnsQueenQiNo ratings yet
- Experiment 1Document9 pagesExperiment 1Rizza Mae RaferNo ratings yet
- Organic Chemistry (Alkane, Alkene and Alkyne)Document7 pagesOrganic Chemistry (Alkane, Alkene and Alkyne)Dazell Varron100% (1)
- 0 B264 D 01Document18 pages0 B264 D 01didikkrisNo ratings yet
- Ternary Phase Diagrams IntroductionDocument41 pagesTernary Phase Diagrams IntroductionGikiTopiNo ratings yet
- Chemistry 12: Solutions Manual Part ADocument38 pagesChemistry 12: Solutions Manual Part AhairtNo ratings yet
- TD WorksheetDocument4 pagesTD WorksheetrtyiookNo ratings yet
- Until Thermal Equilibrium Is Obtained.: Loss GainDocument5 pagesUntil Thermal Equilibrium Is Obtained.: Loss GainCharls DeimoyNo ratings yet
- AtKins Chapter 1Document57 pagesAtKins Chapter 1Ebook Download0% (1)
- Mole To Grams WorksheetDocument1 pageMole To Grams WorksheetJoseph GuerreroNo ratings yet
- Test Gas LawsDocument5 pagesTest Gas LawsCenando BodanioNo ratings yet
- Vanasthali Public School: Notes Chapter-1 Introduction To Chemical Reactions and EquationsDocument8 pagesVanasthali Public School: Notes Chapter-1 Introduction To Chemical Reactions and EquationsPlatinum Gaming Warrior100% (1)
- Rate LawsDocument20 pagesRate LawsReginal MoralesNo ratings yet
- Chemistry Past Paper Ch1.7Document16 pagesChemistry Past Paper Ch1.7Raymond ChanNo ratings yet
- Zumdahl Chemical Kinetics NotesDocument6 pagesZumdahl Chemical Kinetics NotesMalletNjonkemNo ratings yet
- Gas & Its LawDocument47 pagesGas & Its LawMerahouseNo ratings yet
- Practice Questions For Ch. 5: Name: - Class: - Date: - Id: ADocument23 pagesPractice Questions For Ch. 5: Name: - Class: - Date: - Id: APrem MehrotraNo ratings yet
- Chemistry 2 Answer KeyDocument8 pagesChemistry 2 Answer KeyMarielle BuesingNo ratings yet
- Overview of PhotosynthesDocument2 pagesOverview of Photosynthesapi-246212204No ratings yet
- Kinetics Practice Test 1Document25 pagesKinetics Practice Test 1noelNo ratings yet
- Thermochemistry (Important Notes)Document2 pagesThermochemistry (Important Notes)HenrySeowNo ratings yet
- 5 R DPBui QMF ZPUq Exc 6 JFDocument53 pages5 R DPBui QMF ZPUq Exc 6 JFBhfjsukNo ratings yet
- Enzymes: A Practical Introduction to Structure, Mechanism, and Data AnalysisFrom EverandEnzymes: A Practical Introduction to Structure, Mechanism, and Data AnalysisRating: 4 out of 5 stars4/5 (2)
- Chapter 5 Gases PDFDocument49 pagesChapter 5 Gases PDFAbou WalidNo ratings yet
- Chapter 10 Sept13Document57 pagesChapter 10 Sept13chandro57No ratings yet
- Leaching of NaCl Using Water As SolventDocument10 pagesLeaching of NaCl Using Water As SolventKarynne Bernardine Gerona SiclotNo ratings yet
- New VarplusCan - Reactive EnergyDocument40 pagesNew VarplusCan - Reactive EnergyAlfiansyah Dharma Setia Djaketra100% (1)
- Ch2 MC Practice Questions With AnswersDocument3 pagesCh2 MC Practice Questions With AnswersKit SzeNo ratings yet
- Moving Iron Type InstrumentsDocument6 pagesMoving Iron Type InstrumentsAbhishek KatreNo ratings yet
- 1983-Vibration Studies and Tests of Liquid Storage TanksDocument28 pages1983-Vibration Studies and Tests of Liquid Storage TanksbehzadNo ratings yet
- 2365 202 Sample Questions ADocument8 pages2365 202 Sample Questions AarchietomkinsNo ratings yet
- Chapter 16Document25 pagesChapter 16Pandu DoradlaNo ratings yet
- Chapter 1Document19 pagesChapter 1raghu ramNo ratings yet
- Experimental Physics SuperpositionDocument17 pagesExperimental Physics SuperpositionGrace CywrainNo ratings yet
- Quiz EDC UNIT 1Document4 pagesQuiz EDC UNIT 1kalpanadevi0% (1)
- BK 4 (CC 51400)Document9 pagesBK 4 (CC 51400)hasan arifNo ratings yet
- Schmidt Analysis (Updated 12-28-2011)Document5 pagesSchmidt Analysis (Updated 12-28-2011)أمير معروفNo ratings yet
- A Literature Review of Wind Turbines Icing ProblemsDocument8 pagesA Literature Review of Wind Turbines Icing Problemsrahul parmarNo ratings yet
- Tipler The Physics of Immortality Resurrection PDFDocument4 pagesTipler The Physics of Immortality Resurrection PDFKhanNo ratings yet
- 11 Chemistry Chapter 2 Assignment 1Document1 page11 Chemistry Chapter 2 Assignment 1Harman Singh0% (1)
- Chapter 3 (MEC681) - MHI PDFDocument71 pagesChapter 3 (MEC681) - MHI PDFمحمد فائزNo ratings yet
- Prediction of Ship Roll Damping - State of The ArtDocument87 pagesPrediction of Ship Roll Damping - State of The ArtVasil YordanovNo ratings yet
- Syllabus Civil KuDocument148 pagesSyllabus Civil KubashuNo ratings yet
- X.Wang Numericalsimulationofstick SlipbehavioursoftypicalfaultsDocument19 pagesX.Wang Numericalsimulationofstick SlipbehavioursoftypicalfaultsjeovanNo ratings yet
- 22.11.21 Champions SolutionsDocument21 pages22.11.21 Champions SolutionsDamon StarkNo ratings yet
- The Fabric of The CosmosDocument3 pagesThe Fabric of The CosmosTomas Særmark0% (3)
- Instructional Manual For Dead Weight Pressure Gauge TesterDocument6 pagesInstructional Manual For Dead Weight Pressure Gauge TesterRAVI BARTIANo ratings yet
- Code AsterDocument16 pagesCode AsterStefano MilaniNo ratings yet
- PTFE Specifications PDFDocument1 pagePTFE Specifications PDFAulia AnnaNo ratings yet
- Fenomenul WeisenbergDocument7 pagesFenomenul WeisenbergFLORINM_UCDCNo ratings yet
- MENG 302L Lab 4: Poisson's RatioDocument8 pagesMENG 302L Lab 4: Poisson's Ratiosudhir_kumar_33No ratings yet
- E2192-13 Guía Estándar para Dimensionamiento de Altura Plana de Defectos Por UltrasonidosDocument23 pagesE2192-13 Guía Estándar para Dimensionamiento de Altura Plana de Defectos Por Ultrasonidosfredy lopezNo ratings yet
- Eurocode 8-1-3Document18 pagesEurocode 8-1-3joaoNo ratings yet
- HKISO-2019-2020 Mock Heat SSFDocument10 pagesHKISO-2019-2020 Mock Heat SSFChesky Dominic Minas100% (5)
- Orthogonal CuttingDocument43 pagesOrthogonal CuttingMuhammad imranNo ratings yet