Professional Documents
Culture Documents
Chemistry Formula1
Chemistry Formula1
Uploaded by
omoak2015Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
Chemistry Formula1
Chemistry Formula1
Uploaded by
omoak2015Copyright:
Available Formats
All Chemistry Formulas for O levels Chemistry by Ethan Wu
Hydrogen carbonate HCO3-
Hydride H-
Hydroxide OH-
Carbonate CO32-
Nitrate NO3-
Nitrite NO2-
Nitride N3-
Phosphate PO43-
Sulfate SO42-
Sulfite SO32-
Sulfide S2-
Ammonia NH3
Ammonium ion NH4+
Mole Concept
One mole of any substance contains 6 X 1023 particles (Avogadro’s number).
𝑛𝑢𝑚𝑏𝑒𝑟 𝑜𝑓 𝑝𝑎𝑟𝑡𝑖𝑐𝑙𝑒𝑠
𝑛𝑢𝑚𝑏𝑒𝑟 𝑜𝑓 𝑚𝑜𝑙𝑒𝑠 =
6 × 1023
The molar mass of an element is the mass of one mole of atoms of the element.
𝑚𝑎𝑠𝑠 𝑜𝑓 𝑡ℎ𝑒 𝑒𝑙𝑒𝑚𝑒𝑛𝑡 𝑖𝑛 𝑔
𝑛𝑢𝑚𝑏𝑒𝑟 𝑜𝑓 𝑚𝑜𝑙𝑒𝑠 𝑜𝑓 𝑎𝑛 𝑒𝑙𝑒𝑚𝑒𝑛𝑡 =
𝑚𝑜𝑙𝑎𝑟 𝑚𝑎𝑠𝑠 𝑜𝑓 𝑡ℎ𝑒 𝑒𝑙𝑒𝑚𝑒𝑛𝑡 𝑖𝑛 𝑔/𝑚𝑜𝑙
Percentage composition of compounds
𝑛𝑢𝑚𝑏𝑒𝑟 𝑜𝑓 𝑎𝑡𝑜𝑚𝑠 𝑜𝑓 𝑒𝑙𝑒𝑚𝑒𝑛𝑡 × 𝐴𝑟
𝑝𝑒𝑟𝑐𝑒𝑛𝑡𝑎𝑔𝑒 𝑏𝑦 𝑚𝑎𝑠𝑠 𝑜𝑓 𝑎𝑛 𝑒𝑙𝑒𝑚𝑒𝑛𝑡 𝑖𝑛 𝑎 𝑐𝑜𝑚𝑝𝑜𝑢𝑛𝑑 = 100%
𝑀𝑟 𝑜𝑓 𝑐𝑜𝑚𝑝𝑜𝑢𝑛𝑑
Empirical formula
Empirical formula of a compound is the simplest formula of the compound
Element Element 1 Element 2
Mass/g or % by mass
Relative atomic mass
Number of moles
Mole ratio (divide by smallest number)
Simplest whole number
To find molecular formula
E.g. let molecular formula of ethane be (CH3)n
Relative mass of ethane from empirical formula = 15
𝑟𝑒𝑙𝑎𝑡𝑖𝑣𝑒 𝑚𝑜𝑙𝑒𝑐𝑢𝑙𝑎𝑟 𝑚𝑎𝑠𝑠 𝑜𝑓 𝑒𝑡ℎ𝑎𝑛𝑒
𝑛=
𝑟𝑒𝑙𝑎𝑡𝑖𝑣𝑒 𝑚𝑎𝑠𝑠 𝑜𝑓 𝑒𝑡ℎ𝑎𝑛𝑒 𝑓𝑟𝑜𝑚 𝑒𝑚𝑝𝑖𝑟𝑖𝑐𝑎𝑙 𝑓𝑜𝑟𝑚𝑢𝑙𝑎
Compiled by Ethan Wu Ying Tang
Molar volume of gases
𝑣𝑜𝑙𝑢𝑚𝑒 𝑜𝑓 𝑔𝑎𝑠 𝑖𝑛 𝑐𝑚3 𝑎𝑡 𝑟. 𝑡. 𝑝.
𝑛𝑢𝑚𝑏𝑒𝑟 𝑜𝑓 𝑚𝑜𝑙𝑒𝑠 𝑜𝑓 𝑔𝑎𝑠 =
24 000 𝑐𝑚3
Concentration of a solution (can be in terms of mol/dm3 or g/dm3)
𝑚𝑎𝑠𝑠 𝑜𝑓 𝑠𝑜𝑙𝑢𝑡𝑒 𝑖𝑛 𝑔
𝑐𝑜𝑛𝑐𝑒𝑛𝑡𝑟𝑎𝑡𝑖𝑜𝑛 𝑖𝑛 𝑔/𝑑𝑚3 =
𝑣𝑜𝑙𝑢𝑚𝑒 𝑜𝑓 𝑠𝑜𝑙𝑢𝑡𝑖𝑜𝑛 𝑖𝑛 𝑑𝑚3
𝑛𝑢𝑚𝑏𝑒𝑟 𝑜𝑓 𝑚𝑜𝑙𝑒𝑠 𝑜𝑓 𝑠𝑜𝑙𝑢𝑡𝑒
𝑐𝑜𝑛𝑐𝑒𝑛𝑡𝑟𝑎𝑡𝑖𝑜𝑛 𝑖𝑛 𝑚𝑜𝑙/𝑑𝑚3 =
𝑣𝑜𝑙𝑢𝑚𝑒 𝑜𝑓 𝑠𝑜𝑙𝑢𝑡𝑖𝑜𝑛 𝑖𝑛 𝑑𝑚3
𝑎𝑐𝑡𝑢𝑎𝑙 𝑦𝑖𝑒𝑙𝑑
𝑃𝑒𝑟𝑐𝑒𝑛𝑡𝑎𝑔𝑒 𝑦𝑖𝑒𝑙𝑑 = × 100%
𝑡ℎ𝑒𝑜𝑟𝑒𝑡𝑖𝑐𝑎𝑙 𝑦𝑖𝑒𝑙𝑑
𝑚𝑎𝑠𝑠 𝑜𝑓 𝑝𝑢𝑟𝑒 𝑠𝑢𝑏𝑠𝑡𝑎𝑛𝑐𝑒 𝑖𝑛 𝑠𝑎𝑚𝑝𝑙𝑒
𝑃𝑒𝑟𝑐𝑒𝑛𝑡𝑎𝑔𝑒 𝑝𝑢𝑟𝑖𝑡𝑦 = × 100%
𝑚𝑎𝑠𝑠 𝑜𝑓 𝑠𝑎𝑚𝑝𝑙𝑒
Acids and Base Reactions
Acids react with metals to form salt and hydrogen gas (except lead, insoluble salt)
Initial reaction between lead and hydrochloric acid produce lead (II) chloride or lead (II) sulfate
which is insoluble. It will form a coating around metal and protect metal from further attack by acid.
React with carbonates and hydrogen carbonates to form salt, water and carbon dioxide
React with metal oxides and hydroxides to form salt and water
Alkalis heated with ammonium salts give out salt + water +ammonia
Solubility
Soluble salts Insoluble salts
All sodium salts
All potassium salts
All ammonium salts
All nitrates
All chlorides Silver chloride
Lead (II) chloride
All sulfates Barium sulfate
Lead (II) sulfate
Calcium sulfate (sparingly soluble)
Sodium carbonate
Potassium carbonate
Ammonium carbonate
Hydroxide salts of Group I elements are soluble. Hydroxide salts of Group II elements (Ca, Sr, and Ba) are
slightly soluble. Hydroxide salts of transition metals and Al3+ are insoluble.
All oxides are insoluble except calcium (sparingly), barium and Group 1 metals but SO3 is soluble.
Compiled by Ethan Wu Ying Tang
Qualitative Analysis
Test for Cations
Cation Sodium Hydroxide Aqueous Ammonia
On adding a few On adding excess On adding a few On adding excess
drops drops
Zinc ion Zn2+ White ppt Ppt dissolves in White ppt Ppt dissolves in
excess to form excess to form
colourless colourless
solution solution
Aluminium ion AL3+ White ppt Ppt dissolves in White ppt Insoluble in
excess to form excess
colourless
solution
Lead(II) ion PB2+ White ppt Ppt dissolves in White ppt Insoluble in
excess to form excess
colourless
solution
Calcium ion Ca2+ White ppt Insoluble in No ppt No ppt
excess
Copper (II) ion Cu2+ Light blue ppt Insoluble in Light blue ppt Ppt dissolves in
excess excess to form
deep blue
solution
Iron (II) ion Fe2+ Green ppt Insoluble in Green ppt Insoluble in
excess excess
Iron (III) ion Fe3+ Reddish-brown Insoluble in Reddish-brown Insoluble in
ppt excess ppt excess
Ammonium ion No ppt No change - -
+
NH4 On heating,
ammonia gas
give off
The ppt formed in each reaction is the hydroxide of the metal ion.
Test for Anions
Anion Test Observations for positive test
and inference
Carbonate ion CO32- Add dilute hydrochloric acid Effervescence observed.
Pass the gas given off into limewater Gas forms a white ppt with
limewater. Carbon dioxide gas
given off
Nitrate ion NO3- Add sodium hydroxide solution, then add a Effervescence observed
piece of aluminium foil. Warm the mixture. The moist red litmus paper
Test the gas given off with a piece of moist turns blue. Ammonia gas given
red litmus paper. off
Sulfate ion SO42- Add dilute nitric acid, then add barium A white ppt of barium sulfate
nitrate solution formed
Compiled by Ethan Wu Ying Tang
Chloride ion Cl- Add dilute nitric acid, then add silver A white ppt of silver chloride
nitrate solution formed
Iodide ion I- Add dilute nitric acid, then add silver A yellow ppt of silver iodide
nitrate solution formed
Identifying Gases
Gas Colour and Odour Test Observations
Hydrogen H2 Colourless and odourless Place a lighted splint at The light splint is
the mouth of the test extinguished with a
tube ‘pop’ sound
Oxygen O2 Colourless and odourless Insert a glowing splint The glowing splint is
into the test tube rekindled
Carbon Dioxide CO2 Colourless and odourless Bubble gas through A white precipitate is
limewater formed
The ppt dissolves upon
further bubbling
Chlorine Cl2 Greenish-yellow gas with a Place a piece of moist The moist blue litmus
pungent smell blue litmus paper at paper turns red and is
the mouth of the test then bleached
tube
Sulfur Dioxide SO2 Colourless gas with a Place a piece of filter The purple acidified
pungent smell paper soaked with potassium manganate
acidified potassium (VII) turns colourless
manganate (VII) at the
mouth of the test tube
Ammonia NH3 Colourless gas with a Place a piece of moist The moist red litmus
pungent smell red litmus paper at the paper turns blue
mouth of the test tube
Redox Reaction
Oxidising agents Reducing agents
Bromine Carbon
Chlorine Carbon Monoxide
Concentrated sulfuric acid Hydrogen
Nitric acid Hydrogen Sulfide
Oxygen Metals
Ozone Potassium Iodide
Potassium Manganate (VII) (KMnO4) Sulfur Dioxide
Potassium Dichromate (VI) (Kr2Cr2O7)
Reactivity series
Metal Reaction with cold water Reaction with steam
Potassium 2K(s) + 2H2O(l) -> 2KOH(aq) +H2(g) React explosively
Sodium 2Na(s) + 2H2O(l) -> 2NaOH(aq) +H2(g)
Calcium Ca(s) + 2H2O(l) -> Ca(OH)2(aq) + H2(g)
Magnesium Mg(s) + 2H2O(l) -> Mg(OH)2(s) +H2(g) Mg(s) + H2O(g) -> MgO(s) + H2(g)
Compiled by Ethan Wu Ying Tang
Zinc No reaction Zn(s) + H2O(s) + H2O(g) -> ZnO(s) +H2(g)
Zinc oxide is yellow when hot and white when
cold
Iron No reaction (except rusting) 3Fe(s) + 4H2O(g) -> Fe3O4(g)
Reacts slowly, iron must be heated constantly
Lead No reaction No reaction
Copper
Silver
Most reactive to least reactive
K Na Ca Mg Zn Fe Pb H Cu Ag
A more reactive metal can displace a less reactive metal from its salt solution (redox reaction)
A more reactive metal can reduce the oxide of a less reactive metal
Action of heat on metal carbonates
Metal carbonate Observation
Potassium carbonate Unaffected by heat
Sodium carbonate
Calcium carbonate Decompose into metal oxide and carbon dioxide
Magnesium carbonate on heating
Zinc carbonate
Iron (II) carbonate
Lead(II) carbonate
Copper(II) carbonate
Silver carbonate Decomposes into silver and carbon dioxide on
heating
Extracting iron from haematite
1 Carbon dioxide is produced
C(s) + O2(g) -> CO2(g)
2 Carbon monoxide is produced
C(s) + CO2(g) -> 2CO(g)
3 Haematite is reduced to iron
Fe2O3(s) + 3CO(g) -> 2Fe(l) + 3CO2(g)
4 Impurities are removed
CaCO3(s) -> CaO(s) + CO2(g)
CaO(s) + SiO2(s) -> CaSiO3(l)
Compiled by Ethan Wu Ying Tang
Hardest to discharge to easiest to discharge
K+ Na+ Ca2+ MG2+ Zn2+ Fe2+ Pb2+ H+ Cu2+ Ag+
Hardest to discharge to easiest to discharge
SO42- NO3- Cl- Br- I- OH-
Manufacturing ammonia by Haber process
Nitrogen + hydrogen ⇌ ammonia (added in the ratio 1:3)
N2(g) + 3H2(g) -⇌ 2NH3(g)
Conditions: 250atm, 450°C, iron as catalyst
Only about 10-15% of nitrogen and hydrogen concerted to ammonia (ammonia gas cooled to liquid)
SO2 + H2O -> H2SO3 sulfurous acid
Sulfurous acid can be oxidized in air to form sulfuric acid
4NO2 + 2H2O + O2 -> 4HNO3
Reducing pollution
Catalytic converters
2CO(g) + O2(g) -> 2CO2(g)
2NO(g) + 2CO(g) -> N2(g) + 2CO2(g)
2C8H18(l) + 25O2(g) -> 16CO2(g) + 18H2O(g)
Flue Gas Desulfurisation (remove sulfur dioxide pollution)
CaCO3(s) + SO2(g) -> CaSO3(S) + CO2
2CaSO3(s) + O2(g) -> 2CaSO4(s)
CaO(s) + SO2(g) -> CaSO3(s)
Alkanes
Combustion
Alkane + oxygen -> carbon dioxide + water vapour
Substitution (under ultraviolet light)
E.g. methane + chlorine -> chloromethane + hydrogen chloride
Alkenes
Combustion
Alkene + oxygen -> carbon dioxide + water vapour
As alkenes have higher percentage of carbon than alkanes, alkenes will burn with a sootier flame
Alkenes can also undergo substitution reaction under UV light
An Addition reaction is a reaction in which an unsaturated organic compound combines with another
substance to form a single new compound
Addition of hydrogen
Ethene + hydrogen -> ethane
Conditions: 200°C, nickel as catalyst
Convert vegetable oils to solid margarine
Addition of bromine
Compiled by Ethan Wu Ying Tang
Ethene + bromine -> 1,2-dibromooethane
Serve as a chemical test to distinguish between alkane and alkene
Addition of steam
Ethene + steam -> ethanol
Conditions: 300°C, 60 atm, phosphoric (V) acid as catalyst
Addition polymerization
Ethene -> poly(ethene)
Conditions: High temperature and pressure and catalyst
Alcohol
Combustion
Alcohol + oxygen -> carbon dioxide + water vapour
Can be used as a fuel
Burnt on some food such as fruit cake to give it a distinct flavor
Oxidation
Alcohol + oxidizing agent -> carboxylic acid + water
C2H5OH + 2[O] -> CH3COOH +H2O
(2 ways oxidation by acidified potassium manganate (VII) and oxidation by atmospheric oxygen)
Breathalyzer contains an oxidizing agent that has a colour agent when high level of alcohol detected
Producing ethanol
From catalytic addition of steam to ethene
Alcohol fermentation Glucose -> ethanol + carbon dioxide
Carboxylic acids
Are weak acids
Reaction with reactive metals (salts of ethanoic acid are known as ethanoates)
Reaction with carbonates
Reaction with bases
Reaction with alcohols to produce ester and water (esterification)
Ester
An ester is a colourless liquid that is insoluble in water
Ethanoic acid + methanol -> methyl ethanoate (concentrated sulfuric acid as catalyst)
First part of name from alcohol
Functional group
Polymerization
Additional polymerization occurs when unsaturated monomers join together without losing any
molecules or atoms.
Condensation polymerization occurs when monomers combine to form a polymer with the removal of a
small molecule such as water.
Compiled by Ethan Wu Ying Tang
You might also like
- Topic 10 - TradeDocument46 pagesTopic 10 - TradeJaveria AfzaalNo ratings yet
- PDF Cambridge Igcse Chemistry 4th Edition CompressDocument290 pagesPDF Cambridge Igcse Chemistry 4th Edition CompressNilkanth DesaiNo ratings yet
- Harrys Cosmetology Chapter 11 15Document122 pagesHarrys Cosmetology Chapter 11 15Nidhi100% (3)
- Csec Chemistry 2013-18 Long Paper (Solutions)Document75 pagesCsec Chemistry 2013-18 Long Paper (Solutions)Nathan Tate100% (1)
- 7F Quick QuizDocument2 pages7F Quick QuizcenatafioNo ratings yet
- Grammar Tree 7 Teaching GuideDocument52 pagesGrammar Tree 7 Teaching GuideSyed Ashar100% (2)
- O Level Pure Physics Topic by Topic 3 109 112Document4 pagesO Level Pure Physics Topic by Topic 3 109 112Kugan KishurNo ratings yet
- Scheme of Work: Cambridge IGCSE / Cambridge IGCSE (9-1) Computer Science 0478 / 0984Document39 pagesScheme of Work: Cambridge IGCSE / Cambridge IGCSE (9-1) Computer Science 0478 / 0984Syed Ashar80% (10)
- Notes of GeoGraphy Olevel's Chaper 8.Document9 pagesNotes of GeoGraphy Olevel's Chaper 8.ubaidcom786100% (1)
- Once A Week Comprehension Book 1 Answers: Test 1Document30 pagesOnce A Week Comprehension Book 1 Answers: Test 1Syed Ashar75% (4)
- Once A Week Comprehension Book 4 AnswersDocument30 pagesOnce A Week Comprehension Book 4 AnswersSyed Ashar80% (5)
- Brief History of Ban I IsraelDocument39 pagesBrief History of Ban I IsraelSyed AsharNo ratings yet
- History and Culture of Pakistan Chapter-2: What Were The Causes and Consequences of The Decline of Mughal Empire?Document7 pagesHistory and Culture of Pakistan Chapter-2: What Were The Causes and Consequences of The Decline of Mughal Empire?saad saadNo ratings yet
- 4024 Mathematics Example Candidate Responses Booklet 2014 PDFDocument181 pages4024 Mathematics Example Candidate Responses Booklet 2014 PDFEMAAN SARWARNo ratings yet
- Deformation NotesDocument6 pagesDeformation NotesAtif Naseem100% (1)
- 5054 Space Physics Practice Questions For Examination From 2023 1Document25 pages5054 Space Physics Practice Questions For Examination From 2023 1Muhammad QadirNo ratings yet
- Once A Week Comprehension Book 2 Answers Test 1Document30 pagesOnce A Week Comprehension Book 2 Answers Test 1Syed Ashar100% (6)
- O Levels Notes Chapter 1 - Physical Quantities and Measurements - UPDATEDDocument3 pagesO Levels Notes Chapter 1 - Physical Quantities and Measurements - UPDATEDAfroz Ahmad100% (1)
- Learner Guide: Cambridge IGCSE Computer Science 0478 / 0984Document27 pagesLearner Guide: Cambridge IGCSE Computer Science 0478 / 0984Syed Ashar100% (2)
- Geo TopicalDocument215 pagesGeo TopicalAsiya Mahboob0% (1)
- Product Range Textile Auxiliaries - Coating. VersionDocument132 pagesProduct Range Textile Auxiliaries - Coating. VersionRokok Indonesia100% (1)
- 1988 1998 Physics Past PapersDocument168 pages1988 1998 Physics Past PapersAnthony Benson100% (1)
- O Level NotesDocument7 pagesO Level NotesDhanBahadur100% (1)
- High Temperature Corrosion in Molten SaltsDocument250 pagesHigh Temperature Corrosion in Molten SaltsAlex LakeNo ratings yet
- O Level Notes On Mass, Weight and DensityDocument8 pagesO Level Notes On Mass, Weight and DensityRavi MothoorNo ratings yet
- Chemistry Notes On The Periodic Table: Syllabus 5070 O' LevelDocument8 pagesChemistry Notes On The Periodic Table: Syllabus 5070 O' LevelNancy Mohamed0% (1)
- O'level Physics TemperatureDocument8 pagesO'level Physics TemperatureTorettoNo ratings yet
- Cambridge O Level: Second Language Urdu For Examination From 2024Document10 pagesCambridge O Level: Second Language Urdu For Examination From 2024iman 908No ratings yet
- Olevels Islamiyat - Quranic PassagesDocument13 pagesOlevels Islamiyat - Quranic Passagessir umar khanNo ratings yet
- O Level Nuclear Physics and RadioactivityDocument16 pagesO Level Nuclear Physics and RadioactivityMd SafwatNo ratings yet
- Topography of Pakistan: by Sir Ashar Class 9thDocument56 pagesTopography of Pakistan: by Sir Ashar Class 9thSyed AsharNo ratings yet
- O Level Islamiyat P1 TopicalDocument5 pagesO Level Islamiyat P1 TopicalWaleed Zahid100% (1)
- 0610 BIOLOGY: MARK SCHEME For The October/November 2010 Question Paper For The Guidance of TeachersDocument12 pages0610 BIOLOGY: MARK SCHEME For The October/November 2010 Question Paper For The Guidance of TeachersPreethi Sritharan100% (1)
- Experimental Techniques Questions WorksheetDocument12 pagesExperimental Techniques Questions WorksheetRosina KaneNo ratings yet
- 5090 Biology Paper 6 AtpDocument8 pages5090 Biology Paper 6 Atpbiochemistpak50% (12)
- June 2000 - Paper 2Document12 pagesJune 2000 - Paper 2theyaasir67% (3)
- Biology 5090 OL P1 MCQ 2015-2019 All VsDocument317 pagesBiology 5090 OL P1 MCQ 2015-2019 All VsHanaa Ibrahim AliNo ratings yet
- Islamiat O-LevelDocument3 pagesIslamiat O-LevelAbdullah AamirNo ratings yet
- Islamiat References (2058)Document44 pagesIslamiat References (2058)Maheesha AmjaddNo ratings yet
- Mathematics Class 9 Syllabus Break Up AY 2022-23Document5 pagesMathematics Class 9 Syllabus Break Up AY 2022-23عبدل رافعNo ratings yet
- O Level Space Physics NotesDocument40 pagesO Level Space Physics Noteslolshedo100% (2)
- Cambridge O/L PHYSICS (5054) : Motion, Forces and EnergyDocument11 pagesCambridge O/L PHYSICS (5054) : Motion, Forces and EnergySenula Rathgamage100% (2)
- Power Resources Exercise 2059/02Document4 pagesPower Resources Exercise 2059/02mishi moshNo ratings yet
- Topic-18: Static Electricity: Electrostatic PhenomenaDocument9 pagesTopic-18: Static Electricity: Electrostatic Phenomenamuhammad awaisNo ratings yet
- 5070 Chemistry ECR v1.1 080316Document153 pages5070 Chemistry ECR v1.1 080316warisNo ratings yet
- Past Paper Questions May/June 2009 To Oct/Nov 2019: Paper 2 Islamiyat GCE O LEVEL & IGCSEDocument21 pagesPast Paper Questions May/June 2009 To Oct/Nov 2019: Paper 2 Islamiyat GCE O LEVEL & IGCSEJaweria Khan100% (1)
- Unit 5 - Mineral Resources What Are Minerals?: Pakistan StudiesDocument13 pagesUnit 5 - Mineral Resources What Are Minerals?: Pakistan StudiesTaha YousafNo ratings yet
- Gce O' Level Islamiyat (2058) : Topical Question Bank J2009-J2011Document10 pagesGce O' Level Islamiyat (2058) : Topical Question Bank J2009-J2011Ali DonNo ratings yet
- The Periodic Table: Test Yourself 16.1 and 16.2 (Page 312)Document2 pagesThe Periodic Table: Test Yourself 16.1 and 16.2 (Page 312)khalil rehmanNo ratings yet
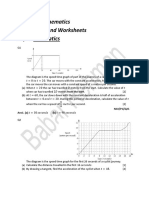
- O' Level Mathematics Past Papers and Worksheets Topic: KinematicsDocument8 pagesO' Level Mathematics Past Papers and Worksheets Topic: Kinematicsadeelthegr8100% (2)
- Travelling by Train in India SummaryDocument1 pageTravelling by Train in India SummaryDhriti GuptaNo ratings yet
- Kinematics Olevel Practice QuestionsDocument2 pagesKinematics Olevel Practice Questionstaha zaidi100% (3)
- Worksheet O Levels Physics PressureDocument30 pagesWorksheet O Levels Physics PressureHZNo ratings yet
- 1 - States of MatterDocument9 pages1 - States of Mattermostafa barakatNo ratings yet
- Revision Checklist For O Level Islamiyat 2058 FINALDocument13 pagesRevision Checklist For O Level Islamiyat 2058 FINALLmfao LmfaoNo ratings yet
- Re-Read Paragraphs 4, 6 and 7 Identify and Write Down One Opinion From Each of These ParagraphsDocument7 pagesRe-Read Paragraphs 4, 6 and 7 Identify and Write Down One Opinion From Each of These ParagraphsMikolas Hovan Susanto100% (2)
- Pak Studies O Levels CAIE Past Paper NotesDocument58 pagesPak Studies O Levels CAIE Past Paper Notesbilal ayub100% (1)
- Quranic Passages Umar KhanDocument17 pagesQuranic Passages Umar KhanHala AlzuhairiNo ratings yet
- 8.1-Transport - in - Plants - Igcse-Cie-Biology - Solved OLIDocument13 pages8.1-Transport - in - Plants - Igcse-Cie-Biology - Solved OLIAmber TeacherNo ratings yet
- 9701 w02 QP 4Document12 pages9701 w02 QP 4Hubbak KhanNo ratings yet
- Land of Pakistan-NotesDocument8 pagesLand of Pakistan-NotesTanvir Ahmad SaleemiNo ratings yet
- Islamiat PapersDocument76 pagesIslamiat PapersMuhammad Nizam-Ud-DinNo ratings yet
- 5090 s18 Ms 12Document3 pages5090 s18 Ms 12AliNo ratings yet
- Science Worksheet For Intervention Classes Grade 8 2nd TermDocument10 pagesScience Worksheet For Intervention Classes Grade 8 2nd TermAmna50% (2)
- O Level Physics P1 June 77 - London University Examination BoardDocument10 pagesO Level Physics P1 June 77 - London University Examination Boarddjenner100% (1)
- Level Past Paper Questions - Physics O: TOPIC-22 Electronics PAPER-1 Multiple ChoiceDocument10 pagesLevel Past Paper Questions - Physics O: TOPIC-22 Electronics PAPER-1 Multiple Choiceelty TanNo ratings yet
- Unit 6.mineralsDocument7 pagesUnit 6.mineralsBurhanNo ratings yet
- Electrode Potential QuestionsDocument2 pagesElectrode Potential QuestionsTeejay MakazhuNo ratings yet
- Islamiyat 2058 Grade Threshold TableDocument1 pageIslamiyat 2058 Grade Threshold TableAbdulAziz ShaikhNo ratings yet
- Chem 5070 RevisionDocument11 pagesChem 5070 Revisionֆɦɛʀաɨռ ֆǟʀʄʀǟʐNo ratings yet
- Revision & High-Level Exercises in Chemistry, Egyptian Curriculum, 1 Sec, For Chemistry Teacher Andrew Medhat 2023-2024Document70 pagesRevision & High-Level Exercises in Chemistry, Egyptian Curriculum, 1 Sec, For Chemistry Teacher Andrew Medhat 2023-2024yassin.study2008No ratings yet
- Chemistry Revision NotesDocument7 pagesChemistry Revision NotesFarhan RahmanNo ratings yet
- TE LP Feb 2015 KBDocument2 pagesTE LP Feb 2015 KBSyed AsharNo ratings yet
- Account CreationDocument7 pagesAccount CreationSyed AsharNo ratings yet
- Home Management Tips 1Document100 pagesHome Management Tips 1Syed AsharNo ratings yet
- Kala Pathar IntoxicationDocument5 pagesKala Pathar IntoxicationSyed AsharNo ratings yet
- Comprehensionqtypes 110425043814 Phpapp02Document14 pagesComprehensionqtypes 110425043814 Phpapp02Syed AsharNo ratings yet
- Urdu - An Essential GrammarDocument318 pagesUrdu - An Essential GrammarSyed AsharNo ratings yet
- Notes On Map ReadingDocument9 pagesNotes On Map ReadingSyed AsharNo ratings yet
- English VII - Differentiated Plan - Valley of Fear Review Playscript - Ayman KamranDocument4 pagesEnglish VII - Differentiated Plan - Valley of Fear Review Playscript - Ayman KamranSyed AsharNo ratings yet
- Topography of Pakistan: by Sir Ashar Class 9thDocument56 pagesTopography of Pakistan: by Sir Ashar Class 9thSyed AsharNo ratings yet
- Correct The Following Scentences Final.Document3 pagesCorrect The Following Scentences Final.Syed AsharNo ratings yet
- ORC7 Teachers GuideDocument28 pagesORC7 Teachers GuideSyed AsharNo ratings yet
- Experiment Salt Analysis Ammonium Chloride 2223Document2 pagesExperiment Salt Analysis Ammonium Chloride 2223ARYAN GOELNo ratings yet
- Caie Igcse Chemistry 0620 Alternative To Practical v2Document11 pagesCaie Igcse Chemistry 0620 Alternative To Practical v2Sanjith KatakamNo ratings yet
- X - CH - 4 - Carbon and Its CompoundDocument16 pagesX - CH - 4 - Carbon and Its CompoundMathanNo ratings yet
- Tests of Retarding Admixtures For ConcreteDocument32 pagesTests of Retarding Admixtures For ConcreteRaynus ArhinNo ratings yet
- BS 1744-1-1998 PDFDocument30 pagesBS 1744-1-1998 PDFMohammed KarasnehNo ratings yet
- A Review of Secondary Aluminum Production and Its ByproductsDocument12 pagesA Review of Secondary Aluminum Production and Its ByproductsmichaelNo ratings yet
- BIOENERGETICS Notes 3Document3 pagesBIOENERGETICS Notes 3Hya LaniogNo ratings yet
- Chemistry: Solubility Rules WorksheetDocument4 pagesChemistry: Solubility Rules WorksheetAlvin AiauNo ratings yet
- Csec Chemistry - A5Document17 pagesCsec Chemistry - A5Mencarty DocentNo ratings yet
- Sample Questions - Chapter 4Document5 pagesSample Questions - Chapter 4Uday Prakash SahuNo ratings yet
- Cleansing Action of Soap and Detergent (Investigatory Project - Class 12)Document22 pagesCleansing Action of Soap and Detergent (Investigatory Project - Class 12)Yati JainNo ratings yet
- 2324 Level M (Gr11 UAE-Gulf) Chemistry Chapter 2 NotesDocument19 pages2324 Level M (Gr11 UAE-Gulf) Chemistry Chapter 2 Notesaminata13536No ratings yet
- Iminium Salt in Organic Synthesis (New) NumberedDocument85 pagesIminium Salt in Organic Synthesis (New) Numberedbosejohn100% (1)
- SA Calcium StearateDocument35 pagesSA Calcium StearateAPJ HollywoodsistersNo ratings yet
- Science 31/2/3 Set-3 Class 10th Question Paper 2023-24Document12 pagesScience 31/2/3 Set-3 Class 10th Question Paper 2023-24Apoorv SahuNo ratings yet
- Aqueous Reactions & Solution StoichiometryDocument125 pagesAqueous Reactions & Solution StoichiometrykumuthaNo ratings yet
- Inorganic Nomenclature Worksheet 2Document7 pagesInorganic Nomenclature Worksheet 2Ji-Shawn PardassieNo ratings yet
- My Separating Mixture Summary Poster 1Document1 pageMy Separating Mixture Summary Poster 1MinduliNo ratings yet
- Inorganic Qualitative AnalysisDocument8 pagesInorganic Qualitative AnalysisMohamed NasihNo ratings yet
- 4.1 Acids, Bases and SaltsDocument22 pages4.1 Acids, Bases and SaltsFestus NanokNo ratings yet
- Harry B. Gray and C. J. Ballhusa - Molecular Orbital Theory For Square Planar Metal ComplexesDocument6 pagesHarry B. Gray and C. J. Ballhusa - Molecular Orbital Theory For Square Planar Metal ComplexesNuansak3No ratings yet
- Chem AnswersDocument8 pagesChem AnswersEunielyn SecretoNo ratings yet
- Rubber Chemical Resistance ChartDocument13 pagesRubber Chemical Resistance ChartQuality ControlNo ratings yet