Professional Documents
Culture Documents
Ácidos y Bases - PH
Ácidos y Bases - PH
Uploaded by
hotmuysoy0 ratings0% found this document useful (0 votes)
13 views13 pagesThis document discusses acids and bases and pH. It defines pH as the negative logarithm of the hydrogen ion concentration and pOH as the negative logarithm of the hydroxide ion concentration. It also states that the sum of pH and pOH equals 14.
Original Description:
Original Title
Ácidos y bases _ PH
Copyright
© © All Rights Reserved
Available Formats
PDF, TXT or read online from Scribd
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentThis document discusses acids and bases and pH. It defines pH as the negative logarithm of the hydrogen ion concentration and pOH as the negative logarithm of the hydroxide ion concentration. It also states that the sum of pH and pOH equals 14.
Copyright:
© All Rights Reserved
Available Formats
Download as PDF, TXT or read online from Scribd
0 ratings0% found this document useful (0 votes)
13 views13 pagesÁcidos y Bases - PH
Ácidos y Bases - PH
Uploaded by
hotmuysoyThis document discusses acids and bases and pH. It defines pH as the negative logarithm of the hydrogen ion concentration and pOH as the negative logarithm of the hydroxide ion concentration. It also states that the sum of pH and pOH equals 14.
Copyright:
© All Rights Reserved
Available Formats
Download as PDF, TXT or read online from Scribd
You are on page 1of 13
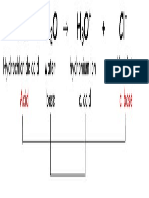
Ácidos y bases / PH
Ph = -log[H+] pOH: -log[OH-]
Ph + pOH = 14
You might also like
- Conjugate Acid Base Pairs: Name - Chem Worksheet 19-2Document2 pagesConjugate Acid Base Pairs: Name - Chem Worksheet 19-2Taylor Delancey100% (2)
- Chapter 16-Acids and BasesDocument33 pagesChapter 16-Acids and BasesGörkem DamdereNo ratings yet
- 100 Hour and 100 Marks PDF With WatermarkDocument8 pages100 Hour and 100 Marks PDF With WatermarkRAHUL SHUKLANo ratings yet
- Chapter 1 NotesDocument8 pagesChapter 1 NotesYuHangTiNo ratings yet
- Unit: Acids, Bases, and SolutionsDocument16 pagesUnit: Acids, Bases, and Solutionssana iqbalNo ratings yet
- Peta Konsep: Kekuatan Asam Basa Dan PH LarutanDocument1 pagePeta Konsep: Kekuatan Asam Basa Dan PH LarutannadilafitrianiNo ratings yet
- 1-Neutralization Theory2Document24 pages1-Neutralization Theory2watersoul.nNo ratings yet
- Ionic Equilibrium: Beginning of HydrolysisDocument8 pagesIonic Equilibrium: Beginning of HydrolysisPiyush GargNo ratings yet
- Homework 10: SO 4) H 2 S) Log SO 4) H2S) Log SO 4) H2S) SO 4) H 2 SDocument3 pagesHomework 10: SO 4) H 2 S) Log SO 4) H2S) Log SO 4) H2S) SO 4) H 2 SKamran AliNo ratings yet
- Acids BasesDocument30 pagesAcids BasesHaniel GalzoteNo ratings yet
- دروس الزوم الوحدة الثانية العلاقة بين بنية ووظيفة البروتين للأستاذDocument10 pagesدروس الزوم الوحدة الثانية العلاقة بين بنية ووظيفة البروتين للأستاذayadilyes79No ratings yet
- Teori Asam Basa # Penentuan Larutan Asam-BasaDocument1 pageTeori Asam Basa # Penentuan Larutan Asam-BasaintanchairunnisaNo ratings yet
- UntitledDocument3 pagesUntitledMaria Zarah Chrisanta AquinoNo ratings yet
- Carbohydrate Metabolism Compiled 1KSDDocument11 pagesCarbohydrate Metabolism Compiled 1KSDHarsh KataraNo ratings yet
- Conjugate Acid Base Pairs: Name - Chem Worksheet 19-2Document1 pageConjugate Acid Base Pairs: Name - Chem Worksheet 19-2Bindu Mohanan PillaiNo ratings yet
- Hydrolysis 47Document2 pagesHydrolysis 47ipsita lahiriNo ratings yet
- Water Chemistry: PH - Log (H)Document2 pagesWater Chemistry: PH - Log (H)Deth-Deth Ramilo MolinaNo ratings yet
- Glycolysis Schematic 2010Document1 pageGlycolysis Schematic 2010ykhan2No ratings yet
- Chapter 3 - Concept of Acid-Base NeutralisationDocument58 pagesChapter 3 - Concept of Acid-Base NeutralisationIkmal FikriNo ratings yet
- Bio 024 - Session 2 Sas Nursing (New Format) - WatermarkDocument6 pagesBio 024 - Session 2 Sas Nursing (New Format) - WatermarkMaria Vannesa Anne SalvacionNo ratings yet
- Phương Trình Con Đư NG EMPDocument2 pagesPhương Trình Con Đư NG EMPDương TháiNo ratings yet
- 5 Shikimic Acid Pathway Lecture 3Document19 pages5 Shikimic Acid Pathway Lecture 3Shar KhanNo ratings yet
- Materi PJJ Phenolic 8 April 2020Document24 pagesMateri PJJ Phenolic 8 April 2020AyuNo ratings yet
- Aldehydes and KetonesDocument5 pagesAldehydes and KetonesBaji Babu BejjankiNo ratings yet
- 1composition 1er Trimestre Lyce Ibn Khatab 2007 2008Document2 pages1composition 1er Trimestre Lyce Ibn Khatab 2007 2008mechkourNo ratings yet
- Acids and Bases KODocument1 pageAcids and Bases KOabhishektheoneNo ratings yet
- Mod2 PDFDocument10 pagesMod2 PDFPrince SinghNo ratings yet
- Chemistry (Full Test) - Paper 2 - SolutionsDocument5 pagesChemistry (Full Test) - Paper 2 - SolutionsRavi Kiran KoduriNo ratings yet
- Asset-V1 UTokyoX+UTokyo007x+1T2018+Type@Asset+Block@Unit 10 Polyprotic Acid-Base EquilibriaDocument5 pagesAsset-V1 UTokyoX+UTokyo007x+1T2018+Type@Asset+Block@Unit 10 Polyprotic Acid-Base EquilibriaVíctor MartínezNo ratings yet
- 9 GlycogenDocument36 pages9 GlycogenSneha Sagar SharmaNo ratings yet
- Ch18Acid Base (A)Document45 pagesCh18Acid Base (A)Michael Conan MaglaqueNo ratings yet
- PH PresDocument8 pagesPH PrestalktotiffanychengNo ratings yet
- Definitions - Topic 12 Acid-Base Equilibria - Edexcel Chemistry A-LevelDocument2 pagesDefinitions - Topic 12 Acid-Base Equilibria - Edexcel Chemistry A-LevelsalmaNo ratings yet
- Pentose Phosphate Pathway: Molecular Biochemistry IIDocument27 pagesPentose Phosphate Pathway: Molecular Biochemistry IIElena PavlicNo ratings yet
- 25.ray ArraneryDocument2 pages25.ray ArraneryKim ParlNo ratings yet
- Chapter3 PKa TableDocument3 pagesChapter3 PKa Tablealexander ramosNo ratings yet
- Pka Table PDFDocument3 pagesPka Table PDFatharvalipareNo ratings yet
- Chapter3-pKa Table PDFDocument3 pagesChapter3-pKa Table PDFAnilesh BansalNo ratings yet
- Acid Base Dissociation ConstantDocument28 pagesAcid Base Dissociation Constantlily augustNo ratings yet
- 14 Acids, Bases and SaltsDocument105 pages14 Acids, Bases and Saltsmika yzobelNo ratings yet
- Binas 6e Druk 68BDocument1 pageBinas 6e Druk 68BGwen Van de WallNo ratings yet
- Nucleotides Metabolism StudDocument41 pagesNucleotides Metabolism Studsyarifah aryaniNo ratings yet
- 1.nucleotide MetabolismDocument37 pages1.nucleotide Metabolismlucky mbaselaNo ratings yet
- Synthesis of Furan and ThiopheneDocument10 pagesSynthesis of Furan and ThiopheneNorman FerdinalNo ratings yet
- Ch16 - Aq Ionic Equil1Document14 pagesCh16 - Aq Ionic Equil1EdcademiaNo ratings yet
- Conjugate Acid and Base PairsDocument1 pageConjugate Acid and Base Pairsfelisita wisangNo ratings yet
- Glycogen MetabolismDocument37 pagesGlycogen MetabolismERIAS TENYWANo ratings yet
- Bronsted-Lowry Acids and BasesDocument2 pagesBronsted-Lowry Acids and Basessikab31632No ratings yet
- Tabella Pka Molecole OrganicheDocument2 pagesTabella Pka Molecole OrganichePaolo Di PalmaNo ratings yet
- Pages From Buffers - 2 PDFDocument1 pagePages From Buffers - 2 PDFKareem_adel_000No ratings yet
- Lecture 14-Gr 15Document25 pagesLecture 14-Gr 15averagestudent838No ratings yet
- Chapter 3 Acid - BaseDocument98 pagesChapter 3 Acid - BaseKhoa Nguyen Viet DangNo ratings yet
- Structure and Function of CarbohydratesDocument27 pagesStructure and Function of CarbohydratesAli SeenaNo ratings yet
- Cells and Sugars 5-Glycolysis-StudentDocument8 pagesCells and Sugars 5-Glycolysis-StudenttyhbbhhNo ratings yet
- Harper's Illustrated Biochemistry PDFDocument1 pageHarper's Illustrated Biochemistry PDFDilip ReddyNo ratings yet
- PPP1B 8.1 B-L Acids BasesDocument20 pagesPPP1B 8.1 B-L Acids BasesRichard LindemannNo ratings yet
- Chemistry (Full Test) - Paper 1 - SolutionsDocument6 pagesChemistry (Full Test) - Paper 1 - SolutionsRavi Kiran KoduriNo ratings yet
- Acid Base Eqm. Test OutlineDocument1 pageAcid Base Eqm. Test OutlineSara MolinaroNo ratings yet
- Acid Base (A)Document30 pagesAcid Base (A)Heather WrightNo ratings yet
- 4 Redox ChemistryDocument10 pages4 Redox Chemistryu3537671No ratings yet