Professional Documents
Culture Documents
Beauty Cream
Uploaded by
muhammad imran aziz0 ratings0% found this document useful (0 votes)
6 views1 pageThis certificate of analysis summarizes test results for Golden Pearl Whitening Beauty Cream Batch 22PS100. The tests show the cream complies with specifications for appearance, color, odor, pH, thermal stability, absence of hydroquinone and corticosteroids, meets minimum fat content requirements, and contains levels of heavy metals, arsenic, and mercury below limits. Microbiology testing found total viable count and gram-negative pathogens within acceptable levels. The batch was released for use.
Original Description:
Original Title
Beauty cream
Copyright
© © All Rights Reserved
Available Formats
PDF, TXT or read online from Scribd
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentThis certificate of analysis summarizes test results for Golden Pearl Whitening Beauty Cream Batch 22PS100. The tests show the cream complies with specifications for appearance, color, odor, pH, thermal stability, absence of hydroquinone and corticosteroids, meets minimum fat content requirements, and contains levels of heavy metals, arsenic, and mercury below limits. Microbiology testing found total viable count and gram-negative pathogens within acceptable levels. The batch was released for use.
Copyright:
© All Rights Reserved
Available Formats
Download as PDF, TXT or read online from Scribd
0 ratings0% found this document useful (0 votes)
6 views1 pageBeauty Cream
Uploaded by
muhammad imran azizThis certificate of analysis summarizes test results for Golden Pearl Whitening Beauty Cream Batch 22PS100. The tests show the cream complies with specifications for appearance, color, odor, pH, thermal stability, absence of hydroquinone and corticosteroids, meets minimum fat content requirements, and contains levels of heavy metals, arsenic, and mercury below limits. Microbiology testing found total viable count and gram-negative pathogens within acceptable levels. The batch was released for use.
Copyright:
© All Rights Reserved
Available Formats
Download as PDF, TXT or read online from Scribd
You are on page 1of 1
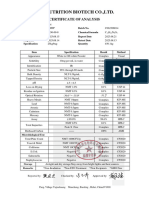
Certificate of Analysis
Document Code: Version #: Effective Date: Revision Date:
GPC/QC/SOR/06 02 24-01-2018 28-02-2020
Product Name GOLDEN PEARL WHITENING BEAUTY CREAM Batch No. 22PS100
Batch Size:20Kg Bulk Mfg. Date:16-11-2022 Date of Analysis: 16-11-2022 QC No. GPC-FP-0327-22 Stage: Bulk
Test Performed Specifications Results
Slightly yellowish viscous mass that must comply
Appearance Complies
with reference
Slightly yellowish that must comply with
Color Complies
reference
Odor Typical fruity that must comply with reference Complies
pH range 4.0-9.0 6.00
Thermal stability To pass the test Pass
Hydroquinone Shall be absent Absent
Corticosteroid Substance Shall be absent Absent
Total fatty substance content
percent by mass, minimum 5.0% 15.55%
Heavy Metals
As Lead Max Maximum 20ppm <1.2ppm
Arsenic (as As2O3), ppm, Max Maximum 2ppm <0.03ppm
Mercury (as Hg) ppm, Max Maximum 1 <0.01ppm
Microbiology Testing
Total Viable count NMT1000cfu/g <100cfu/g
Gram Negative Pathogens NMT10cfu/g Nil
Remarks: Released for use
Analyzed By: (Officer QC)
You might also like
- COA of Calcium D Pantothente - RevisedDocument1 pageCOA of Calcium D Pantothente - RevisedJose.SuarezNo ratings yet
- Certificate of Analysis: Sunflower Oil RefinedDocument1 pageCertificate of Analysis: Sunflower Oil RefinedBetrand MuNo ratings yet
- Ascorbic Acid-Shandong LuweiDocument1 pageAscorbic Acid-Shandong LuweiSai Kiran PalikaNo ratings yet
- Xi'an Haoxuan Bio-Tech Co.,Ltd: Certificate of AnylasisDocument1 pageXi'an Haoxuan Bio-Tech Co.,Ltd: Certificate of AnylasisNilsNo ratings yet
- CoA Sucralose - Supplier Tokped (Shandong Kanbo)Document1 pageCoA Sucralose - Supplier Tokped (Shandong Kanbo)Tantriyani GunadyNo ratings yet
- COA Coco Betaine 40% PDFDocument1 pageCOA Coco Betaine 40% PDFdeepak tatheNo ratings yet
- Coa 3220141170Document1 pageCoa 3220141170Andres FarfanNo ratings yet
- Parameter USP 34 Specification Reagent Plus (C0750) Caffeine Sigma Reference Standard (C1778)Document2 pagesParameter USP 34 Specification Reagent Plus (C0750) Caffeine Sigma Reference Standard (C1778)rushikeshghuleNo ratings yet
- COA - Ascorbic Acid, B# 1101070022-2Document1 pageCOA - Ascorbic Acid, B# 1101070022-2Aboudeh FarranNo ratings yet
- Certificate of Analysis: Polysorbate 80Document1 pageCertificate of Analysis: Polysorbate 80Nurul HidayatriNo ratings yet
- PS 005 - AyuFlexDocument1 pagePS 005 - AyuFlexLuis Castro XtrmNo ratings yet
- SNC - COA - Toltrazuril 98 - 102%Document1 pageSNC - COA - Toltrazuril 98 - 102%ben grugeNo ratings yet
- Certificate of Analysis: 17α-Hydroxy Progesterone AcetateDocument2 pagesCertificate of Analysis: 17α-Hydroxy Progesterone Acetatewindli2012No ratings yet
- Certificado de Analisis: - Certificate of Analysis As Received From Our Supplier - Productos Químicos Gonmisol S.A.Document2 pagesCertificado de Analisis: - Certificate of Analysis As Received From Our Supplier - Productos Químicos Gonmisol S.A.pervaz anwerNo ratings yet
- Coa - Cocoa Ah01 OctubreDocument2 pagesCoa - Cocoa Ah01 OctubreChristopher CaizaNo ratings yet
- Certificate of Analysis: Hubei Xingfa Chemicals Group Co., LTDDocument1 pageCertificate of Analysis: Hubei Xingfa Chemicals Group Co., LTDSpecialty Chemicals100% (1)
- COA Garcinia Cambogia Extract HCA 60Document1 pageCOA Garcinia Cambogia Extract HCA 60Ali Zainal Abidin100% (1)
- Lampiran 3 Dan 4 Proposal DisertasiDocument2 pagesLampiran 3 Dan 4 Proposal DisertasioktariyanaNo ratings yet
- 18mt COA 2210142035Document1 page18mt COA 2210142035nadia ARJDALNo ratings yet
- Auro Labs LTD - Metf HCL USPDocument1 pageAuro Labs LTD - Metf HCL USPsuriana limNo ratings yet
- UntitledDocument3 pagesUntitledDIANELANo ratings yet
- CoA FlexTabDocument2 pagesCoA FlexTabM Zinedine Haryanto SIG LabNo ratings yet
- Certificate of Analysis PDFDocument2 pagesCertificate of Analysis PDFadiNo ratings yet
- Ginseng Leaf ExtractDocument1 pageGinseng Leaf Extractyousfinadjah5No ratings yet
- SampleDocument2 pagesSampleSeara FerminoNo ratings yet
- Coa Polivinilpirrolidona K-90 (PVP K-90) Lote 20221019Document1 pageCoa Polivinilpirrolidona K-90 (PVP K-90) Lote 20221019Ives AlbarracinNo ratings yet
- D Panthenol COADocument1 pageD Panthenol COAwillyvh99No ratings yet
- Coa 76643Document1 pageCoa 76643Rabah ABBASNo ratings yet
- Toyond Industry Limited: Certificate of AnalysisDocument2 pagesToyond Industry Limited: Certificate of AnalysisQf Jhon DonadoNo ratings yet
- COA LonganDocument1 pageCOA LonganBoonyi UnaiNo ratings yet
- COA of Raw MaterialDocument10 pagesCOA of Raw MaterialShafaq ALINo ratings yet
- COA OF MENTHOL 薄荷脑Document1 pageCOA OF MENTHOL 薄荷脑Gioacchino MondelloNo ratings yet
- COA-Carnitine HCL-HengtaiDocument1 pageCOA-Carnitine HCL-Hengtaichurch.hrgNo ratings yet
- Pyrazinamida CoaDocument1 pagePyrazinamida Coawillyvh99No ratings yet
- CoA of EgcgDocument1 pageCoA of EgcgMirna Candra RNo ratings yet
- 5-HTP CoaDocument1 page5-HTP Coawillyvh99No ratings yet
- COA Panax Ginseng ExtDocument1 pageCOA Panax Ginseng ExtFerdian Iwank IriyantoNo ratings yet
- AKBA - 10% CoaDocument1 pageAKBA - 10% CoaDeepak VarmaNo ratings yet
- P18020321 #0 Opq Yl - Opq YlDocument2 pagesP18020321 #0 Opq Yl - Opq YlZainab aboodNo ratings yet
- SAPP FG - SpecificationDocument1 pageSAPP FG - SpecificationNha TranNo ratings yet
- 1 Coa-DcpDocument1 page1 Coa-DcpjivaorganicNo ratings yet
- Alginat-Loba ChemicalDocument2 pagesAlginat-Loba ChemicalNur NurkurniaNo ratings yet
- Pot Clave Syloid B# 403Document1 pagePot Clave Syloid B# 403citi pharmaNo ratings yet
- Maltodextrina Specs - OmniaDocument3 pagesMaltodextrina Specs - OmniaEmanuel Juan Flores ArbulúNo ratings yet
- Vitamin CDocument2 pagesVitamin Cbejoykarim2022No ratings yet
- Certificate of Analysis: Calcium GluconateDocument1 pageCertificate of Analysis: Calcium GluconateEfrain Mendoza MartinezNo ratings yet
- Naturecare Pearly Shampoo (Formulation # HB-DE-07-159-18)Document1 pageNaturecare Pearly Shampoo (Formulation # HB-DE-07-159-18)Patrick FlowerdayNo ratings yet
- BAICAO COA of Evening Primrose OilDocument1 pageBAICAO COA of Evening Primrose OilPolyfine Nutra-SciencesNo ratings yet
- Raw Material Analytical Report of Ciprofloxacin HCLDocument1 pageRaw Material Analytical Report of Ciprofloxacin HCLbejoykarim2022No ratings yet
- Certificate of AnalysisDocument1 pageCertificate of AnalysisDana JuarezNo ratings yet
- Coa Sorbitol-70-LiquidDocument2 pagesCoa Sorbitol-70-LiquidAndriyanto HadiNo ratings yet
- Coa 0893Document1 pageCoa 0893zahid yousufNo ratings yet
- 1, Water Soluble Ginkgo Biloba ExtractDocument1 page1, Water Soluble Ginkgo Biloba ExtractdanilriosNo ratings yet
- Ascorbic Acid (Vitamin C)Document2 pagesAscorbic Acid (Vitamin C)ASHOK KUMAR LENKANo ratings yet
- COA Pelargonium Sidoides Extract4-1Document1 pageCOA Pelargonium Sidoides Extract4-1Bilal MasoodNo ratings yet
- Arges Life Science Limited: Standard Control SpecificationDocument1 pageArges Life Science Limited: Standard Control Specificationইমন বড়ুয়া মইন্যাNo ratings yet
- Citric Acid Anhydrate SpesifikasiDocument1 pageCitric Acid Anhydrate SpesifikasicinnamaldehidNo ratings yet
- Raw Material Analytical Report of Ciprofloxacin HCLDocument1 pageRaw Material Analytical Report of Ciprofloxacin HCLbejoykarim2022No ratings yet
- AllantoinDocument1 pageAllantoinmuhammad imran azizNo ratings yet
- Acrypol 940 - Batch No.11018064Document1 pageAcrypol 940 - Batch No.11018064muhammad imran azizNo ratings yet
- Over Time in The MoralDocument1 pageOver Time in The Moralmuhammad imran azizNo ratings yet
- Internal Audit - 19!08!2022-GP ISO 17025 SlidesDocument33 pagesInternal Audit - 19!08!2022-GP ISO 17025 Slidesmuhammad imran azizNo ratings yet
- Pearson Test of English PDFDocument14 pagesPearson Test of English PDFmuhammad imran azizNo ratings yet
- Directory of Licenced Commercial Banks Authorised NOHCs Jan 2023Document19 pagesDirectory of Licenced Commercial Banks Authorised NOHCs Jan 2023Ajai Suman100% (1)
- 123 AP / 0123 TS: Intermediate Public Examinations - 2024Document3 pages123 AP / 0123 TS: Intermediate Public Examinations - 2024srinivasveeravalli123aNo ratings yet
- Resume of Shabina SanadDocument4 pagesResume of Shabina SanadShabina SanadNo ratings yet
- Crazy in Love-KadebostanyDocument2 pagesCrazy in Love-Kadebostanyvikas kumawatNo ratings yet
- Grade 6 First Periodical Test in EPPDocument6 pagesGrade 6 First Periodical Test in EPPPrecilla Halago88% (8)
- 5Cosc020W Database Systems - Lecture 05Document39 pages5Cosc020W Database Systems - Lecture 05Sanjeeban NiranjankumarNo ratings yet
- Moxa Iologik E1200 Series Manual v15.2Document129 pagesMoxa Iologik E1200 Series Manual v15.2yeng menNo ratings yet
- 1 Whats NewDocument76 pages1 Whats NewTirupati MotiNo ratings yet
- Highland GamesDocument52 pagesHighland GamesolazagutiaNo ratings yet
- Technical Analysis of The Size PremiumDocument4 pagesTechnical Analysis of The Size PremiumRamiro Gamen0% (1)
- Phytoplankton As Bioindicators of Pollution Status in Cirata Reservoir, IndonesiaDocument5 pagesPhytoplankton As Bioindicators of Pollution Status in Cirata Reservoir, IndonesiajoelNo ratings yet
- The Minoan Harem: The Role of Eminent Women and The Knossos FrescoesDocument31 pagesThe Minoan Harem: The Role of Eminent Women and The Knossos FrescoesAlexandros KastanakisNo ratings yet
- Instruction Guide: Playing VolleyballDocument5 pagesInstruction Guide: Playing Volleyballapi-606741192No ratings yet
- Sas Open Source Integration 112134Document21 pagesSas Open Source Integration 112134PapasimaNo ratings yet
- Contrasting MO and VB TheoryDocument4 pagesContrasting MO and VB TheoryPhillimonNo ratings yet
- Rms Due1012Document8 pagesRms Due1012api-273006352No ratings yet
- The Daily Tar Heel For February 1, 2011Document8 pagesThe Daily Tar Heel For February 1, 2011The Daily Tar HeelNo ratings yet
- 1015 - Easement, Nature and Characteristics and ExtinctionDocument25 pages1015 - Easement, Nature and Characteristics and ExtinctionRutwik JoshiNo ratings yet
- Mts 2003 25 1 167Document37 pagesMts 2003 25 1 167Yuri BaerNo ratings yet
- Predestination Vs Free WillDocument4 pagesPredestination Vs Free WillFrancis Heckman100% (2)
- Big Dog's Leadership Page - Presentation Skills: OutlineDocument16 pagesBig Dog's Leadership Page - Presentation Skills: OutlinekathirNo ratings yet
- Sentence Transformation - Perfect TensesDocument3 pagesSentence Transformation - Perfect TensesMaría Esperanza Velázquez Castillo100% (2)
- Pdi Partial EdentulismDocument67 pagesPdi Partial EdentulismAnkeeta ShuklaNo ratings yet
- Document No.: Safety CheckDocument14 pagesDocument No.: Safety CheckRodolfo Alberto Muñoz CarcamoNo ratings yet
- Disassembly and Assembly: 1106D Industrial EngineDocument185 pagesDisassembly and Assembly: 1106D Industrial EnginefranNo ratings yet
- Price Reference Guide For Security Services 2021Document4 pagesPrice Reference Guide For Security Services 2021Dulas DulasNo ratings yet
- Science, Technoloy and Society Prelim ExamDocument14 pagesScience, Technoloy and Society Prelim ExamIman Official (Em-J)No ratings yet
- Is Your: WritingDocument84 pagesIs Your: WritingVito a sapada Arung wetteNo ratings yet
- Irjece: Human Computer Interaction Direct ManipulationDocument4 pagesIrjece: Human Computer Interaction Direct ManipulationgigibowonseetNo ratings yet
- 1 Manalo Vs Sistoza DigestDocument2 pages1 Manalo Vs Sistoza DigestRowell Ian Gana-an0% (1)