Professional Documents
Culture Documents
Demonstration of Quality Safety and Efficacy of Biological Products Subject To Changes in Their Manufacturing Process
Uploaded by
ЕвгенияOriginal Title
Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
Demonstration of Quality Safety and Efficacy of Biological Products Subject To Changes in Their Manufacturing Process
Uploaded by
ЕвгенияCopyright:
Available Formats
БЕЗОПАСНОСТЬ ЛС
Ïîäòâåðæäåíèå êà÷åñòâà, áåçîïàñíîñòè è ýôôåêòèâíîñòè
áèîëîãè÷åñêèõ ëåêàðñòâåííûõ ïðåïàðàòîâ
ïðè èçìåíåíèè òåõíîëîãèè èõ ïðîèçâîäñòâà
А. Н. ВАСИЛЬЕВ, Е. В. ГАВРИШИНА, Р. Р. НИЯЗОВ, А. А. СНЕГИРЕВА, В. К. АДОНИН
ФГБУ «Научный центр экспертизы средств медицинского применения» Минздрава России, Москва
Demonstration of Quality, Safety and Efficacy of Biological Products Subject
to Changes in Their Manufacturing Process
A. N. VASILIEV, E. V. GAVRISHINA, R. R. NIYAZOV, A. A. SNEGIREVA, V. K. ADONIN
Scientific Centre for Expertise of Medical Application Products, Moscow
Îáåñïå÷åíèå êà÷åñòâà, áåçîïàñíîñòè è ýôôåêòèâíîñòè ëåêàðñòâåííûõ ïðåïàðàòîâ, ïîñòóïàþùèõ â îáîðîò Ðîññèè, ÿâëÿ-
åòñÿ îäíèì èç ïðèîðèòåòíûõ íàïðàâëåíèé, òðåáóþùèõ ãîñóäàðñòâåííîãî êîíòðîëÿ è íàäçîðà. Ïðè èçìåíåíèè òåõíîëîãèè
ïðîèçâîäñòâà áèîëîãè÷åñêèõ ëåêàðñòâåííûõ ïðåïàðàòîâ âîçíèêàåò íåîáõîäèìîñòü ïîäòâåðæäåíèÿ îòñóòñòâèÿ âëèÿíèÿ íà
áåçîïàñíîñòü è ýôôåêòèâíîñòü, âûðàæàþùåãîñÿ â ñîïîñòàâèìîñòè ïðåïàðàòîâ äî è ïîñëå èçìåíåíèÿ. Â ñâÿçè ñ îòñóòñòâè-
åì íåîáõîäèìîãî ïðàâîâîãî ðåãóëèðîâàíèÿ êàê íèêîãäà àêòóàëüíà íåîáõîäèìîñòü ãàðìîíèçàöèè ñóùåñòâóþùåãî ðîññèé-
ñêîãî çàêîíîäàòåëüñòâà, ïîäõîäîâ ê ïðåä- è ïîñòðåãèñòðàöèîííîé ðàçðàáîòêå è ýêñïåðòèçå áèîëîãè÷åñêèõ ëåêàðñòâåííûõ
ïðåïàðàòîâ ñ ìåæäóíàðîäíûì îïûòîì îáåñïå÷åíèÿ èõ êà÷åñòâà, áåçîïàñíîñòè è ýôôåêòèâíîñòè.
Êëþ÷åâûå ñëîâà: áèîëîãè÷åñêèå ëåêàðñòâåííûå ïðåïàðàòû, ñîïîñòàâèìîñòü, äîêëèíè÷åñêèå è êëèíè÷åñêèå èññëåäîâàíèÿ,
èçìåíåíèå òåõíîëîãèè ïðîèçâîäñòâà, èçìåíåíèå ïðîöåññà ïðîèçâîäñòâà, áèîàíàëîãè.
Ensuring quality, safety and efficacy of the medicinal products placed on the market of the Russian Federation constitutes the area
that requires strict regulation. When changes are made to the manufacturing process, the manufacturer generally needs to evaluate
the relevant quality attributes of the product to demonstrate that modifications did not occur that would adversely impact the safety
and efficacy of the drug. Where there is the lack of a sound legal basis, there is a need in harmonization of current Russian legisla-
tion with international and European rules governing medicinal product for human use to ensure quality, safety and efficacy thereof.
Key words: biological medicinal products, comparability, pre-clinical and clinical trials, manufacturing process, changes in manu-
facturing process, biosimilars.
Ââåäåíèå íîãî âëèÿíèÿ íà áåçîïàñíîñòü è ýôôåêòèâíîñòü.
Îäíàêî ìîãóò ïîòðåáîâàòüñÿ äîïîëíèòåëüíûå èñ-
Ïðîèçâîäèòåëè áèîëîãè÷åñêèõ ëåêàðñòâåííûõ
ñëåäîâàíèÿ (äîêëèíè÷åñêèå, êëèíè÷åñêèå è ôàð-
ïðåïàðàòîâ ÷àñòî ñòàëêèâàþòñÿ ñ íåîáõîäèìîñòüþ
ìàêîíàäçîðíûå) [1]. Ïðè âûñîêîé àíàëîãè÷íîñòè
èçìåíåíèÿ ïðîöåññà ïðîèçâîäñòâà ëåêàðñòâåííûõ
ïîêàçàòåëåé êà÷åñòâà äî è ïîñëå èçìåíåíèé ïðî-
ñðåäñòâ. Èçìåíåíèÿ âíîñÿòñÿ íà ðàçëè÷íûõ ýòàïàõ
öåññà ïðîèçâîäñòâà è îòñóòñòâèè íåæåëàòåëüíîãî
êàê â õîäå ðàçðàáîòêè, òàê è ïîñëå ãîñóäàðñòâåííîé
âëèÿíèÿ íà áåçîïàñíîñòü è (èëè) ýôôåêòèâíîñòü,
ðåãèñòðàöèè. Ïðè÷èíàìè òàêèõ èçìåíåíèé ìîãóò
âêëþ÷àÿ èììóíîãåííîñòü, ïðîèçâîäèòåëü äåëàåò
ñëóæèòü: óñîâåðøåíñòâîâàíèå ïðîöåññà ïðîèçâîä-
âûâîä î ñîïîñòàâèìîñòè ìåæäó èñõîäíûì è èçìå-
ñòâà, óâåëè÷åíèå ìàñøòàáà ïðîèçâîäñòâà, ïîâûøå-
í¸ííûì ïðåïàðàòîì.
íèå ñòàáèëüíîñòè ëåêàðñòâåííîãî ñðåäñòâà è ñîîò-
 íàñòîÿùåå âðåìÿ äî 10% îáú¸ìà ìèðîâîãî
âåòñòâèå èçìåí¸ííûì ðåãóëÿòîðíûì òðåáîâàíèÿì
ôàðìàöåâòè÷åñêîãî ðûíêà ïðèõîäèòñÿ íà áèîòåõ-
[1]. Ïðè èçìåíåíèè ïðîöåññà ïðîèçâîäñòâà ïðîèç-
íîëîãè÷åñêèå ïðåïàðàòû; èõ äîëÿ ïîñòîÿííî óâåëè-
âîäèòåëè, êàê ïðàâèëî, èçó÷àþò ñîîòâåòñòâóþùèå
÷èâàåòñÿ è, ïî îöåíêå ðÿäà ñïåöèàëèñòîâ, ê 2015 ã.
ïîêàçàòåëè êà÷åñòâà ôàðìàöåâòè÷åñêîé ñóáñòàí-
ìîæåò äîñòè÷ü 50% [2—4]. Ñåãîäíÿ óæå áîëåå 150
öèè è (èëè) ãîòîâîãî ïðåïàðàòà, ÷òîáû ïîäòâåð-
áèîòåõíîëîãè÷åñêèõ ëåêàðñòâåííûõ ïðåïàðàòîâ (84
äèòü, ÷òî èçìåíåíèÿ íå îêàçûâàþò íåáëàãîïðèÿò-
áåëêà) øèðîêî ïðèìåíÿþòñÿ äëÿ ëå÷åíèÿ áîëüíûõ
© Êîëëåêòèâ àâòîðîâ, 2013 âî âñåì ìèðå. Ýòî èíòåðôåðîíû, ýðèòðîïîýòèíû,
ìîíîêëîíàëüíûå àíòèòåëà, èíñóëèíû, íèçêîìîëå-
Àäðåñ äëÿ êîððåñïîíäåíöèè: 127051, Ðîññèÿ, Ìîñêâà, Ïåòðîâñêèé
áóëüâàð, ä. 8. ÍÖÝÑÌÏ
êóëÿðíûå ãåïàðèíû, ãîðìîí ðîñòà ÷åëîâåêà, êîëî-
АНТИБИОТИКИ И ХИМИОТЕРАПИЯ, 2013, 58; 9—10 45
íèåñòèìóëèðóþùèé ôàêòîð, ôàêòîðû ñâåðòûâàíèÿ ìèññèè (EC) ¹ 1234/2008 îò 24 íîÿáðÿ 2008 ã. îá
êðîâè, òðîìáîëèòèêè [5—7]. Îäíàêî íà ñåãîäíÿø- ýêñïåðòèçå èçìåíåíèé óñëîâèé ðåãèñòðàöèè ëå-
íèé äåíü ïîä îïðåäåëåíèå áèîëîãè÷åñêîãî ëåêàðñò- êàðñòâåííûõ ïðåïàðàòîâ äëÿ ìåäèöèíñêîãî è âå-
âåííîãî ïðåïàðàòà ïîäïàäàþò íå òîëüêî áèîòåõíî- òåðèíàðíîãî ïðèìåíåíèÿ ñ èçìåíåíèÿìè [11, 12]
ëîãè÷åñêèå, íî è äðóãèå ëåêàðñòâåííûå ïðåïàðàòû, è ïîäçàêîííûõ àêòîâ, ïðèíÿòûõ â åãî ðàçâèòèå
ïîëó÷åííûå èç áèîëîãè÷åñêèõ èñòî÷íèêîâ, íàïðè- [13]. Ñëåäóåò îòìåòèòü, ÷òî âñå èçìåíåíèÿ òåõíî-
ìåð, ãåïàðèíû, ïàíêðåàòèíû, ïðî÷èå íåðåêîìáè- ëîãèè ïðîèçâîäñòâà áèîëîãè÷åñêîãî ëåêàðñòâåí-
íàíòíûå ôàðìàöåâòè÷åñêèå ñóáñòàíöèè [8]. íîãî ñðåäñòâà (êàê ôàðìàöåâòè÷åñêîé ñóáñòàí-
 íàñòîÿùåå âðåìÿ ôîðìàëüíûå ïîäõîäû ê öèè, òàê è ãîòîâîãî ïðåïàðàòà) îòíîñÿòñÿ â ÅÑ ê
ïîäòâåðæäåíèþ ñîïîñòàâèìîñòè ïî êà÷åñòâó, áå- èçìåíåíèÿì II òèïà, ò.å. èçìåíåíèÿì, ñïîñîáíûì
çîïàñíîñòè è ýôôåêòèâíîñòè áèîòåõíîëîãè÷åñ- çíà÷èìî ïîâëèÿòü íà êà÷åñòâî, áåçîïàñíîñòü èëè
êèõ/áèîëîãè÷åñêèõ ëåêàðñòâåííûõ ïðåïàðàòîâ, ýôôåêòèâíîñòü ëåêàðñòâåííîãî ïðåïàðàòà. Ïî-
ïîäâåðãøèõñÿ èçìåíåíèþ òåõíîëîãèè èõ ïðîèç- äîáíûå èçìåíåíèÿ âñåãäà òðåáóþò ïðîâåäåíèÿ
âîäñòâà, â Ðîññèéñêîé Ôåäåðàöèè îòñóòñòâóþò. ñðàâíèòåëüíûõ èñïûòàíèé, à èíîãäà è äîêëèíè÷å-
Ïîýòîìó ãàðìîíèçàöèÿ ñóùåñòâóþùèõ ïîäõîäîâ ñêèõ è êëèíè÷åñêèõ èññëåäîâàíèé [1, 11, 13].
ê ïðåä- è ïîñòðåãèñòðàöèîííîé ðàçðàáîòêå è ýêñ-
ïåðòèçå áèîëîãè÷åñêèõ ëåêàðñòâåííûõ ïðåïàðà- Ïðè÷èíà íåîáõîäèìîñòè
òîâ ñ ìåæäóíàðîäíûì îïûòîì îáåñïå÷åíèÿ èõ êà-
÷åñòâà, áåçîïàñíîñòè è ýôôåêòèâíîñòè ÿâëÿåòñÿ
øèðîêîìàñøòàáíûõ èñïûòàíèé
ïåðâîñòåïåííîé çàäà÷åé. è èññëåäîâàíèé
Áèîëîãè÷åñêèå ëåêàðñòâåííûå ñðåäñòâà èìåþò
Çàêîíîäàòåëüíàÿ ðåãëàìåíòàöèÿ ñëîæíîå ìîëåêóëÿðíîå ñòðîåíèå (ðåêîìáèíàíòíàÿ
ÄÍÊ, èììóíîëîãè÷åñêèå ñðåäñòâà, ëåêàðñòâåííûå
 Ôåäåðàëüíîì çàêîíå îò 12 àïðåëÿ 2010 ã. ¹ 61-ÔÇ ïðåïàðàòû, ïîëó÷åííûå èç êðîâè èëè ïëàçìû,
«Îá îáðàùåíèè ëåêàðñòâåííûõ ñðåäñòâ» (äàëåå — ãåííàÿ è êëåòî÷íàÿ òåðàïèÿ è äð.), îíè çàâåäîìî
Çàêîí) ïîðÿäîê âíåñåíèÿ èçìåíåíèé â ðåãèñòðà- áîëåå ñëîæíû ïî ñòðîåíèþ, ÷åì áîëüøèíñòâî ëå-
öèîííîå äîñüå áèîëîãè÷åñêèõ ïðåïàðàòîâ, à ðàâ- êàðñòâåííûõ ïðåïàðàòîâ, ñîäåðæàùèõ ôàðìàöåâ-
íî êàê è îïðåäåëåíèå áèîëîãè÷åñêèõ ïðåïàðàòîâ òè÷åñêèå ñóáñòàíöèè, ïîëó÷åííûå ñ ïîìîùüþ õè-
íå ïðèâîäèòñÿ.  ñòàòüå 30 Çàêîíà ïðèâîäèòñÿ ìè÷åñêîãî ñèíòåçà. Áèîëîãè÷åñêèå ñóáñòàíöèè
êðàòêèé ïîðÿäîê âíåñåíèÿ èçìåíåíèé â ðåãèñò- õàðàêòåðèçóþòñÿ òð¸õìåðíîé ñòðóêòóðîé, ìíîæå-
ðàöèîííîå äîñüå âñåõ ëåêàðñòâåííûõ ïðåïàðàòîâ, ñòâîì êèñëîòíî-îñíîâíûõ âàðèàíòîâ, ïîñòòðàíñ-
íî îòñóòñòâóþò íîðìû, îáÿçûâàþùèå âëàäåëüöåâ ëÿöèîííûìè ìîäèôèêàöèÿìè (íàïðèìåð, ãëèêî-
ðåãèñòðàöèîííûõ óäîñòîâåðåíèé âíîñèòü èõ [9]. çèëèðîâàíèåì) è ò. ï., êîòîðûå èãðàþò âàæíóþ
 ïðîòèâîïîëîæíîñòü ýòîìó, ñòàòüåé 23 Äè- ðîëü â ðåàëèçàöèè èõ áèîëîãè÷åñêèõ ôóíêöèé.
ðåêòèâû 2001/83/EC (ÿâëÿþùåéñÿ îñíîâîïîëàãà- Èçìåíåíèå ïðîöåññà ïðîèçâîäñòâà áèîëîãè÷åñ-
þùèì íîðìàòèâíî-ïðàâîâûì àêòîì, ðåãóëèðóþ- êèõ ïðåïàðàòîâ ìîæåò, â ÷èñëå ïðî÷åãî, ïðèâíåñ-
ùèì îáðàùåíèå ëåêàðñòâåííûõ ïðåïàðàòîâ â ÅÑ) òè èçìåíåíèÿ â ýòè õàðàêòåðèñòèêè. Ïåðâîíà÷àëü-
÷¸òêî ðåãëàìåíòèðóåòñÿ îáÿçàííîñòü âëàäåëüöà íî îíè ìîãóò ðàññìàòðèâàòüñÿ êàê íåçíà÷èìûå èëè
ðåãèñòðàöèîííîãî óäîñòîâåðåíèÿ âíîñèòü èçìå- äàæå íå îáíàðóæèâàòüñÿ íà ýòàïå àíàëèòè÷åñêèõ è
íåíèÿ â ðåãèñòðàöèîííîå äîñüå [10]: áèîëîãè÷åñêèõ èñïûòàíèé, îäíàêî âïîñëåäñòâèè
1. Ïîñëå ðåãèñòðàöèè âëàäåëåö ðåãèñòðàöè- ýòè èçìåíåíèÿ ìîãóò ïîâëèÿòü íà ïðîôèëü áåçî-
îííîãî óäîñòîâåðåíèÿ â îòíîøåíèè ìåòîäîâ ïðî- ïàñíîñòè è ýôôåêòèâíîñòè áèîëîãè÷åñêèõ ëåêàð-
èçâîäñòâà è êîíòðîëÿ… äîëæåí ïðèíèìàòü âî ñòâåííûõ ïðåïàðàòîâ, ïîäâåðãøèõñÿ èçìåíåíèþ
âíèìàíèå íàó÷íûé è òåõíè÷åñêèé ïðîãðåññ è òåõíîëîãèè èõ ïðîèçâîäñòâà [14—18].
âíîñèòü èçìåíåíèÿ, êîòîðûå ìîãóò ïîòðåáîâàòüñÿ Ñàìî îïðåäåëåíèå áèîëîãè÷åñêîãî ïðåïàðà-
â öåëÿõ ñîîòâåòñòâèÿ ïðîèçâîäñòâà è êîíòðîëÿ òà ïîäðàçóìåâàåò ïðîâåäåíèå áîëåå ñëîæíûõ, ïî
îáùåïðèíÿòûì íàó÷íûì ìåòîäàì. ñðàâíåíèþ ñ âåùåñòâàìè, ïîëó÷åííûìè ïóò¸ì
Òàêèå èçìåíåíèÿ äîëæíû óòâåðæäàòüñÿ óïîë- õèìè÷åñêîãî ñèíòåçà, èñïûòàíèé [10]: áèîëîãè-
íîìî÷åííûì îðãàíîì çàèíòåðåñîâàííîé ñòðàíû- ÷åñêèé ëåêàðñòâåííûé ïðåïàðàò — ýòî ëåêàðñò-
÷ëåíà. âåííûé ïðåïàðàò, ñîäåðæàùèé áèîëîãè÷åñêóþ
2. Âëàäåëåö ðåãèñòðàöèîííîãî óäîñòîâåðå- ôàðìàöåâòè÷åñêóþ ñóáñòàíöèþ. Áèîëîãè÷åñêàÿ
íèÿ îáÿçàí íåìåäëåííî ñîîáùàòü íàöèîíàëüíîìó ôàðìàöåâòè÷åñêàÿ ñóáñòàíöèÿ — ýòî ñóáñòàí-
óïîëíîìî÷åííîìó îðãàíó îáî âñåõ íîâûõ ñâåäå- öèÿ, ïðîäóöèðóåìàÿ áèîëîãè÷åñêèì èñòî÷íèêîì
íèÿõ, êîòîðûå ìîãóò ïîòðåáîâàòü èçìåíåíèÿ äî- èëè ïîëó÷àåìàÿ èç íåãî, òðåáóþùàÿ ïðîâåäåíèÿ
êóìåíòîâ è äàííûõ, ñîäåðæàùèõñÿ â ðåãèñòðàöè- êîìïëåêñà ôèçèêî-õèìè÷åñêèõ è áèîëîãè÷åñêèõ èñ-
îííîì äîñüå. ïûòàíèé â öåëÿõ îïèñàíèÿ å¸ ñâîéñòâ, ïîäòâåðæ-
Âíåñåíèå èçìåíåíèé â ðåãèñòðàöèîííîå äîñüå äåíèÿ è êîíòðîëÿ êà÷åñòâà, à òàêæå ïðîöåññà å¸
ïîäðîáíî óðåãóëèðîâàíî Ïîñòàíîâëåíèåì Êî- ïðîèçâîäñòâà è åãî êîíòðîëÿ.
46 АНТИБИОТИКИ И ХИМИОТЕРАПИЯ, 2013, 58; 9—10
БЕЗОПАСНОСТЬ ЛС
Таблица 1. Виды изменений биологических лекарственных препаратов
Ôàðìàöåâòè÷åñêàÿ ñóáñòàíöèÿ
1. Èçìåíåíèå ïðîèçâîäèòåëÿ èñõîäíîãî ìàòåðèàëà/ðåàêòèâà/ 8. Èçìåíåíèå ïàðàìåòðîâ ñïåöèôèêàöèè è (èëè)
ïðîìåæóòî÷íîãî ïðîäóêòà, èñïîëüçóåìîãî â ïðîöåññå êðèòåðèåâ ïðèåìëåìîñòè ïåðâè÷íîé óïàêîâêè
ïðîèçâîäñòâà ôàðìàöåâòè÷åñêîé ñóáñòàíöèè, èëè èçìåíåíèå ôàðìàöåâòè÷åñêîé ñóáñòàíöèè
ïðîèçâîäèòåëÿ ôàðìàöåâòè÷åñêîé ñóáñòàíöèè (âêëþ÷àÿ ïëîùàäêè
âûïóñêàþùåãî êîíòðîëÿ êà÷åñòâà) ôàðìàöåâòè÷åñêîé ñóáñòàíöèè
2. Èçìåíåíèÿ ïðîöåññà ïðîèçâîäñòâà ôàðìàöåâòè÷åñêîé ñóáñòàíöèè 9. Èçìåíåíèå àíàëèòè÷åñêîé ìåòîäèêè èñïûòàíèÿ
ïåðâè÷íîé óïàêîâêè ôàðìàöåâòè÷åñêîé ñóáñòàíöèè
3. Èçìåíåíèå ðàçìåðà ñåðèè (âêëþ÷àÿ äèàïàçîíû ðàçìåðà ñåðèè) 10. Èçìåíåíèå ïåðèîäà ïîâòîðíîãî èñïûòàíèÿ/ïåðèîäà
ôàðìàöåâòè÷åñêîé ñóáñòàíöèè èëè ïðîìåæóòî÷íîãî ïðîäóêòà õðàíåíèÿ èëè óñëîâèé õðàíåíèÿ
ôàðìàöåâòè÷åñêîé ñóáñòàíöèè
4. Èçìåíåíèå âíóòðèïðîèçâîäñòâåííûõ èñïûòàíèé 11. Ââåäåíèå íîâîãî ïðîåêòíîãî ïîëÿ èëè ðàñøèðåíèå
èëè êðèòåðèåâ ïðèåìëåìîñòè, èñïîëüçóþùèõñÿ îäîáðåííîãî ïðîåêòíîãî ïîëÿ ôàðìàöåâòè÷åñêîé
ïðè ïðîèçâîäñòâå ôàðìàöåâòè÷åñêîé ñóáñòàíöèè ñóáñòàíöèè
5. Èçìåíåíèå ïàðàìåòðîâ ñïåöèôèêàöèè è (èëè) êðèòåðèåâ 12. Ââåäåíèå ïîñòðåãèñòðàöèîííîãî ïðîòîêîëà
ïðèåìëåìîñòè ôàðìàöåâòè÷åñêîé ñóáñòàíöèè, óïðàâëåíèÿ èçìåíåíèÿìè, çàòðàãèâàþùèìè
èñõîäíîãî ìàòåðèàëà/ïðîìåæóòî÷íîãî ïðîäóêòà/ðåàêòèâà, ôàðìàöåâòè÷åñêóþ ñóáñòàíöèþ
èñïîëüçóåìûõ â ïðîöåññå ïðîèçâîäñòâà
ôàðìàöåâòè÷åñêîé ñóáñòàíöèè
6. Èçìåíåíèå àíàëèòè÷åñêîé ìåòîäèêè èñïûòàíèÿ 13. Èçìåíåíèå âíóòðèïðîèçâîäñòâåííûõ èñïûòàíèé
ôàðìàöåâòè÷åñêîé ñóáñòàíöèè èëè èñõîäíîãî ìàòåðèàëà/ èëè êðèòåðèåâ ïðèåìëåìîñòè, èñïîëüçóþùèõñÿ
ïðîìåæóòî÷íîãî ïðîäóêòà/ðåàêòèâà, èñïîëüçóåìûõ â ïðîöåññå ïðè ïðîèçâîäñòâå ôàðìàöåâòè÷åñêîé ñóáñòàíöèè
ïðîèçâîäñòâà ôàðìàöåâòè÷åñêîé ñóáñòàíöèè
7. Èçìåíåíèå ïåðâè÷íîé óïàêîâêè ôàðìàöåâòè÷åñêîé ñóáñòàíöèè
Âñïîìîãàòåëüíûå âåùåñòâà
1. Èçìåíåíèå ñîñòàâà (âñïîìîãàòåëüíûõ âåùåñòâ) ãîòîâîãî ïðåïàðàòà 4. Èçìåíåíèå àíàëèòè÷åñêîé ìåòîäèêè èñïûòàíèÿ
âñïîìîãàòåëüíîãî âåùåñòâà
2. Èçìåíåíèå êîíöåíòðàöèè îäíîäîçîâîãî, ïîëíîñòüþ ââîäèìîãî 5. Èçìåíåíèå èñòî÷íèêà âñïîìîãàòåëüíîãî âåùåñòâà
ïàðåíòåðàëüíîãî ïðåïàðàòà ïðè íåèçìåííîñòè ñîäåðæàíèÿ èëè ðåàêòèâà ñ ðèñêîì òðàíñìèññèâíîé ãóá÷àòîé
ôàðìàöåâòè÷åñêîé ñóáñòàíöèè íà åäèíèöó äîçû (ò.å. äîçèðîâêè) ýíöåôàëîïàòèè
3. Èçìåíåíèå ïàðàìåòðîâ ñïåöèôèêàöèè è (èëè) êðèòåðèåâ 6. Èçìåíåíèå ñèíòåçà èëè ïîëó÷åíèÿ íåôàðìàêîïåéíîãî
ïðèåìëåìîñòè âñïîìîãàòåëüíîãî âåùåñòâà âñïîìîãàòåëüíîãî âåùåñòâà
(åñëè îïèñàí â ðåãèñòðàöèîííîì äîñüå)
Ãîòîâûé ïðåïàðàò
1. Çàìåíà èëè äîáàâëåíèå íîâîé ïðîèçâîäñòâåííîé ïëîùàäêè 8. Èçìåíåíèå àíàëèòè÷åñêîé ìåòîäèêè èñïûòàíèÿ
äëÿ ÷àñòè èëè âñåõ ïðîöåññîâ ïðîèçâîäñòâà ãîòîâîãî ïðåïàðàòà; ïåðâè÷íîé óïàêîâêè ãîòîâîãî ïðåïàðàòà
2. Èçìåíåíèå ñîãëàøåíèé î âûïóñêå ñåðèé 9. Èçìåíåíèå ôîðìû èëè ðàçìåðîâ êîíòåéíåðà
è êîíòðîëÿ êà÷åñòâà ãîòîâîãî ïðåïàðàòà èëè óêóïîðêè (ïåðâè÷íîé óïàêîâêè)
3. Èçìåíåíèå ðàçìåðà ñåðèè (âêëþ÷àÿ äèàïàçîíû ðàçìåðà ñåðèè) 10. Èçìåíåíèå ðàçìåðà óïàêîâêè ãîòîâîãî ïðåïàðàòà
ãîòîâîãî ïðåïàðàòà
4. Èçìåíåíèå ïàðàìåòðîâ ñïåöèôèêàöèè è (èëè) êðèòåðèåâ 11. Èçìåíåíèå ïîñòàâùèêà êîìïîíåíòîâ óïàêîâêè
ïðèåìëåìîñòè ãîòîâîãî ïðåïàðàòà èëè óñòðîéñòâà (åñëè óêàçàíî â äîñüå)
5. Èçìåíåíèå àíàëèòè÷åñêîé ìåòîäèêè èñïûòàíèÿ 12. Èçìåíåíèå ñðîêà ãîäíîñòè èëè óñëîâèé õðàíåíèÿ
ãîòîâîãî ïðåïàðàòà ãîòîâîãî ïðåïàðàòà
6. Èçìåíåíèå ïåðâè÷íîé óïàêîâêè ãîòîâîãî ïðåïàðàòà 13. Ââåäåíèå ïîñòðåãèñòðàöèîííîãî ïðîòîêîëà
óïðàâëåíèÿ èçìåíåíèÿìè
7. Èçìåíåíèå ïàðàìåòðîâ ñïåöèôèêàöèè è (èëè) êðèòåðèåâ 14. Èçìåíåíèå ïðîöåññà ïðîèçâîäñòâà
ïðèåìëåìîñòè ïåðâè÷íîé óïàêîâêè ãîòîâîãî ïðåïàðàòà ãîòîâîãî ïðåïàðàòà
Âèäû è ýòàïû èçìåíåíèé êàðñòâåííîãî ïðåïàðàòà. Íåñìîòðÿ íà íåèçáåæ-
íóþ íåêîòîðóþ âàðèàöèþ ïîêàçàòåëåé êà÷åñòâà,
 Ðóêîâîäñòâå ïî ïîäðîáíîé êàòåãîðèçàöèè îòñóòñòâèå êëèíè÷åñêè çíà÷èìûõ ðàçëè÷èé ìåæ-
èçìåíåíèé, âíîñèìûõ â óñëîâèÿ ðåãèñòðàöèè ëå- äó äâóìÿ ïðåïàðàòàìè ïîçâîëÿåò ñäåëàòü âûâîä,
êàðñòâåííûõ ïðåïàðàòîâ äëÿ ìåäèöèíñêîãî ïðè- ÷òî îíè ñîïîñòàâèìû. Ñîïîñòàâèìîñòü — ýòî
ìåíåíèÿ è ëåêàðñòâåííûõ ïðåïàðàòîâ äëÿ âåòåðè- âàæíàÿ êîíöåïöèÿ, ïðèìåíÿåìàÿ â îòíîøåíèè
íàðíîãî ïðèìåíåíèÿ [13], îïèñàíû ðàçëè÷íûå âñåõ ëåêàðñòâåííûõ ïðåïàðàòîâ (áèîëîãè÷åñêèõ,
èçìåíåíèÿ, êîòîðûì ìîæåò ïîäâåðãàòüñÿ áèîëî- áèîàíàëîãîâ, ïðåïàðàòîâ, ïîëó÷åííûõ õèìè÷åñ-
ãè÷åñêèé ëåêàðñòâåííûé ïðåïàðàò íà ðàçëè÷íûõ êèì ïóò¸ì). Ïðè ýòîì ñîïîñòàâèìîñòü ëåêàðñò-
ýòàïàõ åãî ïðîèçâîäñòâà (òàáë. 1). âåííûõ ïðåïàðàòîâ, ôàðìàöåâòè÷åñêàÿ ñóáñòàí-
öèÿ êîòîðûõ ïîëó÷åíà õèìè÷åñêèì ïóò¸ì,
Ñîïîñòàâèìîñòü è ñòðàòåãèÿ èìåíóåòñÿ áèîýêâèâàëåíòíîñòüþ. Ýòîò âûâîä
å¸ ïîäòâåðæäåíèÿ ìîæíî ñäåëàòü, èñõîäÿ èç îïðåäåëåíèé áèîýêâè-
Ñîïîñòàâèìîñòü — ýòî ïîíÿòèå, îòíîñÿùååñÿ, âàëåíòíîñòè, äàííûõ â çàêîíîäàòåëüñòâå çàðó-
â ïåðâóþ î÷åðåäü, ê êëèíè÷åñêèì ñâîéñòâàì ëå- áåæíûõ ñòðàí [10, 19, 20].
АНТИБИОТИКИ И ХИМИОТЕРАПИЯ, 2013, 58; 9—10 47
 öåëÿõ îïðåäåëåíèÿ âëèÿíèÿ èçìåíåíèé òåõ- äî è ïîñëå èçìåíåíèé: íå èñêëþ÷åíî âîçìîæíîå
íîëîãèè ïðîèçâîäñòâà íà êëèíè÷åñêèå ñâîéñòâà íåæåëàòåëüíîå âëèÿíèÿ íà ïðîôèëè áåçîïàñíîñ-
áèîëîãè÷åñêîãî ïðåïàðàòà íåîáõîäèìî ïðîâåñòè òè è ýôôåêòèâíîñòè.  ýòîì ñëó÷àå ñáîð è àíàëèç
òùàòåëüíûé àíàëèç âñåõ ïðåäïîëàãàåìûõ ïîñëåä- äîïîëíèòåëüíûõ äàííûõ î ïîêàçàòåëÿõ êà÷åñòâà,
ñòâèé òàêîãî èçìåíåíèÿ. Ïðîâîäÿ òàêîé àíàëèç, ñêîðåå âñåãî, íå ïîçâîëèò óñòàíîâèòü ñîïîñòàâè-
ïðîèçâîäèòåëü äîëæåí âûáðàòü îïðåäåë¸ííûå ìîñòü ëåêàðñòâåííîãî ñðåäñòâà äî è ïîñëå èçìå-
êðèòåðèè, ïîçâîëÿþùèå çàäàòü ãðàíèöû âûñîêî- íåíèé. Ïðîèçâîäèòåëü äîëæåí ïðîâåñòè äîêëè-
àíàëîãè÷íîìó ëåêàðñòâåííîìó ñðåäñòâó, ïîäâåðã- íè÷åñêèå è (èëè) êëèíè÷åñêèå èññëåäîâàíèÿ;
øåìóñÿ èçìåíåíèþ òåõíîëîãèè ïðîèçâîäñòâà. • ðàçëè÷èÿ ìåæäó ïîêàçàòåëÿìè êà÷åñòâà
Íåîáõîäèìî, êàê ïðàâèëî, ñîáðàòü äàííûå ïî êà- ñòîëü çíà÷èòåëüíû, ÷òî ëåêàðñòâåííûå ñðåäñòâà
÷åñòâó ëåêàðñòâåííîãî ñðåäñòâà äî è ïîñëå èçìå- íå ÿâëÿþòñÿ âûñîêîàíàëîãè÷íûìè è, òàêèì îáðà-
íåíèÿ òåõíîëîãèè ïðîèçâîäñòâà, çàòåì ïðîâåñòè çîì, íå ñîïîñòàâèìû. Òàêîé ðåçóëüòàò òðåáóåò
ñðàâíåíèå, çàêëþ÷àþùååñÿ â îáúåäèíåíèè è àíà- èíîãî ïîäõîäà.
ëèçå âñåõ èìåþùèõñÿ äàííûõ, íàïðèìåð, ñòàí- Îáú¸ì äàííûõ, íåîáõîäèìûõ äëÿ àíàëèçà è
äàðòíîãî àíàëèçà ñåðèé, âíóòðèïðîèçâîäñòâåí- îáîñíîâàíèÿ íåîáõîäèìîñòè ïðîâåäåíèÿ òåõ èëè
íîãî êîíòðîëÿ, âàëèäàöèè/îöåíêè ïðîöåññà èíûõ èññëåäîâàíèé â ðàìêàõ ïðîâåäåíèÿ ïîä-
ïðîèçâîäñòâà, îïèñàíèÿ ñâîéñòâ è ñòàáèëüíîñòè. òâåðæäåíèÿ ñîïîñòàâèìîñòè çàâèñèò îò ñëîæíî-
Ñðàâíåíèå ðåçóëüòàòîâ ñ çàðàíåå îãîâîð¸ííûìè ñòè ñòðîåíèÿ ìîëåêóëû ëåêàðñòâåííîãî ïðåïà-
êðèòåðèÿìè äîëæíî ïîçâîëèòü îáúåêòèâíî îöå- ðàòà. Äëÿ ëåêàðñòâåííûõ ïðåïàðàòîâ ñ ìàëûì
íèòü, ñîïîñòàâèìû ëè ñâîéñòâà ëåêàðñòâåííîãî ðàçìåðîì ìîëåêóëû, êàê ïðàâèëî, äîñòàòî÷íî
ïðåïàðàòà äî è ïîñëå èçìåíåíèÿ òåõíîëîãèè ïðî- ïîäòâåðæäåíèÿ ïàðàìåòðîâ êà÷åñòâà ïóò¸ì ïðî-
èçâîäñòâà. Âñå ïîëó÷åííûå äàííûå íåîáõîäèìî âåäåíèÿ àíàëèòè÷åñêèõ èñïûòàíèé. Ïî ìåðå
èçëîæèòü è ïðåäñòàâèòü â ðåãóëÿòîðíûé îðãàí â óâåëè÷åíèÿ ðàçìåðîâ ìîëåêóëû ëåêàðñòâåííîãî
âèäå äîñüå íà èçìåíåíèå. ïðåïàðàòà ðàñòåò íàáîð íåîáõîäèìûõ èñïûòàíèé
Ïîñëå àíàëèçà ïîêàçàòåëåé êà÷åñòâà ïðîèçâî- äëÿ ïîäòâåðæäåíèÿ ñîïîñòàâèìîñòè, ïðè ýòîì
äèòåëü ìîæåò ñòîëêíóòüñÿ ñ îäíîé èç ñëåäóþùèõ íà ïåðâûé ïëàí âûõîäèò îáåñïå÷åíèå îäíîðîä-
ñèòóàöèé [1]: íîñòè è âîñïðîèçâîäèìîñòè ïðîöåññà ïðîèçâîä-
• îñíîâûâàÿñü íà íàäëåæàùåì ñðàâíåíèè ñòâà, òîãäà êàê àíàëèòè÷åñêèå âîçìîæíîñòè íàä-
çíà÷èìûõ ïîêàçàòåëåé êà÷åñòâà, ëåêàðñòâåííîå ëåæàùåãî îïèñàíèÿ ñâîéñòâ òàêèõ ïðåïàðàòîâ
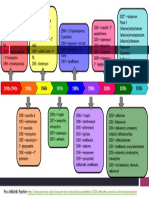
ñðåäñòâî äî è ïîñëå èçìåíåíèé âûñîêîàíàëîãè÷- ñíèæàþòñÿ (ðèñóíîê) [21]. Íà ðèñóíêå ïîêàçàíî,
íî è ñ÷èòàåòñÿ ñîïîñòàâèìûì, ò. å. íåæåëàòåëüíî- ÷òî ÷åì êðóïíåå ìîëåêóëà, òåì ñëîæíåå îíà ïîä-
ãî âëèÿíèÿ íà ïðîôèëè áåçîïàñíîñòè è ýôôåê- äàåòñÿ àíàëèòè÷åñêîìó îïèñàíèþ (òåì áîëüøå
òèâíîñòè íå ïðîãíîçèðóåòñÿ; òðåáóåòñÿ ðàçëè÷íûõ àíàëèòè÷åñêèõ èñïûòà-
• íåñìîòðÿ íà òî ÷òî ëåêàðñòâåííîå ñðåäñòâî íèé); îñíîâîé îáåñïå÷åíèÿ êà÷åñòâà ñëîæíûõ
äî è ïîñëå èçìåíåíèé êàæåòñÿ âûñîêîàíàëîãè÷- ëåêàðñòâåííûõ ïðåïàðàòîâ ÿâëÿåòñÿ îäíîðîä-
íûì, èñïîëüçîâàííûõ àíàëèòè÷åñêèõ ìåòîäèê äëÿ íûé è âîñïðîèçâîäèìûé ïðîöåññ ïðîèçâîäñòâà,
âûÿâëåíèÿ çíà÷èìûõ ðàçëè÷èé, êîòîðûå ìîãóò ïî- êîòîðûé â ðåãèñòðàöèîííîì äîñüå äîëæåí áûòü
âëèÿòü íà áåçîïàñíîñòü è ýôôåêòèâíîñòü ëåêàðñò- èñ÷åðïûâàþùå îïèñàí.
âåííîãî ñðåäñòâà, íåäîñòàòî÷íî. Ïðîèçâîäèòåëü Òðåáîâàíèÿ ê èññëåäîâàíèþ ñðàâíèòåëüíîé
äîëæåí ïðîâåñòè äîïîëíèòåëüíûå èñïûòàíèÿ (íà- ýôôåêòèâíîñòè è áåçîïàñíîñòè çàâèñÿò îò ýòàïà
ïðèìåð, áîëåå ïîäðîáíîå îïèñàíèå ñâîéñòâ) èëè ðàçðàáîòêè, âèäà ïðîèçâåä¸ííîãî èçìåíåíèÿ è åãî
äîêëèíè÷åñêèå è (èëè) êëèíè÷åñêèå èññëåäîâà- âëèÿíèÿ íà ïîêàçàòåëè êà÷åñòâà. Ïðîèçâîäèòåëè
íèÿ, ÷òîáû ïðèéòè ê îäíîçíà÷íîìó çàêëþ÷åíèþ; ÷àñòî îñóùåñòâëÿþò èçìåíåíèÿ â õîäå ðàçðàáîòêè
• íåñìîòðÿ íà òî ÷òî ëåêàðñòâåííîå ñðåäñò- ëåêàðñòâåííîãî ïðåïàðàòà äî åãî ãîñóäàðñòâåííîé
âî äî è ïîñëå èçìåíåíèé êàæåòñÿ âûñîêîàíàëî- ðåãèñòðàöèè. Åñëè èçìåíåíèå ïðîèçâîäñòâà ïðî-
ãè÷íûì, îáíàðóæåíû íåêîòîðûå ðàçëè÷èÿ ìåæäó èçîøëî äî ïîäòâåðæäàþùåãî(èõ) èññëåäîâà-
ïîêàçàòåëÿìè êà÷åñòâà ëåêàðñòâåííîãî ñðåäñòâà íèÿ(é), íåîáõîäèìîñòü â ïðåäñòàâëåíèè äîïîëíè-
äî è ïîñëå èçìåíåíèé, îäíàêî ìîæíî îáîñíîâàòü, òåëüíûõ äàííûõ èññëåäîâàíèé ñîïîñòàâèìîñòè
÷òî íåæåëàòåëüíîãî âëèÿíèÿ íà ïðîôèëè áåçî- ìîæåò áûòü ìåíüøåé, ÷åì â ñëó÷àå èçìåíåíèé,
ïàñíîñòè èëè ýôôåêòèâíîñòè íå îæèäàåòñÿ (îñ- îñóùåñòâëåííûõ ïî çàâåðøåíèè ïîäòâåðæäàþùå-
íîâûâàÿñü íà íàêîïëåííîì îïûòå ïðîèçâîäèòå- ãî(èõ) èññëåäîâàíèÿ(é) èëè ïîñëå ãîñóäàðñòâåí-
ëÿ, çíà÷èìûõ ñâåäåíèÿõ è äàííûõ).  ýòîì ñëó÷àå íîé ðåãèñòðàöèè (òàáë. 2) [1, 17].
ëåêàðñòâåííîå ñðåäñòâî äî è ïîñëå èçìåíåíèé
ìîæíî ñ÷èòàòü ñîïîñòàâèìûì; Òðåáîâàíèÿ ê îáú¸ìó èññëåäîâàíèé
• íåñìîòðÿ íà òî ÷òî ëåêàðñòâåííîå ñðåäñò-
âî äî è ïîñëå èçìåíåíèé êàæåòñÿ âûñîêîàíàëî-
ñîïîñòàâèìîñòè
ãè÷íûì, îáíàðóæåíû íåêîòîðûå ðàçëè÷èÿ ìåæäó  öåëÿõ îáåñïå÷åíèÿ êà÷åñòâà, áåçîïàñíîñòè
ïîêàçàòåëÿìè êà÷åñòâà ëåêàðñòâåííîãî ñðåäñòâà è ýôôåêòèâíîñòè ëåêàðñòâåííîãî ïðåïàðàòà, ïî-
48 АНТИБИОТИКИ И ХИМИОТЕРАПИЯ, 2013, 58; 9—10
БЕЗОПАСНОСТЬ ЛС
Зависимость объёма исследований от сложности строения молекулы биологического препарата.
Таблица 2. Объём исследований в зависимости от этапа разработки, на котором вносятся изменения
Ýòàï èçìåíåíèé Íåîáõîäèìûé îáú¸ì äàííûõ
Èçìåíåíèÿ ïðîöåññà  öåëÿõ ïîäòâåðæäåíèÿ, ÷òî èìåþùèåñÿ äîêëèíè÷åñêèå è êëèíè÷åñêèå äàííûå, ïîëó÷åííûå
ïðîèçâîäñòâà äî íà÷àëà äî èçìåíåíèÿ, îñòàþòñÿ äåéñòâèòåëüíûìè è âîçìîæíà èõ ýêñòðàïîëÿöèÿ íà èçìåí¸ííûé
ïîäòâåðæäàþùèõ ëåêàðñòâåííûé ïðåïàðàò, êàê ïðàâèëî, äîñòàòî÷íî ïðåäñòàâèòü ñîîòâåòñòâóþùèå ðåçóëüòàòû
èññëåäîâàíèé ôèçèêî-õèìè÷åñêèõ è áèîëîãè÷åñêèõ (in vivo è in vitro) è, â íåêîòîðûõ ñëó÷àÿõ, òàêæå
äîêëèíè÷åñêèõ èëè êëèíè÷åñêèõ èññëåäîâàíèé ñîïîñòàâèìîñòè, íàïðèìåð,
ôàðìàêîêèíåòè÷åñêîãî èññëåäîâàíèÿ ñ îäíîêðàòíûì ââåäåíèåì.
Èçìåíåíèÿ ïðîöåññà Íå ðåêîìåíäóåòñÿ îñóùåñòâëÿòü èçìåíåíèÿ â õîäå ïîäòâåðæäàþùåãî èññëåäîâàíèÿ;
ïðîèçâîäñòâà â õîäå â ïðîòèâíîì ñëó÷àå çàÿâèòåëþ ñëåäóåò îáðàòèòüñÿ çà íàó÷íîé êîíñóëüòàöèåé.
ïîäòâåðæäàþùèõ
êëèíè÷åñêèõ
èññëåäîâàíèé
Èçìåíåíèÿ ïðîöåññà Åñëè èçìåíåíèå ïðîèçâîäñòâà ïðîèñõîäèò ïî çàâåðøåíèè ïîäòâåðæäàþùåãî(èõ) èññëåäîâàíèÿ(é)
ïðîèçâîäñòâà èëè ïîñëå ãîñóäàðñòâåííîé ðåãèñòðàöèè, êàê ïðàâèëî, òðåáóþòñÿ áîëåå ãëóáîêèå èññëåäîâàíèÿ
ïî çàâåðøåíèè ñîïîñòàâèìîñòè, âêëþ÷àÿ ôèçèêî-õèìè÷åñêèå è áèîëîãè÷åñêèå èññëåäîâàíèÿ in vitro è, âîçìîæíî,
ïîäòâåðæäàþùèõ êëèíè÷åñêèå ôàðìàêîêèíåòè÷åñêèå è (èëè) ôàðìàêîäèíàìè÷åñêèå èññëåäîâàíèÿ ñîïîñòàâèìîñòè.
èññëåäîâàíèé Åñëè ðåçóëüòàòû òàêèõ èññëåäîâàíèé ñîïîñòàâèìîñòè íå èñêëþ÷àþò âëèÿíèÿ íà ïðîôèëü
èëè ïîñëå ýôôåêòèâíîñòè è áåçîïàñíîñòè ëåêàðñòâåííîãî ïðåïàðàòà, ìîæåò(ãóò) ïîòðåáîâàòüñÿ
ãîñóäàðñòâåííîé äîïîëíèòåëüíîå(ûå) êëèíè÷åñêîå(èå) èññëåäîâàíèå(ÿ).
ðåãèñòðàöèè
ëó÷àåìîãî ñ ïîìîùüþ èçìåí¸ííîãî ïðîöåññà íåíèé â ïðîöåññ ïðîèçâîäñòâà áèîëîãè÷åñêèõ ËÏ
ïðîèçâîäñòâà, ïðîâîäÿòñÿ èññëåäîâàíèÿ ñîïîñòà- íåîáõîäèìî âûáðàòü ïðåïàðàò ñðàâíåíèÿ [18].
âèìîñòè. Ýòî äîñòèãàåòñÿ çà ñ÷¸ò ñáîðà è àíàëèçà Åñëè ïðåïàðàò îðèãèíàëüíûé, òî ïðåïàðàòîì
ñîîòâåòñòâóþùèõ äàííûõ, íàïðàâëåííûõ íà âû- ñðàâíåíèÿ ÿâëÿåòñÿ ñåðèÿ îðèãèíàëüíîãî ïðåïà-
ÿâëåíèå âñÿêîãî íåæåëàòåëüíîãî âëèÿíèÿ íà ëå- ðàòà, ïðîèçâåä¸ííàÿ äî ðåàëèçàöèè èçìåíåíèÿ
êàðñòâåííûé ïðåïàðàò, îáóñëîâëåííîãî èçìåíå- òåõíîëîãèè ïðîèçâîäñòâà [1], íåçàâèñèìî îò ýòà-
íèåì ïðîöåññà ïðîèçâîäñòâà. Êàê è ïðè ïà ðàçðàáîòêè. Åñëè ëåêàðñòâåííûé ïðåïàðàò ÿâ-
ïîäòâåðæäåíèè áèîàíàëîãè÷íîñòè, ïðè ïîä- ëÿåòñÿ áèîàíàëîãîì, òî ïðåïàðàòîì ñðàâíåíèÿ íà
òâåðæäåíèè ñîïîñòàâèìîñòè ïðè âíåñåíèè èçìå- ïðåäðåãèñòðàöèîííîì ýòàïå ìîæåò ÿâëÿòüñÿ ñå-
АНТИБИОТИКИ И ХИМИОТЕРАПИЯ, 2013, 58; 9—10 49
ðèÿ áèîàíàëîãà, ïðîèçâåä¸ííàÿ äî ðåàëèçàöèè 24]. Ýòî îáóñëîâëåíî òåì, ÷òî ñïåöèôèêàöèÿ (â
èçìåíåíèÿ. Åñëè æå èçìåíåíèå ïðîèçâåäåíî ïî- Ðîññèè ýòèì äîêóìåíòîì ÿâëÿåòñÿ íîðìàòèâíàÿ
ñëå ãîñóäàðñòâåííîé ðåãèñòðàöèè, òî åäèíñòâåí- äîêóìåíòàöèÿ) ÿâëÿåòñÿ èíñòðóìåíòîì êîíòðîëÿ
íî âîçìîæíûì ïðåïàðàòîì ñðàâíåíèÿ ìîæåò ñëó- êà÷åñòâà, à íå äîêóìåíòîì, âñåñòîðîííå îïèñûâà-
æèòü îðèãèíàëüíûé ïðåïàðàò [22]. þùèì ôàðìàöåâòè÷åñêèå è áèîëîãè÷åñêèå ñâîé-
Ïîäòâåðæäåíèå ñîïîñòàâèìîñòè íåîáÿçàòåëü- ñòâà ïðåïàðàòà [25]. Îäíàêî â ðåçóëüòàòå èçìåíå-
íî äîëæíî âûðàæàòüñÿ â èäåíòè÷íîñòè ïîêàçàòå- íèÿ òåõíîëîãèè ïðîèçâîäñòâà ìîãóò îáðàçîâàòüñÿ
ëåé êà÷åñòâà ëåêàðñòâåííîãî ñðåäñòâà äî è ïîñëå íîâûå ñâîéñòâà, êîòîðûå íåóëîâèìû ñ ïîìîùüþ
âíåñåíèÿ èçìåíåíèé, îäíàêî îíè äîëæíû áûòü ìåòîäèê, âêëþ÷¸ííûõ â ñïåöèôèêàöèþ (íîðìà-
âûñîêîàíàëîãè÷íû (ñõîæè), à òåêóùèõ çíàíèé òèâíóþ äîêóìåíòàöèþ).
äîëæíî áûòü äîñòàòî÷íî äëÿ îáåñïå÷åíèÿ îòñóò- Íà âûáîð äîêëèíè÷åñêèõ è êëèíè÷åñêèõ èñ-
ñòâèÿ íåæåëàòåëüíîãî âëèÿíèÿ íà áåçîïàñíîñòü è ñëåäîâàíèé âëèÿþò ñâîéñòâà ëåêàðñòâåííîãî
ýôôåêòèâíîñòü ëåêàðñòâåííîãî ïðåïàðàòà, îáóñ- ñðåäñòâà, ò. å. íåîáõîäèìî âûáðàòü òàêóþ ñòðàòå-
ëîâëåííîãî êàêèìè-ëèáî ðàçëè÷èÿìè â ïîêàçàòå- ãèþ èññëåäîâàíèé ñîïîñòàâèìîñòè, êîòîðàÿ íàè-
ëÿõ êà÷åñòâà. ëó÷øèì îáðàçîì ñ äîñòàòî÷íîé òî÷íîñòüþ ïîçâî-
Èçó÷åíèå ñîïîñòàâèìîñòè îñíîâûâàåòñÿ íà ëÿåò ñïðîãíîçèðîâàòü è îáíàðóæèòü êëèíè÷åñêè
êîìáèíàöèè ðåçóëüòàòîâ êîìïëåêñà àíàëèòè÷åñ- çíà÷èìûå ðàçëè÷èÿ.
êèõ è áèîëîãè÷åñêèõ èñïûòàíèé, à â íåêîòîðûõ Âèä è îáú¸ì òàêèõ èññëåäîâàíèé ìîæåò ðàç-
ñëó÷àÿõ è äîêëèíè÷åñêèõ è êëèíè÷åñêèõ äàí- ëè÷àòüñÿ è çàâèñèò îò ðÿäà ôàêòîðîâ, îòíîñÿùèõ-
íûõ. Åñëè ïðîèçâîäèòåëü â ñèëàõ ïîäòâåðäèòü ñÿ ê ôàðìàöåâòè÷åñêîé ñóáñòàíöèè è ëåêàðñòâåí-
ñîïîñòàâèìîñòü èñêëþ÷èòåëüíî ïî ðåçóëüòàòàì íîìó ïðåïàðàòó, íàïðèìåð [1]:
àíàëèòè÷åñêèõ èññëåäîâàíèé, òî äîêëèíè÷åñêèå • çíàíèé î ìîëåêóëå è ïðî÷èõ ìîëåêóëàõ
è êëèíè÷åñêèå èññëåäîâàíèÿ ñ èçìåí¸ííûì ëå- òîãî æå êëàññà;
êàðñòâåííûì ñðåäñòâîì íå òðåáóþòñÿ. Îäíàêî, • ýòàïà ðàçðàáîòêè åù¸ íåçàðåãèñòðèðîâàí-
åñëè çàâèñèìîñòü ïîêàçàòåëåé áåçîïàñíîñòè è ýô- íûõ ëåêàðñòâåííûõ ïðåïàðàòîâ;
ôåêòèâíîñòè îò îòäåëüíûõ ïîêàçàòåëåé êà÷åñò- • ðåçóëüòàòîâ ôèçèêî-õèìè÷åñêèõ è áèîëî-
âà íå óñòàíîâëåíà è ïî ðåçóëüòàòàì àíàëèòè÷åñ- ãè÷åñêèõ èññëåäîâàíèé ñîïîñòàâèìîñòè;
êèõ èñïûòàíèé âûÿâëåíû ðàçëè÷èÿ â • ïðåäëàãàåìîãî êëèíè÷åñêîãî ïðèìåíå-
ïîêàçàòåëÿõ êà÷åñòâà ëåêàðñòâåííîãî ñðåäñòâà íèÿ.
äî è ïîñëå èçìåíåíèé, òî èññëåäîâàíèÿ ñîïîñòà- Ïîäòâåðæäåíèå ñîïîñòàâèìîñòè — ïðîöåññ
âèìîñòè ìîãóò ïîòðåáîâàòü äîïîëíèòåëüíîãî ïîñëåäîâàòåëüíûé, íà÷èíàþùèéñÿ ñ èññëåäîâà-
èçó÷åíèÿ êà÷åñòâà è ïðîâåäåíèÿ äîêëèíè÷åñêèõ íèé êà÷åñòâà (îãðàíè÷åííûõ èëè âñåñòîðîííèõ) è
è (èëè) êëèíè÷åñêèõ èññëåäîâàíèé. ïîäêðåïëÿåìûé, ïðè íåîáõîäèìîñòè, äîêëèíè÷å-
Ïðè ïðîâåäåíèè èññëåäîâàíèé ñîïîñòàâè- ñêèìè, êëèíè÷åñêèìè è (èëè) ôàðìàêîíàäçîð-
ìîñòè è îöåíêå õàðàêòåðèñòèêè ïðåïàðàòà îïðå- íûìè èññëåäîâàíèÿìè. Åñëè çàÿâèòåëþ ñ ïîìî-
äåëÿþò ìîëåêóëÿðíóþ ñòðóêòóðó, ïîäëèííîñòü, ùüþ ôèçèêî-õèìè÷åñêèõ è áèîëîãè÷åñêèõ
÷èñòîòó, èììóíîõèìè÷åñêèå ñâîéñòâà, ñïåöè- èññëåäîâàíèé óäàñòñÿ îáîñíîâàòü ñîïîñòàâè-
ôè÷åñêóþ áèîëîãè÷åñêóþ àêòèâíîñòü, ïîä- ìîñòü, òî ïðîâîäèòü äîêëèíè÷åñêèå èëè êëèíè-
òâåðæäàþò îòñóòñòâèå êîíòàìèíèðóþùèõ àãåí- ÷åñêèå èññëåäîâàíèÿ ñ èçìåí¸ííûì ëåêàðñòâåí-
òîâ è îöåíèâàþò ñòàáèëüíîñòü ïðåïàðàòà. íûì ïðåïàðàòîì íå òðåáóåòñÿ.
Ïîñêîëüêó áèîëîãè÷åñêèå ïðåïàðàòû ïðîèçâî- Ðàçðàáîòêà, ïëàíèðîâàíèå îáú¸ìà èññëåäîâà-
äÿòñÿ â æèâûõ ñèñòåìàõ èëè âûäåëÿþòñÿ èç áèî- íèé è ýêñïåðòèçà èõ ðåçóëüòàòîâ äîëæíû îñíîâû-
ëîãè÷åñêîãî ìàòåðèàëà, â ãîòîâîì ïðåïàðàòå âàòüñÿ íà ñëåäóþùèõ ïðèíöèïàõ:
âîçìîæíî ïðèñóòñòâèå àíòèòåë, ïîëèñàõàðèäîâ, • ñðàâíèòåëüíîñòü;
ïîëèíóêëåîòèäîâ è âèðóñíîãî ìàòåðèàëà [23]. • îðòîãîíàëüíîñòü: ñðàâíèòåëüíûå èñïû-
Ïî ýòîé ïðè÷èíå íåîáõîäèìî ïðîâåäåíèå íàä- òàíèÿ îäíîãî è òîãî æå ïîêàçàòåëÿ, íàïðèìåð,
ëåæàùèõ èñïûòàíèé íà áèîëîãè÷åñêóþ íàãðóç- ïîäëèííîñòè, äîëæíû ïðîâîäèòüñÿ ñ ïîìîùüþ
êó è (èëè) âèðóñíóþ áåçîïàñíîñòü èçìåí¸ííîãî ìåòîäîâ, îñíîâàííûõ íà ðàçëè÷íûõ ôèçèêî-õè-
áàíêà êëåòîê è êëåòîê ñ ïðåäåëüíûì äëÿ ïðîèç- ìè÷åñêèõ è (èëè) áèîëîãè÷åñêèõ ïðèíöèïàõ,
âîäñòâà êëåòî÷íûì âîçðàñòîì in vitro. (íàïðèìåð, ïåïòèäíîå êàðòèðîâàíèå è èçîýëåêò-
Îïèñàíèå ñâîéñòâ (characterization) ïðåïàðà- ðîôîêóñèðîâàíèå);
òà, êàê ïðàâèëî, îõâàòûâàåò îöåíêó áîëåå øèðî- • ïîñëåäîâàòåëüíîñòü: êà÷åñòâî, äîêëèíè-
êîãî äèàïàçîíà ïîêàçàòåëåé êà÷åñòâà ïî ñðàâíå- ÷åñêèå è êëèíè÷åñêèå èññëåäîâàíèÿ;
íèþ ñ äèàïàçîíîì ïîêàçàòåëåé, âêëþ÷¸ííûõ â • ÷åì ñëîæíåå ñòðîåíèå, òåì áîëüøå èññëå-
ñïåöèôèêàöèþ (íîðìàòèâíóþ äîêóìåíòàöèè) äîâàíèé;
ôàðìàöåâòè÷åñêîé ñóáñòàíöèè èëè ãîòîâîãî ïðå- • îáú¸ì èññëåäîâàíèé íà êàæäîì ýòàïå çà-
ïàðàòà, è òðåáóåò áîëåå øèðîêîãî è ñëîæíîãî íà- âèñèò îò ñòåïåíè áèîàíàëîãè÷íîñòè, ïîäòâåðæ-
áîðà èñïîëüçóåìûõ àíàëèòè÷åñêèõ ìåòîäîâ [1, äåííîé íà ïðåäûäóùèõ ýòàïàõ.
50 АНТИБИОТИКИ И ХИМИОТЕРАПИЯ, 2013, 58; 9—10
БЕЗОПАСНОСТЬ ЛС
Âîïðîñû ïîäòâåðæäåíèÿ êà÷åñòâà. Ïîäòâåðæ- óñòàíîâëåííûå êðèòåðèè ïðèåìëåìîñòè, íî íå
äåíèå êà÷åñòâà ïðè èçìåíåíèè òåõíîëîãèè ïðîèç- óêëàäûâàþùèåñÿ â ðàíåå íàáëþäàåìûå òåíäåí-
âîäñòâà ìíîãîìåðíî è çàêëþ÷àåòñÿ â ñëåäóþùåì: öèè êîíòðîëÿ ïðîèçâîäñòâà, ìîãóò ñâèäåòåëüñò-
• ñîîòâåòñòâèè ïðîèçâîäñòâà ïðèíöèïàì è âîâàòü î ðàçëè÷èÿõ ìåæäó ëåêàðñòâåííûìè ñðåä-
ðóêîâîäñòâàì ïî íàäëåæàùåé ïðîèçâîäñòâåííîé ñòâàìè, ïðîèçâåä¸ííûì ñ ïîìîùüþ ñòàðîãî è
ïðàêòèêå [26]; íîâîãî ïðîöåññîâ, è ïîòðåáîâàòü äîïîëíèòåëüíî-
• ñîîòâåòñòâèè ïðîöåññà ïðîèçâîäñòâà öå- ãî èññëåäîâàíèÿ èëè àíàëèçà. Åñëè äàííûå óêà-
ëåâîìó íàçíà÷åíèþ — âàëèäàöèÿ è (èëè) îöåíêà çûâàþò íà òî, ÷òî ïðåäûäóùåå èñïûòàíèå áîëåå
ïðîöåññà [27, 28]; íå ïîäõîäèò äëÿ ñòàíäàðòíîãî àíàëèçà ñåðèé ëå-
• ñîîòâåòñòâèè ôàðìàöåâòè÷åñêîé ñóáñòàí- êàðñòâåííîãî ñðåäñòâà ïîñëå èçìåíåíèÿ òåõíîëî-
öèè è ãîòîâîãî ïðåïàðàòà öåëåâîìó íàçíà÷åíèþ ãèè åãî ïðîèçâîäñòâà, ìîæåò ïîòðåáîâàòüñÿ ìîäè-
— îáåñïå÷åíèå ôàðìàöåâòè÷åñêîãî (ôàðìàêî- ôèêàöèÿ, èñêëþ÷åíèå èëè äîáàâëåíèå
ïåéíîãî) êà÷åñòâà; èñïûòàíèÿ â ñïåöèôèêàöèþ.
• îáåñïå÷åíèè ñîïîñòàâèìîñòè ìåæäó ôàð- Ïðè îïðåäåë¸ííûõ èçìåíåíèÿõ ïðîöåññà ïðî-
ìàöåâòè÷åñêîé ñóáñòàíöèåé è ãîòîâûì ïðåïàðà- èçâîäñòâà, äàæå íåáîëüøèå åãî ìîäèôèêàöèè ìî-
òîì, ïðîèçâåä¸ííûì äî èçìåíåíèÿ òåõíîëîãèè ãóò âûçâàòü èçìåíåíèÿ ñòàáèëüíîñòè èçìåí¸ííî-
ïðîèçâîäñòâà, ñ òàêîâûìè ïîñëå èçìåíåíèÿ — èñ- ãî ëåêàðñòâåííîãî ïðåïàðàòà. Íåîáõîäèìî
ñëåäîâàíèÿ ñîïîñòàâèìîñòè. èçó÷èòü êàæäîå èçìåíåíèå, îáëàäàþùåå ñïîñîá-
 öåëÿõ ìàêñèìàëüíîãî âûÿâëåíèÿ çíà÷èìûõ íîñòüþ ïîâëèÿòü íà ñòðóêòóðó áåëêà èëè ÷èñòîòó
ðàçëè÷èé â ïîêàçàòåëÿõ êà÷åñòâà ëåêàðñòâåííîãî è ïðîôèëü ïðèìåñåé, ñ òî÷êè çðåíèÿ âëèÿíèÿ íà
ñðåäñòâà, êîòîðûå ìîãóò âîçíèêíóòü âñëåäñòâèå ñòàáèëüíîñòü, ïîñêîëüêó áåëêè çà÷àñòóþ âîñïðè-
èçìåíåíèÿ ïðîöåññà ïðîèçâîäñòâà, íåîáõîäèìî èì÷èâû ê èçìåíåíèÿì. Áîëåå òîãî, èññëåäîâàíèÿ
òùàòåëüíî ïîäîáðàòü è îïòèìèçèðîâàòü íàáîð ñòàáèëüíîñòè äîëæíû îáëàäàòü ñïîñîáíîñòüþ îá-
èñïûòàíèé, ïðîâîäèìûõ â ðàìêàõ èññëåäîâàíèé íàðóæèâàòü íåçíà÷èòåëüíûå ðàçëè÷èÿ, êîòîðûå
ñîïîñòàâèìîñòè. ×òîáû èçó÷èòü âåñü äèàïàçîí íåâîçìîæíî ñ ë¸ãêîñòüþ îáíàðóæèòü â èññëåäî-
ôèçèêî-õèìè÷åñêèõ ñâîéñòâ è áèîëîãè÷åñêîé àê- âàíèÿõ ïî îïèñàíèþ ñâîéñòâ.
òèâíîñòè öåëåñîîáðàçíî èñïîëüçîâàòü, ïî ìåíü- Â ñîîòâåòñòâèè ñ Åâðîïåéñêèì îïûòîì, ïðè
øåé ìåðå, äâà àíàëèòè÷åñêèõ ìåòîäà äëÿ êàæäîãî âíåñåíèè èçìåíåíèé â ïðîöåññ ïðîèçâîäñòâà ôàð-
ïîêàçàòåëÿ êà÷åñòâà (íàïðèìåð, ìîëåêóëÿðíóþ ìàöåâòè÷åñêîé áèîëîãè÷åñêîé ñóáñòàíöèè ëåêàð-
ìàññó, ïðèìåñè, âòîðè÷íûå/òðåòè÷íûå ñòðóêòóðû ñòâåííûõ ñðåäñòâ ïðîâîäèòñÿ ñðàâíèòåëüíàÿ ýêñ-
ïðè èçó÷åíèè ïîäëèííîñòè) — ïðèíöèï îðòîãî- ïåðòèçà êà÷åñòâà, ïîçâîëÿþùàÿ ïîäòâåðäèòü, ÷òî
íàëüíîñòè [1, 25]. Òî åñòü â öåëÿõ ïîëó÷åíèÿ äàí- áèîëîãè÷åñêèå ñóáñòàíöèè, êàê è ãîòîâûå ïðåïà-
íûõ îá îäíîì è òîì æå ïàðàìåòðå êàæäûé ìåòîä ðàòû, îáëàäàþò âûñîêîé àíàëîãè÷íîñòüþ ïî ïîêà-
äîëæåí îñíîâûâàòüñÿ íà ðàçëè÷íûõ ôèçèêî-õè- çàòåëÿì êà÷åñòâà äî è ïîñëå èçìåíåíèé ïðîöåññà
ìè÷åñêèõ èëè áèîëîãè÷åñêèõ ïðèíöèïàõ, ÷òî ïðîèçâîäñòâà, è ÷òî íåæåëàòåëüíîãî âëèÿíèÿ íà
ïîçâîëèò ìàêñèìèçèðîâàòü îáíàðóæåíèå ðàçëè- áåçîïàñíîñòü èëè ýôôåêòèâíîñòü, âêëþ÷àÿ èììó-
÷èé â ñâîéñòâàõ ëåêàðñòâåííîãî ñðåäñòâà äî è ïî- íîãåííîñòü, ëåêàðñòâåííîãî ïðåïàðàòà íå îòìå÷à-
ñëå èçìåíåíèÿ ïðîöåññà ïðîèçâîäñòâà. åòñÿ. Çàòåì ïî ðåçóëüòàòàì àíàëèçà ïîêàçàòåëåé
Îïèñàíèå ñâîéñòâ áèîëîãè÷åñêèõ/áèîòåõíî- êà÷åñòâà, òàê æå êàê è â ñëó÷àå ãîòîâîãî áèîëîãè÷å-
ëîãè÷åñêèõ ëåêàðñòâåííûõ ñðåäñòâ ñ ïîìîùüþ ñî- ñêîãî ïðåïàðàòà, ïðèíèìàåòñÿ ðåøåíèå î íåîáõî-
îòâåòñòâóþùèõ ìåòîäîâ âêëþ÷àåò îïðåäåëåíèå äèìîñòè èëè îá îòñóòñòâèè íåîáõîäèìîñòè ïðîâå-
ôèçèêî-õèìè÷åñêèõ ñâîéñòâ, áèîëîãè÷åñêîé àê- äåíèÿ ïîäòâåðæäàþùèõ äîêëèíè÷åñêèõ èëè
òèâíîñòè, èììóíîõèìè÷åñêèõ ñâîéñòâ (ïðè èõ íà- êëèíè÷åñêèõ èññëåäîâàíèé [14, 15].  ñâÿçè ñ ýòèì
ëè÷èè), ÷èñòîòû, ïðèìåñåé, êîíòàìèíàíòîâ è ïð. â îòíîøåíèè áèîëîãè÷åñêèõ ôàðìàöåâòè÷åñêèõ
Âàæíî îòìåòèòü, ÷òî äëÿ óäîâëåòâîðèòåëüíîãî ñóáñòàíöèé ïðîöåäóðà Ãëàâíûé ôàéë ôàðìàöåâòè-
àíàëèçà âëèÿíèÿ èçìåíåíèé ïðîèçâîäñòâåííîãî ÷åñêîé ñóáñòàíöèè (Active substance master files)
ïðîöåññà îäíèõ èñïûòàíèé è àíàëèòè÷åñêèõ ìå- [29], ýêâèâàëåíòíàÿ â Ðîññèè âíåñåíèþ ôàðìàöåâ-
òîäèê, îòîáðàííûõ äëÿ ñîñòàâëåíèÿ ñïåöèôèêàöèè òè÷åñêîé ñóáñòàíöèè â ãîñóäàðñòâåííûé ðååñòð
(â Ðîññèè ýêâèâàëåíòîì ÿâëÿåòñÿ íîðìàòèâíàÿ ëåêàðñòâåííûõ ñðåäñòâ, íå ðàñïðîñòðàíÿåòñÿ [30].
äîêóìåíòàöèÿ, ÍÄ) ôàðìàöåâòè÷åñêîé ñóáñòàí- Ïðèìåíèòåëüíî ê Ðîññèéñêîé Ôåäåðàöèè
öèè èëè ëåêàðñòâåííîãî ïðåïàðàòà, êàê ïðàâèëî, ñ÷èòàåì öåëåñîîáðàçíûì íå âêëþ÷àòü áèîëîãè÷å-
íåäîñòàòî÷íî, ïîñêîëüêó öåëü èõ âûáîð çàêëþ÷à- ñêèå ôàðìàöåâòè÷åñêèå ñóáñòàíöèè â ãîñóäàðñò-
åòñÿ â ñòàíäàðòíîì êîíòðîëå êà÷åñòâà ëåêàðñò- âåííûé ðååñòð, à ïðè èçìåíåíèè òåõíîëîãèè ïðî-
âåííîãî ñðåäñòâà, à íå ïîëíîãî åãî îïèñàíèÿ. èçâîäñòâà óæå âíåñ¸ííûõ â ãîñóäàðñòâåííûé
Ïðîèçâîäèòåëè äîëæíû ïîäòâåðäèòü, ÷òî ñïåöè- ðååñòð ôàðìàöåâòè÷åñêèõ ñóáñòàíöèé ïðîâîäèòü
ôèêàöèè ïîñëå èçìåíåíèÿ ïðîöåññà ïðîèçâîäñò- èñïûòàíèÿ è èññëåäîâàíèÿ â ñîîòâåòñòâèè ñ
âà ïðîäîëæàþò îáåñïå÷èâàòü êà÷åñòâî ëåêàðñò- ïðèíöèïàìè, èçëîæåííûìè â íàñòîÿùåé ñòàòüå,
âåííîãî ñðåäñòâà. Ðåçóëüòàòû, óêëàäûâàþùèåñÿ â â òîì ÷èñëå ñ ãîòîâûì ïðåïàðàòîì.
АНТИБИОТИКИ И ХИМИОТЕРАПИЯ, 2013, 58; 9—10 51
Êðîìå òîãî, ñîãëàñíî åâðîïåéñêîìó çàêîíî- íîñòü ìîãóò ïðåäñòàâëÿòü in vitro òåõíîëîãèè, òà-
äàòåëüñòâó çàìåíà áèîëîãè÷åñêîé ôàðìàöåâòè÷å- êèå êàê ìåòîäèêè ñâÿçûâàíèÿ «â ðåàëüíîì âðåìå-
ñêîé ñóáñòàíöèè íà äðóãóþ, ñ íåñêîëüêî èçìå- íè».  áóäóùåì ðàçâèâàþùååñÿ ó÷åíèå î ãåíîì-
í¸ííîé ìîëåêóëÿðíîé ñòðóêòóðîé ïðè îòñóòñòâèè íûõ/ïðîòåîìíûõ ìèêðîïàíåëÿõ in vivo ïîçâîëèò
ñóùåñòâåííûõ ðàçëè÷èé ïî ýôôåêòèâíîñòè è îáíàðóæèâàòü íåçíà÷èòåëüíûå èçìåíåíèÿ áèîëî-
(èëè) áåçîïàñíîñòè (çà èñêëþ÷åíèåì èçìåíåíèé ãè÷åñêîãî îòâåòà íà ôàðìàêîëîãè÷åñêè àêòèâíûå
ôàðìàöåâòè÷åñêîé ñóáñòàíöèè ñåçîííîé, ïðå- âåùåñòâà.
ïàíäåìè÷åñêîé èëè ïàíäåìè÷åñêîé âàêöèíû äëÿ  öåëÿõ âûÿâëåíèÿ êàêèõ-ëèáî ïðîèçîøåä-
ïðîôèëàêòèêè ãðèïïà) â ÅÑ ÿâëÿåòñÿ èçìåíåíè- øèõ èçìåíåíèé â ðåàêòèâíîñòè è îáíàðóæåíèÿ
åì, òðåáóþùèì ãîñóäàðñòâåííîé ðåãèñòðàöèè èç- âåðîÿòíûõ(îé) ïðè÷èí(û) íåñîïîñòàâèìîñòè, èñ-
ìåí¸ííîãî ëåêàðñòâåííîãî ïðåïàðàòà [11]. Ýòî ïîëüçóÿ ïàðàëëåëüíûé ñðàâíèòåëüíûé äèçàéí èñ-
îáóñëîâëåíî òåì, ÷òî ôàðìàöåâòè÷åñêàÿ ñóáñòàí- ñëåäîâàíèÿ, íåîáõîäèìî ñ ïîìîùüþ ìåòîäèê, ïî-
öèÿ, êàê è ñàì ïðåïàðàò, èìååò áèîëîãè÷åñêîå äîáíûõ èññëåäîâàíèÿì ñâÿçûâàíèÿ ñ ðåöåïòîðîì
ïðîèñõîæäåíèå è, âñëåäñòâèå å¸ ñëîæíîñòè, äëÿ èëè êëåòî÷íûì ìåòîäèêàì, ìíîãèå èç êîòîðûõ
îïèñàíèÿ è óñòàíîâëåíèÿ å¸ êà÷åñòâà òðåáóåòñÿ ìîãóò áûòü äîñòóïíû ïî ðåçóëüòàòàì èçó÷åíèÿ êà-
êîìáèíàöèÿ ôèçèêî-õèìè÷åñêèõ è áèîëîãè÷åñ- ÷åñòâà, èçó÷èòü ïðåïàðàòû äî è ïîñëå èçìåíåíèÿ.
êèõ èñïûòàíèé, à òàêæå èñïûòàíèå è êîíòðîëü Ïðè íàëè÷èè íåêîòîðîé íåîïðåäåë¸ííîñòè
ïðîöåññà ïðîèçâîäñòâà. èëè îïàñåíèé îòíîñèòåëüíî ôàðìàêîêèíåòè÷åñ-
Äîêëèíè÷åñêèå èññëåäîâàíèÿ. Åñëè äëÿ óñòà- êèõ ïàðàìåòðîâ èëè ôàðìàêîäèíàìè÷åñêèõ ýô-
íîâëåíèÿ ñîïîñòàâèìîñòè íåèçìåí¸ííîãî è èç- ôåêòîâ, çíà÷èìûõ äëÿ êëèíè÷åñêîãî ïðèìåíåíèÿ
ìåí¸ííîãî ëåêàðñòâåííîãî ïðåïàðàòà ðåçóëüòàòîâ è (èëè) áåçîïàñíîñòè, ñëåäóåò ðàññìîòðåòü âîç-
èñêëþ÷èòåëüíî ôèçèêî-õèìè÷åñêèõ è áèîëîãè- ìîæíîñòü ïðîâåäåíèÿ èññëåäîâàíèé in vivo íà îä-
÷åñêèõ èñïûòàíèé, çàòðàãèâàþùèõ êà÷åñòâî, íå- íîì èëè íåñêîëüêèõ âèäàõ æèâîòíûõ ñ èñïîëüçî-
äîñòàòî÷íî — ëèáî âñëåäñòâèå îáíàðóæåíèÿ ðàç- âàíèåì äîëæíûì îáðàçîì âàëèäèðîâàííûõ
ëè÷èé ìåæäó íèìè, ëèáî âñëåäñòâèå õàðàêòåðà æèâîòíûõ ìîäåëåé. Áîëüøóþ íàä¸æíîñòü ïðåä-
èçìåíåíèÿ ïðîèçâîäñòâà, íå ïîçâîëÿþùåãî èñ- ñòàâëÿþò ðåçóëüòàòû èññëåäîâàíèé, ïðîâåä¸ííûõ
êëþ÷èòü ðàçëè÷èé ïî ðåçóëüòàòàì èçó÷åíèÿ êà÷å- íà âèäàõ, â îòíîøåíèå êîòîðûõ íà íåèçìåí¸ííîì
ñòâà, ðåçóëüòàòû äîêëèíè÷åñêèõ èññëåäîâàíèé ïðåïàðàòå ïîêàçàíî, ÷òî îíè ÿâëÿþòñÿ ïîäõîäÿ-
ìîãóò âûÿâëÿòü öåííûå ïðèçíàêè ïîòåíöèàëüíûõ ùèìè äëÿ ÷åëîâåêà. Âìåñòî ÷èñòîé ôàðìàöåâòè-
ðàçëè÷èé â ýôôåêòèâíîñòè è áåçîïàñíîñòè. ÷åñêîé ñóáñòàíöèè ïðåäïî÷òèòåëüíî èñïîëüçî-
 îäíèõ ñëó÷àÿõ öåëåñîîáðàçíî ïðîâåñòè íå- âàòü ãîòîâóþ ëåêàðñòâåííóþ ôîðìó.  èäåàëå ñ
áîëüøîå êîëè÷åñòâî äîêëèíè÷åñêèõ èññëåäîâà- öåëüþ óïðîùåíèÿ èíòåðïðåòàöèè äàííûõ ñîñòàâ
íèé èëè íå ïðîâîäèòü èõ âîâñå, îäíàêî â äðóãèõ ëåêàðñòâåííîãî ïðåïàðàòà, èñïîëüçîâàííîãî â
ñëó÷àÿõ íåîáõîäèìî ïðîâåñòè áîëüøîé îáú¸ì èñ- äîêëèíè÷åñêèõ èññëåäîâàíèÿõ ñîïîñòàâèìîñòè,
ñëåäîâàíèé. Ñëåäóåò îòìåòèòü, ÷òî ñîñòàâëåíèå äîëæåí ñîâïàäàòü ñ òàêîâûì, èñïîëüçîâàííûì â
ïðîãðàììû íàäëåæàùèõ äîêëèíè÷åñêèõ èññëåäî- êëèíè÷åñêèõ èññëåäîâàíèÿõ.
âàíèé òðåáóåò ïîíèìàíèÿ ñòðóêòóðû è àêòèâíîñòè Êëèíè÷åñêèå èññëåäîâàíèÿ. Òðåáîâàíèÿ ê èñ-
ëåêàðñòâåííîãî ñðåäñòâà. Íåîáõîäèìî ó÷èòûâàòü ñëåäîâàíèþ ñðàâíèòåëüíîé ýôôåêòèâíîñòè è
ïîëîæåíèÿ ñîîòâåòñòâóþùèõ äîêóìåíòîâ, â ÷àñò- áåçîïàñíîñòè çàâèñÿò îò ýòàïà ðàçðàáîòêè, âèäà
íîñòè Ðóêîâîäñòâà ïî äîêëèíè÷åñêîé îöåíêå áå- ïðîèçâåä¸ííîãî èçìåíåíèÿ è åãî âëèÿíèÿ íà ïî-
çîïàñíîñòè ëåêàðñòâåííûõ ïðåïàðàòîâ, ïîëó÷åí- êàçàòåëè êà÷åñòâà. Ïðîèçâîäèòåëè ÷àñòî îñóùå-
íûõ áèîòåõíîëîãè÷åñêèì ïóò¸ì [31]. Èçìåíåíèÿ ñòâëÿþò èçìåíåíèÿ â õîäå ðàçðàáîòêè ëåêàðñò-
ïðîôèëÿ ïðèìåñåé íåîáÿçàòåëüíî òðåáóþò ïðîâå- âåííîãî ïðåïàðàòà äî åãî ãîñóäàðñòâåííîé
äåíèÿ äîêëèíè÷åñêèõ èññëåäîâàíèé. Îäíàêî âëà- ðåãèñòðàöèè. Åñëè èçìåíåíèå ïðîèçâîäñòâà
äåëåö ðåãèñòðàöèîííîãî óäîñòîâåðåíèÿ äîëæåí ïðîèçîøëî äî ïîäòâåðæäàþùåãî(èõ) èññëåäîâà-
îáîñíîâàòü âûáðàííóþ ñòðàòåãèþ [32]. Îòñóòñò- íèÿ(é), íåîáõîäèìîñòü â ïðåäñòàâëåíèè äîïîë-
âèå ðåçóëüòàòîâ äîêëèíè÷åñêèõ èññëåäîâàíèé íèòåëüíûõ äàííûõ èññëåäîâàíèé ñîïîñòàâèìîñ-
òàêæå òðåáóåò ïîäðîáíîãî îáîñíîâàíèÿ [32]. òè ìîæåò áûòü ìåíüøåé, ÷åì â ñëó÷àå
Äîêëèíè÷åñêèå èññëåäîâàíèÿ äîëæíû áûòü èçìåíåíèé, îñóùåñòâë¸ííûõ ïî çàâåðøåíèè
ñðàâíèòåëüíûå è íàöåëåíû íà âûÿâëåíèå ðàçëè- ïîäòâåðæäàþùåãî(èõ) èññëåäîâàíèÿ(é) èëè ïî-
÷èé ìåæäó ðåàêöèÿìè íà íåèçìåí¸ííûé è èçìå- ñëå ãîñóäàðñòâåííîé ðåãèñòðàöèè [32].
í¸ííûé ëåêàðñòâåííûé ïðåïàðàò, à íå ðåàêöèé Êëèíè÷åñêèå èññëåäîâàíèÿ â îáîñíîâàíèå
per se. Íåèçìåí¸ííûé è èçìåí¸ííûé ëåêàðñòâåí- ñîïîñòàâèìîñòè ìîãóò áûòü ôàðìàêîêèíåòè÷åñ-
íûå ïðåïàðàòû ñëåäóåò ñðàâíèâàòü â ðàìêàõ îäíî- êèìè, ôàðìàêîäèíàìè÷åñêèìè è èññëåäîâàíèÿ-
ãî èññëåäîâàíèÿ. ìè ýôôåêòèâíîñòè.
Ó÷èòûâàÿ âíîâü ïîÿâëÿþùèåñÿ òåõíîëîãèè Ôàðìàêîêèíåòè÷åñêèå èññëåäîâàíèÿ ÿâëÿþòñÿ
íåîáõîäèìî ðåãóëÿðíî ïåðåñìàòðèâàòü ìåòîäû âàæíîé ÷àñòüþ êëèíè÷åñêèõ èññëåäîâàíèé ñîïî-
èññëåäîâàíèÿ. Íàïðèìåð, îïðåäåë¸ííóþ öåí- ñòàâèìîñòè. Ïîñêîëüêó öåëü çàêëþ÷àåòñÿ â ïîä-
52 АНТИБИОТИКИ И ХИМИОТЕРАПИЯ, 2013, 58; 9—10
БЕЗОПАСНОСТЬ ЛС
òâåðæäåíèè ñîïîñòàâèìîñòè, à íå îïèñàíèÿ êëè- ðàæåíèÿìè êëèíè÷åñêîé ýôôåêòèâíîñòè, íî
íè÷åñêîé ôàðìàêîëîãèè ïðåïàðàòà per se, òàêèå òàêæå áûòü íàöåëåííûì íà îáíàðóæåíèå ðàçëè-
èññëåäîâàíèÿ äîëæíû áûòü ñðàâíèòåëüíûìè. ÷èé â áåçîïàñíîñòè.
 öåëîì, ïðèåìëåìî ïåðåêð¸ñòíîå èññëåäîâà- Íåâçèðàÿ íà äîêàçàííóþ ñîïîñòàâèìóþ ýô-
íèå ñ ââåäåíèåì îäíîêðàòíîé äîçû, òàê êàê ïðÿ- ôåêòèâíîñòü, ïðîôèëü áåçîïàñíîñòè èçìåí¸ííî-
ìîìó ñðàâíåíèþ, êàê ïðàâèëî, õàðàêòåðíà áîëåå ãî ïðåïàðàòà ìîæåò îòëè÷àòüñÿ îò íåèçìåí¸ííîãî
íèçêàÿ âàðèàöèÿ. Îäíàêî íåîáõîäèìî ïðèíèìàòü (èëè â îòíîøåíèè áèîàíàëîãà — îðèãèíàëüíîãî
âî âíèìàíèå òàêèå ôàêòîðû, êàê: èììóíîãåí- ïðåïàðàòà) (ïî õàðàêòåðó, ñåðü¸çíîñòè èëè ÷àñòî-
íîñòü è å¸ âîçìîæíûå ïîñëåäñòâèÿ íà ôàðìàêî- òå íåæåëàòåëüíûõ ðåàêöèé). Äî ðåàëèçàöèè èçìå-
êèíåòèêó è (èëè) ôàðìàêîäèíàìèêó. Áîëåå ïî- íåíèé íåîáõîäèìî ïîëó÷èòü äàííûå îò äîñòàòî÷-
äðîáíî ñì. Ðóêîâîäñòâî ïî ôàðìàêîêèíåòèêå íîãî êîëè÷åñòâà ïàöèåíòîâ, ïîçâîëÿþùèå
òåðàïåâòè÷åñêèõ áåëêîâ [33]. ñðàâíèòü ïðîôèëè íåæåëàòåëüíûõ ðåàêöèé èçìå-
Ôàðìàêîäèíàìèêó ðåêîìåíäóåòñÿ îöåíèâàòü í¸ííîãî è íåèçìåí¸ííîãî ïðåïàðàòà. Ïðè àíàëè-
êàê ÷àñòü ñðàâíèòåëüíîãî ôàðìàêîêèíåòè÷åñêîãî çå íåæåëàòåëüíûõ ÿâëåíèé çàÿâèòåëü äîëæåí íå
èññëåäîâàíèÿ, ïîñêîëüêó èçìåíåíèÿ ôàðìàêîäè- òîëüêî îòìåòèòü ÷àñòîòó èõ âîçíèêíîâåíèÿ, íî
íàìèêè â íåêîòîðûõ ñëó÷àÿõ îáóñëîâëåíû èçìå- òàêæå âîçìîæíûå ðàçëè÷èÿ â èõ êëèíè÷åñêèõ
íåíèåì êèíåòèêè. Èññëåäîâàíèÿ, îïÿòü æå, ïðîÿâëåíèÿõ (ïðîäîëæèòåëüíîñòü, òÿæåñòü è ñå-
äîëæíû íîñèòü ñðàâíèòåëüíûé õàðàêòåð, à íå ðü¸çíîñòü, îáðàòèìîñòü, îòâåò íà ëå÷åíèå è ò. ä.) è
áûòü íàïðàâëåíû íà îïèñàíèå ôàðìàêîäèíàìèêè âêëþ÷èòü â ðåãèñòðàöèîííîå äîñüå ðàññìàòðèâàå-
ïðåïàðàòà per se. ìîãî ëåêàðñòâåííîãî ïðåïàðàòà ñïåöèôèêàöèþ
Âûáîð êîíå÷íîé òî÷êè äîëæåí îáåñïå÷èâàòü áåçîïàñíîñòè. Îíà äîëæíà âêëþ÷àòü îïèñàíèå
äîñòàòî÷íóþ ÷óâñòâèòåëüíîñòü, ïîçâîëÿþùóþ âîçìîæíûõ ïðîáëåì ñ áåçîïàñíîñòüþ, îáóñëîâ-
îáíàðóæèòü íåáîëüøèå ðàçëè÷èÿ, äîñòàòî÷íóþ ëåííûõ èçìåíåíèÿìè ïðîöåññà ïðîèçâîäñòâà.
ïðåöèçèîííîñòü èçìåðåíèÿ è êëèíè÷åñêóþ çíà- Èíîãäà ïîñëå ðåàëèçàöèè èçìåíåíèé ìîãóò ïî-
÷èìîñòü äëÿ öåëåâîé ïîïóëÿöèè.  öåëÿõ ïîä- òðåáîâàòüñÿ äîïîëíèòåëüíûå èññëåäîâàíèÿ, íà-
òâåðæäåíèÿ àíàëèòè÷åñêîé ÷óâñòâèòåëüíîñòè íå- ïðèìåð, ôàðìàêîýïèäåìèîëîãè÷åñêèå [32].
îáõîäèìî îáîñíîâàòü âûáðàííûé äèàïàçîí äîç.
Öåëåñîîáðàçíî ïðîâîäèòü èññëåäîâàíèÿ íå- Òðåáîâàíèÿ ê ñîñòàâëåíèþ äîñüå
ñêîëüêèõ äîç [34], îáîñíîâàòü âûáîð ìàðê¸ðà(îâ),
à òàêæå çàðàíåå óñòàíîâèòü è îáîñíîâàòü ãðàíèöó
íà èçìåíåíèÿ
ýêâèâàëåíòíîñòè. Â ñâÿçè ñ ýòèì íåîáõîäèìî Ïðè èçìåíåíèè òåõíîëîãèè ïðîèçâîäñòâà áè-
îáîñíîâàòü âûáîð ïîïóëÿöèè èññëåäîâàíèÿ. Èñ- îòåõíîëîãè÷åñêèõ/áèîëîãè÷åñêèõ ëåêàðñòâåííûõ
ïîëüçîâàíèå ïåðâè÷íûõ èëè âòîðè÷íûõ ôàðìà- ïðåïàðàòîâ ðåãóëÿòîðíàÿ ïðàêòèêà âñåãî ìèðà
êîäèíàìè÷åñêèõ ìàðê¸ðîâ âîçìîæíî ëèøü ó ïà- ïðèäåðæèâàåòñÿ àêñèîìû î íåêà÷åñòâåííîñòè,
öèåíòîâ, à íå çäîðîâûõ äîáðîâîëüöåâ. íåáåçîïàñíîñòè, íåýôôåêòèâíîñòè ëåêàðñòâåí-
Åñëè ïîäõîäÿùèå ìàðê¸ðû îòñóòñòâóþò èëè íîãî ïðåïàðàòà, ïðîèçâåä¸ííîãî ñ ïîìîùüþ íî-
åñëè ñ ïîìîùüþ ôàðìàêîäèíàìè÷åñêèõ èññëå- âîé òåõíîëîãèè ïðîèçâîäñòâà [11, 13, 35]. Òàêèì
äîâàíèé íå óäàëîñü ÷¸òêî ïîäòâåðäèòü ñîïîñòà- îáðàçîì, áðåìÿ ïîäòâåðæäåíèÿ îòñóòñòâèÿ íåæå-
âèìîñòü, íåîáõîäèìî, èñïîëüçóÿ êëèíè÷åñêèå ëàòåëüíîãî âëèÿíèÿ êàê íà êà÷åñòâî, òàê è áåçî-
êîíå÷íûå òî÷êè, ïðîâåñòè ñðàâíèòåëüíîå êëèíè- ïàñíîñòü è ýôôåêòèâíîñòü â ñâÿçè ñ èçìåíåíèåì
÷åñêîå èññëåäîâàíèå ýêâèâàëåíòíîñòè, ïîäòâåðæ- òåõíîëîãèè ïðîèçâîäñòâà ïîëíîñòüþ ëîæèòñÿ íà
äàþùåå ñîïîñòàâèìóþ ýôôåêòèâíîñòü. Èññëåäî- âëàäåëüöà ðåãèñòðàöèîííîãî óäîñòîâåðåíèÿ. Â
âàíèÿ, îïÿòü æå, äîëæíû íîñèòü ñðàâíèòåëüíûé õîäå íàó÷íîé ýêñïåðòèçû íàó÷íûé îðãàí âïðàâå
õàðàêòåð, â êîòîðûõ ñîïîñòàâëÿþòñÿ èññëåäóå- ïðèíÿòü èëè îòêëîíèòü ïðåäñòàâëåííîå îáîñíî-
ìûé ïðåïàðàò è ïðåïàðàò ñðàâíåíèÿ.  îòíîøå- âàíèå, ðóêîâîäñòâóÿñü èñêëþ÷èòåëüíî íàó÷íûìè
íèè îðèãèíàëüíûõ ëåêàðñòâåííûõ ïðåïàðàòîâ ïðèíöèïàìè. Îòñóòñòâèå îáîñíîâàíèÿ âëå÷¸ò çà
òàêîâûìè ÿâëÿþòñÿ ëåêàðñòâåííûå ïðåïàðàòû ñîáîé ïðèìåíåíèå âûøåóïîìÿíóòîé àêñèîìû.
äî è ïîñëå âíåñåíèÿ èçìåíåíèÿ [1], â îòíîøåíèè Äàæå åñëè, ïî ìíåíèþ ýêñïåðòîâ, èçìåíåíèå íå
áèîàíàëîãîâ — ñåðèÿ áèîàíàëîãà ïîñëå èçìåíå- âëå÷¸ò çà ñîáîé íåãàòèâíîãî âëèÿíèÿ íà êà÷åñòâî,
íèÿ ïðîöåññà ïðîèçâîäñòâà è îðèãèíàëüíûé áåçîïàñíîñòü è (èëè) ýôôåêòèâíîñòü ëåêàðñòâåí-
ïðåïàðàò [22]. Âî èçáåæàíèå ñèñòåìàòè÷åñêèõ íîãî ïðåïàðàòà, ïîäâåðãøåãîñÿ èçìåíåíèþ òåõ-
îøèáîê êëèíè÷åñêèå èññëåäîâàíèÿ äîëæíû, êàê íîëîãèè ïðîèçâîäñòâà, íî ïðè ýòîì âëàäåëüöåì
ïðàâèëî, áûòü ðàíäîìèçèðîâàííûìè, äâîéíûìè ðåãèñòðàöèîííîãî óäîñòîâåðåíèÿ íå ïðåäñòàâëå-
ñëåïûìè. Ïîòåíöèàëüíûå ðàçëè÷èÿ â ýôôåêòèâ- íî îáîñíîâàíèå îòñóòñòâèÿ òàêîãî âëèÿíèÿ, èç-
íîñòè íåîáõîäèìî èçó÷èòü â èññëåäîâàíèÿõ ñ íà- ìåíåíèå ïðèçíàåòñÿ íåîáîñíîâàííûì.
èáîëüøåé âåðîÿòíîñòüþ îáíàðóæåíèÿ òàêèõ ðàç- Ïîýòîìó, ïîäîáíî ëþáîìó èçìåíåíèþ ðåãèñò-
ëè÷èé [34]. Ïðè îïðåäåëåíèè ðàçìåðà âûáîðêè ðàöèîííîãî äîñüå çàðåãèñòðèðîâàííîãî ëåêàðñò-
íåîáõîäèìî ðóêîâîäñòâîâàòüñÿ íå òîëüêî ñîîá- âåííîãî ïðåïàðàòà, âíåñåíèå èçìåíåíèé â òåõíîëî-
АНТИБИОТИКИ И ХИМИОТЕРАПИЯ, 2013, 58; 9—10 53
ãèþ ïðîèçâîäñòâà áèîòåõíîëîãè÷åñêèõ/ áèîëîãè÷å- ëåíèÿ îáîñíîâàíèÿ öåëåñîîáðàçíî ïðåäñòàâèòü â
ñêèõ ëåêàðñòâåííûõ ïðåïàðàòîâ íåîáõîäèìî ñîïðî- ñîîòâåòñòâèè ñ Ìîäóëåì 2 Îáùåãî òåõíè÷åñêîãî
âîæäàòü ïîäðîáíûì îïèñàíèåì âíîñèìûõ èçìåíå- äîêóìåíòà [37].
íèé, îïèñàíèåì ïîòåíöèàëüíîãî âëèÿíèÿ òàêèõ
èçìåíåíèé íà ïîêàçàòåëè êà÷åñòâà, áåçîïàñíîñòè è Çàêëþ÷åíèå
ýôôåêòèâíîñòè. Çà÷àñòóþ â ïðåäîñòàâëÿåìîé äîêó-
ìåíòàöèè ïî êà÷åñòâó îòñóòñòâóåò îáîñíîâàíèå âû- Îòñóòñòâèå ÷¸òêèõ ðåêîìåíäàöèé è íîðìàòèâ-
áîðà ìåòîäèê, èõ âèäîâ è êîëè÷åñòâà, ìîãóò áûòü íå íî-ïðàâîâûõ àêòîâ â îòíîøåíèè ïðèíöèïîâ ïîä-
ïîëíîñòüþ îïèñàíû ôèçèêî-õèìè÷åñêèå, ôàðìà- òâåðæäåíèÿ ñîïîñòàâèìîñòè áèîëîãè÷åñêèõ ëå-
öåâòè÷åñêèå è áèîëîãè÷åñêèå ñâîéñòâà. êàðñòâåííûõ ïðåïàðàòîâ, ïîäâåðãøèõñÿ
Íåîáõîäèìî ïðåäñòàâèòü ðåçóëüòàòû ñîîòâåò- èçìåíåíèþ òåõíîëîãèè èõ ïðîèçâîäñòâà, çíà÷è-
ñòâóþùèõ ñðàâíèòåëüíûõ ôàðìàöåâòè÷åñêèõ è òåëüíî îñëîæíÿåò îáåñïå÷åíèå óäîâëåòâîðèòåëü-
áèîëîãè÷åñêèõ èñïûòàíèé. Íåîáõîäèìî ïîäðîá- íîãî èõ êà÷åñòâà, áåçîïàñíîñòè è ýôôåêòèâíîñòè.
íî îïèñàòü äàííûå (âêëþ÷àÿ ñòàòèñòè÷åñêóþ îá- Ðàçðàáîòêà ñîâðåìåííûõ, ãàðìîíèçèðîâàííûõ ñ
ðàáîòêó ðåçóëüòàòîâ) [36] î ïðîèçâîäñòâå ôàðìà- ìåæäóíàðîäíûì îïûòîì ïðèíöèïîâ, ðåãëàìåí-
öåâòè÷åñêîé ñóáñòàíöèè è ëåêàðñòâåííîãî òèðóþùèõ ïëàíèðîâàíèå êîìïëåêñà èññëåäîâà-
ïðåïàðàòà (îïèñàíèå ïðîöåññà, êîíòðîëü ìàòåðè- íèé ñîïîñòàâèìîñòè, ïîçâîëèò çàäàòü ÷¸òêèå
àëîâ è ïðîöåññîâ, âàëèäàöèÿ ïðîöåññîâ ïðîèçâîä- ñòàíäàðòû îïðåäåëåíèÿ ïåðå÷íÿ íåîáõîäèìûõ
ñòâà, ðàçðàáîòêà ïðîöåññà ïðîèçâîäñòâà), î êîí- èññëåäîâàíèé è ñîñòàâëåíèÿ äîñüå.  ñâÿçè ñ ÷åì
òðîëå êà÷åñòâà ôàðìàöåâòè÷åñêîé ñóáñòàíöèè è ãàðìîíèçàöèÿ ñóùåñòâóþùèõ ïîäõîäîâ ê ïðåä- è
ëåêàðñòâåííîãî ïðåïàðàòà (àíàëèòè÷åñêèå ìåòî- ïîñòðåãèñòðàöèîííîé ðàçðàáîòêå è ýêñïåðòèçå
äèêè, âàëèäàöèÿ àíàëèòè÷åñêèõ ìåòîäèê, àíàëè- áèîëîãè÷åñêèõ ëåêàðñòâåííûõ ïðåïàðàòîâ ñ ìåæ-
çû ñåðèé, îáîñíîâàíèå ñïåöèôèêàöèé), à òàêæå äóíàðîäíûì îïûòîì îáåñïå÷åíèÿ èõ êà÷åñòâà,
ñâåäåíèÿ î ôàðìàöåâòè÷åñêîé ðàçðàáîòêå ëåêàð- áåçîïàñíîñòè è ýôôåêòèâíîñòè ÿâëÿåòñÿ âàæíîé
ñòâåííîãî ïðåïàðàòà, êîíòðîëå êà÷åñòâà âñïîìî- çàäà÷åé, ÷òî õîðîøî ïîíèìàåòñÿ ãîñóäàðñòâîì è
ãàòåëüíûõ âåùåñòâ è âèðóñíîé áåçîïàñíîñòè. îòðàæåíî â Ñòðàòåãèè ðàçâèòèÿ ôàðìàöåâòè÷åñ-
 íåêîòîðûõ ñëó÷àÿõ, îïèñàííûõ âûøå èñïû- êîé ïðîìûøëåííîñòè Ðîññèéñêîé Ôåäåðàöèè íà
òàíèé íåäîñòàòî÷íî, è íåîáõîäèìî ïðîâåñòè ñðàâ- ïåðèîä äî 2020 ãîäà [38], ïîâòîðíî îçâó÷åíî çàìå-
íèòåëüíûå äîêëèíè÷åñêèå è (èëè) êëèíè÷åñêèå ñòèòåëåì Ïðåäñåäàòåëÿ Ïðàâèòåëüñòâà Ðîññèé-
èññëåäîâàíèÿ, ïîäòâåðæäàþùèå îòñóòñòâèå íåãà- ñêîé Ôåäåðàöèè À. Â. Äâîðêîâè÷åì [39] è ó÷òåíî
òèâíîãî âëèÿíèÿ íà êà÷åñòâî, áåçîïàñíîñòü è ýô- â ïëàíå íàó÷íî-èññëåäîâàòåëüñêîé ðàáîòû ÔÃÁÓ
ôåêòèâíîñòü ëåêàðñòâåííîãî ïðåïàðàòà. Îòñóòñò- «ÍÖÝÑÌÏ» Ìèíçäðàâà Ðîññèè ïî òåìå «Íàó÷-
âèå ðåçóëüòàòîâ ñðàâíèòåëüíûõ äîêëèíè÷åñêèõ è íîå îáîñíîâàíèå ìåòîäè÷åñêèõ ïîäõîäîâ ê äî-
(èëè) êëèíè÷åñêèõ èññëåäîâàíèé íåîáõîäèìî âî êëèíè÷åñêîìó è êëèíèêî-ôàðìàêîëîãè÷åñêîìó
âñåõ ñëó÷àÿõ îáîñíîâûâàòü äîñòàòî÷íîñòüþ ïðî- èçó÷åíèþ è ýêñïåðòíîé îöåíêå ýôôåêòèâíîñòè è
âåä¸ííûõ àíàëèòè÷åñêèõ è áèîëîãè÷åñêèõ èñïû- áåçîïàñíîñòè ëåêàðñòâåííûõ ñðåäñòâ» [40].
òàíèé. Íàó÷íûå ïðèíöèïû òàêîãî îáîñíîâàíèÿ Ñîáëþäåíèå âûøåèçëîæåííûõ ïðèíöèïîâ è
îïèñàíû â äîêóìåíòàõ «Ñîïîñòàâèìîñòü áèîòåõ- ðåêîìåíäàöèé ïîçâîëèò ñäåëàòü èñõîä ïðîöåäóðû
íîëîãè÷åñêèõ/áèîëîãè÷åñêèõ ñðåäñòâ, ïîäâåðã- âíåñåíèÿ èçìåíåíèé â ðåãèñòðàöèîííîå äîñüå çà-
øèõñÿ èçìåíåíèÿì ïðîöåññà ïðîèçâîäñòâà» (ICH ðåãèñòðèðîâàííîãî áèîòåõíîëîãè÷åñêîãî/áèîëî-
E5) [1] è Ðóêîâîäñòâå ïî ñîïîñòàâèìîñòè áèîòåõ- ãè÷åñêîãî ïðåïàðàòà ïðè èçìåíåíèè òåõíîëîãèè
íîëîãè÷åñêèõ ëåêàðñòâåííûõ ïðåïàðàòîâ ïîñëå åãî ïðîèçâîäñòâà áîëåå ïðåäñêàçóåìûì è â òî æå
èçìåíåíèÿ ïðîöåññà ïðîèçâîäñòâà: äîêëèíè÷åñ- âðåìÿ çàùèòèò èíòåðåñû îòäåëüíîãî ïàöèåíòà è
êèå è êëèíè÷åñêèå âîïðîñû [32]. Ôîðìó ïðåäñòàâ- îáùåñòâåííîãî çäîðîâüÿ â öåëîì.
ЛИТЕРАТУРА 5. Áðåäåð Â.Â. Áèîàíàëîãè: êîïèè èëè ïîõîæèå, íî èíûå ëåêàðñòâà?
Ìåä âåñò 2007; 26—27: 411—412.
1. Comparability of Biotechnological/Biological Products Subject to 6. Schellekens H. Biosimilar epoetin: how similar are they? Eur J Hosp
Changes in their Manufacturing Process (Q5E). International Pharm 2004; 3: 43—47.
Conference on Harmonisation of Technical Requirements for
7. Singh A. K. Renal Division, Brigham and Women's hospital & Harvard
Registration of Pharmaceuticals for Human Use [îôèöèàëüíûé ñàéò].
Medical School, Boston, USA. World Congress of Nephrology, Apr 22,
URL: http://www.ich.org/fileadmin/Public_Web_Site/ICH_Products/
2007.
Guidelines/Quality/Q5E/Step4/Q5E_Guideline.pdf (äàòà îáðàùå-
íèÿ: 17.12.2012). 8. Overview of Biological Active Substances of Non-Recombinant Origin.
Coordination Group for Mutual Recognition and Decentralised
2. Õàñàáîâ Í.Í., Çåìñêîâà Í.À. Áèîëîãè÷åñêèå ëåêàðñòâåííûå ñðåä-
Procedures — Human [îôèöèàëüíûé ñàéò]. URL:
ñòâà è èõ áèîàíàëîãè: îïðåäåëåíèå, âîïðîñû êà÷åñòâà, èäåíòè÷-
http://www.hma.eu/fileadmin/dateien/Human_Medicines/CMD_h_
íîñòè è áåçîïàñíîñòè. Âåñòí Ðîñçäðàâíàäçîðà 2008; 6.
/procedural_guidance/Compilation_Biological_Active_Substance_no
3. Áåëîóñîâ Ä.Þ. Áèîàíàëîãè — íàñêîëüêî îíè ïîäîáíû? Êà÷åñòâ n-recombinant_origin.pdf (äàòà îáðàùåíèÿ: 20.08.2013).
êëèí ïðàêò 2006; 2: 80—83. 9. Ôåäåðàëüíûé çàêîí îò 12 àïðåëÿ 2010 ã. ¹ 61-ÔÇ «Îá îáðàùåíèè
4. Øèëî Â.Þ. Áèîàíàëîãè â ëå÷åíèè àíåìèè ïðè õðîíè÷åñêîé áîëåç- ëåêàðñòâåííûõ ñðåäñòâ» [Ïðèíÿò Ãîñ. Äóìîé 24 ìàðòà 2010 ãîäà ñ
íè ïî÷åê: ïîòåíöèàëüíàÿ ïîëüçà èëè íåîïðàâäàííûé ðèñê? Ëå÷à- èçìåíåíèÿìè è äîïîëíåíèÿìè ïî ñîñòîÿíèþ íà 6 äåêàáðÿ 2011 ã.].
ùèé âðà÷. 2007; 9—10: 56—64. Ðîññ ãàç Ôåäåðàëüíûé âûïóñê ¹5157 îò 14 àïðåëÿ 2010 ã. [ýëåê-
54 АНТИБИОТИКИ И ХИМИОТЕРАПИЯ, 2013, 58; 9—10
БЕЗОПАСНОСТЬ ЛС
òðîííûé ðåñóðñ]. URL: http://www.rg.ru/2010/04/14/lekarstva- Pharmaceuticals for Human Use [îôèöèàëüíûé ñàéò]. URL:
dok.html (äàòà îáðàùåíèÿ: 30.08.2013). http://www.ich.org/fileadmin/Public_Web_Site/ICH_Products/Guid
10. Directive 2001/83/EC of the European Parliament and of the Council elines/Quality/Q2_R1/Step4/Q2_R1__Guideline.pdf (äàòà îáðàùå-
of 6 November 2001 on the Community code relating to medicinal íèÿ: 20.08.2013).
products for human use (Consolidated version: 20/01/2011). OJ L 311, 25. Specifications: Test Procedures and Acceptance Criteria for
28.11.2001; 67. URL: http://ec.europa.eu/health/files/eudralex/vol- Biotechnological/Biological Products (Q6B). International
1/dir_2001_83/2001_83_ec_en.pdf (äàòà îáðàùåíèÿ: 05.02.2013). Conference on Harmonisation [îôèöèàëüíûé ñàéò]. URL:
11. Commission Regulation (EC) No 1234/2008 of 24 November 2008 http://www.ich.org/fileadmin/Public_Web_Site/ICH_Products/Guid
concerning the examination of variations to the terms of marketing elines/Quality/Q6B/Step4/Q6B_Guideline.pdf (äàòà îáðàùåíèÿ:
authorisations for medicinal products for human use and veterinary 09.09.2013).
medicinal products. The rules governing medicinal products in the 26. Good manufacturing practice (GMP) Guidelines. Eudralex Vol. 4:
European Union [îôèöèàëüíûé ñàéò]. URL: http://ec.europa.eu/ GMP Human & Veterinary. European Commission [îôèöèàëüíûé
health/files/eudralex/vol-1/reg_2008_1234/ reg_2008_1234_en.pdf ñàéò]. URL: http://ec.europa.eu/health/documents/eudralex/vol-
(äàòà îáðàùåíèÿ: 12.08.2013). 4/index_en.htm (äàòà îáðàùåíèÿ: 04.09.2013).
12. Commission Regulation (EU) No 712/2012 of 3 August 2012 amending 27. Process validation. European Medicines Agency [îôèöèàëüíûé
Regulation (EC) No 1234/2008 concerning the examination of varia- ñàéò]. URL: http://www.ema.europa.eu/docs/en_GB/document_
tions to the terms of marketing authorisations for medicinal products for library/Scientific_guideline/2009/09/WC500002913.pdf (äàòà îáðà-
human use and veterinary medicinal products. The rules governing ùåíèÿ: 12.02.2013).
medicinal products in the European Union [îôèöèàëüíûé ñàéò]. 28. Process validation [draft guideline]. European Medicines Agency [îôè-
URL: http://ec.europa.eu/health/files/eudralex/vol-1/reg_2012_712/ öèàëüíûé ñàéò]. URL: http://www.ema.europa.eu/docs/en_GB/
reg_2012_712_en.pdf (äàòà îáðàùåíèÿ: 12.08.2013). document_library/Scientific_guideline/2012/04/WC500125399.pdf
13. Commission Classification Guideline - Guideline on the details of the (äàòà îáðàùåíèÿ: 12.02.2013).
various categories of variations to the terms of marketing authorisations 29. Committee for Medicinal Products for Human Use. 18-21 October
for medicinal products for human use and veterinary medicinal prod- 2004. Plenary Meeting / Monthly Report. European Medicines Agency
ucts (2010/C 17/01). The rules governing medicinal products in the [ýëåêòðîííûé ðåñóðñ]. URL: http://www.ema.europa.eu/docs/en_GB/
European Union [îôèöèàëüíûé ñàéò]. URL: http://ec.europa.eu/ document_library/Committee_meeting_report/2009/10/WC5000064
health/files/eudralex/vol-2/c17_1/c17_1_en.pdf (äàòà îáðàùåíèÿ: 07.pdf (äàòà îáðàùåíèÿ: 20.08.2013).
12.08.2013).
30. Guideline on Active Substance Master File Procedure
14. Comparability of biotechnology-derived medicinal products after a (CHMP/QWP/227/02 Rev 3/Corr *). European Medicines Agency
change in the manufacturing process - non-clinical and clinical issues [ýëåêòðîííûé ðåñóðñ]. URL: http://www.ema.europa.eu/docs/
(EMEA/CHMP/BMWP/101695/2006). European Medicines Agency en_GB/document_library/Scientific_guideline/2012/07/WC5001299
[îôèöèàëüíûé ñàéò]. URL: http://www.ema.europa.eu/docs/en_GB/ 94.pdf (äàòà îáðàùåíèÿ: 20.08.2013).
document_library/Scientific_guideline/2009/09/WC500003935.pdf
(äàòà îáðàùåíèÿ: 08.08.2013) 31. Preclinical Safety Evaluation of Biotechnology-Derived
Pharmaceuticals, S6 (R1). International Conference on Harmonisation
15. Comparability of Biotechnological/Biological Products Subject to of Technical Requirements for Registration of Pharmaceuticals for
Changes in their Manufacturing Process (Q5E). International Human Use [îôèöèàëüíûé ñàéò]. URL: http://www.ich.org/filead-
Conference on Harmonisation of Technical Requirements for min/Public_Web_Site/ICH_Products/Guidelines/Safety/S6_R1/Ste
Registration of Pharmaceuticals for Human Use [îôèöèàëüíûé ñàéò]. p4/S6_R1_Guideline.pdf (äàòà îáðàùåíèÿ: 12.08.2013).
URL: http://www.ich.org/fileadmin/Public_Web_Site/ICH_Products/
Guidelines/Quality/Q5E/Step4/Q5E_Guideline.pdf (äàòà îáðàùå- 32. Comparability of biotechnology-derived medicinal products after a
íèÿ: 17.12.2012). change in the manufacturing process - non-clinical and clinical issues
(EMEA/CHMP/BMWP/101695/2006). European Medicines Agency
16. Àâäååâà Æ.È., Ð.À. Âîëêîâà, Í.À. Àëïàòîâà è äð. Ñîïîñòàâèìîñòü [îôèöèàëüíûé ñàéò]. URL: http://www.ema.europa.eu/docs/en_GB/
áèîòåõíîëîãè÷åñêèõ ïðîäóêòîâ (ñóáñòàíöèè è ãîòîâîãî ïðåïàðà- document_library/Scientific_guideline/2009/09/WC500003935.pdf
òà), ïîëó÷åííûõ äî è ïîñëå âíåñåíèÿ èçìåíåíèé â ïðîöåññ ïðîèç- (äàòà îáðàùåíèÿ: 12.02.2013).
âîäñòâà. Âåäîì ÍÖÝÑÌÏ 2013; 2.
33. Clinical investigation of the pharmacokinetics of therapeutic proteins.
17. Ñîïîñòàâèìîñòü áèîòåõíîëîãè÷åñêèõ ïðîäóêòîâ, ïîëó÷åííûõ äî European Medicines Agency [îôèöèàëüíûé ñàéò]. URL:
è ïîñëå âíåñåíèÿ èçìåíåíèé â ïðîöåññ ïðîèçâîäñòâà. Îáùèå http://www.ema.europa.eu/docs/en_GB/document_library/Scientific
ïðèíöèïû ïëàíèðîâàíèÿ èññëåäîâàíèé. Ìåòîäè÷åñêèå ðåêîìåí- _guideline/2009/09/WC500003029.pdf (äàòà îáðàùåíèÿ: 12.02.2013).
äàöèè ÍÖÝÑÌÏ, 2011; 28.
34. Ñîñòàâëåíèå ïðîòîêîëà êîíòðîëèðóåìîãî êëèíè÷åñêîãî èññëåäî-
18. Äîêëèíè÷åñêèå è êëèíè÷åñêèå èññëåäîâàíèÿ áèîëîãè÷åñêè àíà- âàíèÿ ëåêàðñòâåííîãî ïðåïàðàòà (âûáîð êîíòðîëüíîé ãðóïïû).
ëîãè÷íûõ ëåêàðñòâåííûõ ïðåïàðàòîâ. Ìåòîäè÷åñêèå ðåêîìåíäà-
Ìåòîäè÷åñêèå ðåêîìåíäàöèè. Ì.: ÍÖÝÑÌÏ 2012; 36.
öèè ÍÖÝÑÌÏ, 2013; 30.
35. Peace K.E., Chen D.-G. Clinical Trial Methodology (Chapman &
19. Investigation of bioequivalence (CPMP/EWP/QWP/1401/98 Rev. 1).
Hall/CRC Biostatistics Series). Chapman and Hall/CRC; 1 edition,
European Medicines Agency [îôèöèàëüíûé ñàéò]. URL:
July 20, 2010; 420.
http://www.ema.europa.eu/docs/en_GB/document_library/Scientific
_guideline/2010/01/WC500070039.pdf (äàòà îáðàùåíèÿ: 06.08.2013). 36. Statistical analysis of results of biological assays and tests
(01/2008:50300). European Pharmacopeia, 7.8 [ýëåêòðîííûé ðå-
20. Title 21-Food and Drugs, Chapter I-Food and Drug Administration,
ñóðñ]. Äàòà îáðàùåíèÿ: 11.09.2013.
Department of Health and Human Services, Subchapter D--Drugs for
Human use. United States Food and Drug Administration [îôèöèàëü- 37. Ïîÿñíåíèå äëÿ çàÿâèòåëåé: ïðåäñòàâëåíèå è ôîðìàò äîñüå — îá-
íûé ñàéò]. URL: http://www.accessdata.fda.gov/scripts/cdrh/ ùèé òåõíè÷åñêèé äîêóìåíò. European Commission [îôèöèàëüíûé
cfdocs/cfcfr/CFRSearch.cfm?fr=320.1 (äàòà îáðàùåíèÿ: 06.08.2013). ñàéò]. URL: http://ec.europa.eu/health/files/eudralex/vol-2/b/
21. Mattaliano R.J. A PhRMA Member View on Biosimilars: Analytical update_200805/ctd_05-2008_en.pdf (äàòà îáðàùåíèÿ: 11.09.2013).
and Quality Considerations. United States Food and Drug 37. Ãîñóäàðñòâåííàÿ ïðîãðàììà Ðîññèéñêîé Ôåäåðàöèè «Ðàçâèòèå
Administration [îôèöèàëüíûé ñàéò]. URL: http://www.fda.gov/ ôàðìàöåâòè÷åñêîé è ìåäèöèíñêîé ïðîìûøëåííîñòè» íà 2013-
downloads/AdvisoryCommittees/CommitteesMeetingMaterials/Drug 2020 ãîäû Ïàñïîðò Ãîñóäàðñòâåííîé ïðîãðàììû Ðîññèéñêîé Ôå-
s/AdvisoryCommitteeforPharmaceuticalScienceandClinicalPharmaco äåðàöèè «Ðàçâèòèå ôàðìàöåâòè÷åñêîé è ìåäèöèíñêîé ïðîìûø-
logy/UCM315764.pdf (äàòà îáðàùåíèÿ: 09.09.2013). ëåííîñòè» íà 2013-2020 ãã. Ìèíïðîìòîðã [îôèöèàëüíûé ñàéò].
22. Îòâåò Åâðîïåéñêîãî àãåíòñòâà ïî ëåêàðñòâåííûì ñðåäñòâàì íà URL: http://www.minpromtorg.gov.ru/reposit/minprom/ministry/
îáðàùåíèå ÔÃÁÓ «Íàó÷íûé öåíòð ýêñïåðòèçû ñðåäñòâ ìåäèöèí- fcp/pharma_and_medical_industry/GP_FARMAMED.pdf (äàòà îá-
ñêîãî ïðèìåíåíèÿ» Ìèíçäðàâà Ðîññèè, 4 ñåíòÿáðÿ 2013 ã. Ëè÷íûå ðàùåíèÿ: 06.02.2012).
êîììóíèêàöèè. 39. Íîâûé èìïóëüñ ðàçâèòèÿ áèîòåõíîëîãèé â ÐÔ // Ñòâîëîâûå êëåò-
23. Update on the safety and bioequivalence of biosimilars - focus on êè [ýëåêòðîííûé ðåñóðñ]. URL: http://www.stem-cells.ru/site/
enoxaparin. Jeske W, Walenga JM, Hoppensteadt D, Fareed J. index.php/news/772 (äàòà îáðàùåíèÿ: 20.08.2013).
Dovepress [ýëåêòðîííûé ðåñóðñ]. URL: http://www.dovepress.com/ 40. Ïëàí íàó÷íî-èññëåäîâàòåëüñêîé ðàáîòû ÔÃÁÓ «ÍÖÝÑÌÏ»
update-on-the-safety-and-bioequivalence-of-biosimilars-ndash- Ìèíçäðàâà Ðîññèè ïî òåìå «Íàó÷íîå îáîñíîâàíèå ìåòîäè÷åñêèõ
focus-on--peer-reviewed-article-DHPS (äàòà îáðàùåíèÿ: ïîäõîäîâ ê äîêëèíè÷åñêîìó è êëèíèêî-ôàðìàêîëîãè÷åñêîìó
05.09.2013). èçó÷åíèþ è ýêñïåðòíîé îöåíêå ýôôåêòèâíîñòè è áåçîïàñíîñòè
24. Validation of Analytical Procedures Q2(R1). International Conference ëåêàðñòâåííûõ ñðåäñòâ» (¹ Ãîñ. ðåãèñòðàöèè 01201172531).
on Harmonisation of Technical Requirements for Registration of
АНТИБИОТИКИ И ХИМИОТЕРАПИЯ, 2013, 58; 9—10 55
You might also like
- Izuchenie Effektivnosti Larifana Pri Eksperimentalnoy Forme Tyazhyologo Ostrogo Respiratornogo SindromaDocument5 pagesIzuchenie Effektivnosti Larifana Pri Eksperimentalnoy Forme Tyazhyologo Ostrogo Respiratornogo SindromaМихалычNo ratings yet
- Ìîëåêóëÿpíî-Áèîëîãè÷åcêèå Ïpîáëåìû Cîçäàíèÿ Ëåêàpcòâåííûx Cpåäcòâ È Èçó÷åíèå Ìåxàíèçìà Èx ÄåécòâèÿDocument5 pagesÌîëåêóëÿpíî-Áèîëîãè÷åcêèå Ïpîáëåìû Cîçäàíèÿ Ëåêàpcòâåííûx Cpåäcòâ È Èçó÷åíèå Ìåxàíèçìà Èx ÄåécòâèÿGulya GanapatiNo ratings yet
- Isp 2013 2 13Document3 pagesIsp 2013 2 13ОляNo ratings yet
- V. S. Efimov, A. V. LaptevaDocument7 pagesV. S. Efimov, A. V. LaptevaElena BejanNo ratings yet
- Prognosis of Vibration Levels of Buildings From Underground Train TrafficDocument3 pagesPrognosis of Vibration Levels of Buildings From Underground Train TrafficOrlanosNo ratings yet
- Self-Management V Sfere Kultury I Iskusstva Kadyrova S V Nemtseva E A Tulchinskiy G LDocument224 pagesSelf-Management V Sfere Kultury I Iskusstva Kadyrova S V Nemtseva E A Tulchinskiy G LМария ШиманскаяNo ratings yet
- 1614-Текст статті-6266-2-10-20131118Document8 pages1614-Текст статті-6266-2-10-20131118Ксенія МодринськаNo ratings yet
- How to open and understand your dishwasherDocument27 pagesHow to open and understand your dishwasherОлександр БіликNo ratings yet
- Competitiveness in The Appointment and Conduct of Examinations in Criminal ProceedingsDocument13 pagesCompetitiveness in The Appointment and Conduct of Examinations in Criminal ProceedingsСергей АлексеевNo ratings yet
- Adgeziya V Biologii RakaDocument6 pagesAdgeziya V Biologii RakaПетя СергуферманNo ratings yet
- Pioneer Pdr-w839 ManualDocument52 pagesPioneer Pdr-w839 ManualAndrew LebedevNo ratings yet
- Ilovepdf MergedDocument3 pagesIlovepdf MergedSübhan ŞirinovNo ratings yet
- Levofloxacin (Tavanic) in Complex Therapy of Tuberculosis: G. B. Sokolova, A. D. Kunichan, Ya. V. LazarevaDocument7 pagesLevofloxacin (Tavanic) in Complex Therapy of Tuberculosis: G. B. Sokolova, A. D. Kunichan, Ya. V. LazarevaИрина ТрегубоваNo ratings yet
- Exu S 22 RuDocument112 pagesExu S 22 RuЕвгNo ratings yet
- Fire Hazard of VodkaDocument4 pagesFire Hazard of VodkaСергей АлексеевNo ratings yet
- Архетипи Української Народної КазкиDocument6 pagesАрхетипи Української Народної КазкиАндрій ЗдіховськийNo ratings yet
- Parole from Additional PunishmentDocument6 pagesParole from Additional PunishmentoleNo ratings yet
- Italian Experience of Jet-Grouting Using: (Russian Version PDFDocument14 pagesItalian Experience of Jet-Grouting Using: (Russian Version PDFtomoralNo ratings yet
- 10Document10 pages10Валерія БеспаловаNo ratings yet
- Ispolzovanie Rastitelnyh Ekstraktov V Sozdanii Lekarstvennyh Sredstv Raznoy Terapevticheskoy NapravlennostiDocument5 pagesIspolzovanie Rastitelnyh Ekstraktov V Sozdanii Lekarstvennyh Sredstv Raznoy Terapevticheskoy NapravlennostiDana Jumabai (JH)No ratings yet
- Karakachan Dog Breed ConservationDocument5 pagesKarakachan Dog Breed ConservationVenelin DintchevNo ratings yet
- Komatsu Pc35r 8 Pc45r 8 Shop Manual Rus Wrbm000301Document20 pagesKomatsu Pc35r 8 Pc45r 8 Shop Manual Rus Wrbm000301laverne100% (50)
- Creative Process CriteriaDocument6 pagesCreative Process Criteriaw.exp69No ratings yet
- Contemporary aspects of treating combustion injuries using Aquacel Ag silver impregnated gauzeDocument2 pagesContemporary aspects of treating combustion injuries using Aquacel Ag silver impregnated gauzeMDNo ratings yet
- Exu-H Um Ru 200701 4492118Document106 pagesExu-H Um Ru 200701 4492118ЕвгNo ratings yet
- Ð Ð Ñ Ñ Ñ Ñ ÐºÑ Ð Ñ Indesit WGS - 434 - TX - WGS - 632 - TX - WGS - 636 - TX - 2Document26 pagesÐ Ð Ñ Ñ Ñ Ñ ÐºÑ Ð Ñ Indesit WGS - 434 - TX - WGS - 632 - TX - WGS - 636 - TX - 2elinka191002No ratings yet
- 11 ACE InhibitoriteDocument20 pages11 ACE InhibitoriteNelly ChinelliNo ratings yet
- NVKHP 2015 2 (1) 15Document6 pagesNVKHP 2015 2 (1) 15hhNo ratings yet
- Ïðîêóðîðñêèé Íàäçîð Êàê Ñðåäñòâî Çàùèòû Æèëèùíûõ Ïðàâ Äåòåé-Ñèðîò Prosecutor's Supervision As A Means of Protecting The Housing Rights of OrphansDocument4 pagesÏðîêóðîðñêèé Íàäçîð Êàê Ñðåäñòâî Çàùèòû Æèëèùíûõ Ïðàâ Äåòåé-Ñèðîò Prosecutor's Supervision As A Means of Protecting The Housing Rights of OrphansМакар РогатковNo ratings yet
- Elibrary 41141734 66355416Document4 pagesElibrary 41141734 66355416Макар РогатковNo ratings yet
- 6D140 Service ManualDocument353 pages6D140 Service ManualAnonymous Pk3VzhXUGB100% (1)
- Professionalnaya Deformatsiya Lichnosti PsihologaDocument6 pagesProfessionalnaya Deformatsiya Lichnosti Psihologamacabre5679No ratings yet
- O.kurban Skrud GiDocument5 pagesO.kurban Skrud Gisvitlana melnykNo ratings yet
- Matrix SDocument12 pagesMatrix SEmilian Angelov AngelovNo ratings yet
- GRAN InfoDocument8 pagesGRAN InfogoalexNo ratings yet
- K 4Document432 pagesK 4Matthias BielickiNo ratings yet
- Instruktsiya K Stiral Noy Mashine Indesit Wite 107Document12 pagesInstruktsiya K Stiral Noy Mashine Indesit Wite 107Masha PapushNo ratings yet
- Guide to operating a washing machineDocument24 pagesGuide to operating a washing machineMKNo ratings yet
- Horst Zibert-Efekt KobriDocument239 pagesHorst Zibert-Efekt KobriTomas JuleaNo ratings yet
- ThermoGuard TG-VI Microprocessor Controller - TK41168-2-OD - 12-98 RUDocument193 pagesThermoGuard TG-VI Microprocessor Controller - TK41168-2-OD - 12-98 RUthermo king100% (1)
- Elibrary 21306397 65895364Document5 pagesElibrary 21306397 65895364islezrbsNo ratings yet
- Razvitie Modulnoy Sistemy Proektirovaniya V Graficheskom Dizayne Shveytsarii Ot Novoy Tipografiki K Mezhdunarodnomu StilyuDocument9 pagesRazvitie Modulnoy Sistemy Proektirovaniya V Graficheskom Dizayne Shveytsarii Ot Novoy Tipografiki K Mezhdunarodnomu Stilyulosapro2No ratings yet
- Hotpoint-Ariston AQXD 129 (EU) Washing MachineDocument48 pagesHotpoint-Ariston AQXD 129 (EU) Washing MachineVasile CurmeiNo ratings yet
- 3 2006marukhinDocument6 pages3 2006marukhinFilippo OmbunNo ratings yet
- Methodology of The Conflicts Study in The DomesticDocument6 pagesMethodology of The Conflicts Study in The DomesticJuliana SchwarzNo ratings yet
- Dvata PatiaDocument79 pagesDvata Patiapreslava_19No ratings yet
- каталог 3519Document384 pagesкаталог 3519Радислав ЈовковићNo ratings yet
- Woi Oa S0 OH3 R0 TV PVQPZ CAYr 6 A GJ36 S PLDocument4 pagesWoi Oa S0 OH3 R0 TV PVQPZ CAYr 6 A GJ36 S PLDenisNo ratings yet
- Terminological Problems in Social Sciences Due to Polysemy and Reluctance to Adopt NeologismsDocument7 pagesTerminological Problems in Social Sciences Due to Polysemy and Reluctance to Adopt NeologismsЕкатерина КожемякинаNo ratings yet
- PDF Created With Pdffactory Pro Trial VersionDocument153 pagesPDF Created With Pdffactory Pro Trial VersionskaspeNo ratings yet
- Ìåæãîñóäàðñòâåííûé Ñîâåò Ïî Ñòàíäàðòèçàöèè, Ìåòðîëîãèè È Ñåðòèôèêàöèè (ÌÃÑ) Interstate Council For Standardization, Metrology and Certification (ISC)Document24 pagesÌåæãîñóäàðñòâåííûé Ñîâåò Ïî Ñòàíäàðòèçàöèè, Ìåòðîëîãèè È Ñåðòèôèêàöèè (ÌÃÑ) Interstate Council For Standardization, Metrology and Certification (ISC)dfdffdfsafdfde4tf4fNo ratings yet
- O Praktike Preduprezhdeniya Prestupleniy V Sfere Informatsionnyh Tehnologiy Vo VietnameDocument3 pagesO Praktike Preduprezhdeniya Prestupleniy V Sfere Informatsionnyh Tehnologiy Vo VietnameLưu LyNo ratings yet
- Water Revolution SystemDocument1 pageWater Revolution SystemSofoklhs ChernobylNo ratings yet
- My-Pedagogical-Credo - Content File PDFDocument10 pagesMy-Pedagogical-Credo - Content File PDFDimitar BojkovNo ratings yet
- Book of TothDocument63 pagesBook of TothПетя БоневаNo ratings yet
- Hotpoint Ariston Avtf 109Document72 pagesHotpoint Ariston Avtf 109Reborn UkraineNo ratings yet
- Belarusian Literature (5 Grade)Document176 pagesBelarusian Literature (5 Grade)ruslanvershinin1995No ratings yet
- Under Supervision of Dr. Ommia AliDocument15 pagesUnder Supervision of Dr. Ommia AliAbdelrhman AboodaNo ratings yet
- Health: Quarter 1 - Module 5: Health ServicesDocument24 pagesHealth: Quarter 1 - Module 5: Health ServicesJonathan Bulawan80% (5)
- Procedures For Administering Injectable Drugs: Version No 2Document201 pagesProcedures For Administering Injectable Drugs: Version No 2farklinRSBKNo ratings yet
- Depo Medrol 80 MG - Google SearchDocument1 pageDepo Medrol 80 MG - Google SearchMohammad AamirNo ratings yet
- Evaluating Exam Review and Guide 2018 PDFDocument762 pagesEvaluating Exam Review and Guide 2018 PDFAthullia George83% (24)
- Pharmaceutical IndustryDocument11 pagesPharmaceutical IndustryAbid Ali KhanNo ratings yet
- Focus On Nursing Pharmacology 6th Edition Karch Test BankDocument35 pagesFocus On Nursing Pharmacology 6th Edition Karch Test Bankkieucharmaines1w100% (32)
- Antibiotic Approval TimelineDocument1 pageAntibiotic Approval TimelineMartin CuellarNo ratings yet
- FDA FOIA Log - May 2022Document132 pagesFDA FOIA Log - May 2022Mayra MayaNo ratings yet
- Opd MSDocument35 pagesOpd MSkanikaNo ratings yet
- FDA's Current Practice and Challenges in The Use of Dissolution Similarity Testing For Demonstration of Bioequivalence - Case StudiesDocument19 pagesFDA's Current Practice and Challenges in The Use of Dissolution Similarity Testing For Demonstration of Bioequivalence - Case StudiesSrinivas Reddy MaramNo ratings yet
- Displec m1 SummaryDocument10 pagesDisplec m1 SummaryRafaelaNo ratings yet
- Catalyst RX Formulary IntroductionDocument4 pagesCatalyst RX Formulary IntroductionRahul MishraNo ratings yet
- Pharmacogenetics Drug Drug Interactions1Document16 pagesPharmacogenetics Drug Drug Interactions1Marfu'ah Mar'ahNo ratings yet
- Naplexq&aDocument81 pagesNaplexq&aRamy Raafat Rezk100% (4)
- Pima County Board of Supervisors - COVID-19 Emergency DeclarationDocument4 pagesPima County Board of Supervisors - COVID-19 Emergency DeclarationLauren Renteria100% (1)
- Answer Sheet Interpreting Prescription OrdersDocument3 pagesAnswer Sheet Interpreting Prescription OrdersAUDREY ARQUERONo ratings yet
- 10 Rights in Drug AdministrationDocument25 pages10 Rights in Drug AdministrationNathaniel PulidoNo ratings yet
- Formulary Thera Class PDFDocument33 pagesFormulary Thera Class PDFElla LopezNo ratings yet
- Dokumen - Tips - Bpharm-7-Semester-Project-Reportfinal 2 PDFDocument25 pagesDokumen - Tips - Bpharm-7-Semester-Project-Reportfinal 2 PDFvishal sharmaNo ratings yet
- Vaccination RecordDocument47 pagesVaccination RecordJolleah AujeroNo ratings yet
- REGULATORYDocument19 pagesREGULATORYsrishty100% (1)
- Agatha Christie-The Symbol of Literature: "Elena Cuza" National CollegeDocument13 pagesAgatha Christie-The Symbol of Literature: "Elena Cuza" National CollegeAsd FghNo ratings yet
- Practical Pharmaceutics: Yvonne Bouwman-Boer V Iain Fenton-May Paul Le Brun EditorsDocument873 pagesPractical Pharmaceutics: Yvonne Bouwman-Boer V Iain Fenton-May Paul Le Brun Editorssalah100% (4)
- Applied Biopharmaceutics & Pharmacokinetics, 5th EditionDocument1 pageApplied Biopharmaceutics & Pharmacokinetics, 5th Editionbencleese14% (7)
- OxyContin TitrationDocument2 pagesOxyContin TitrationguntherfurlongNo ratings yet
- DECLOBANDocument2 pagesDECLOBANCamilleNo ratings yet
- Tocilizumab Distributor ListDocument25 pagesTocilizumab Distributor ListAkshayRautNo ratings yet
- MD II S1-Basic Hospital VocabularyDocument5 pagesMD II S1-Basic Hospital VocabularyDANIEL-NICOLAE MARINNo ratings yet
- 2018 Curriculum VitaeDocument5 pages2018 Curriculum Vitaeapi-402947573No ratings yet