Professional Documents
Culture Documents
2 Carnot Cycle
Uploaded by
Bhavin GandhiCopyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
2 Carnot Cycle
Uploaded by
Bhavin GandhiCopyright:
Available Formats
Gas Power Cycles
Prof. U.S.P. Shet , Prof. T. Sundararajan and Prof. J.M . Mallikarjuna
4.1 Carnot Cycle :
A Carnot gas cycle operating in a given temperature range is shown in the T-s diagram in Fig. 4.1(a). One way to carry out the processes of this cycle is through the use of steady-state, steady-flow devices as shown in Fig. 4.1(b). The isentropic expansion process 2-3 and the isentropic compression process 4-1 can be simulated quite well by a well-designed turbine and compressor respectively, but the isothermal expansion process 1-2 and the isothermal compression process 3-4 are most difficult to achieve. Because of these difficulties, a steady-flow Carnot gas cycle is not practical.
The Carnot gas cycle could also be achieved in a cylinder-piston apparatus (a reciprocating engine) as shown in Fig. 4.2(b). The Carnot cycle on the p-v diagram is as shown in Fig. 4.2(a), in which processes 1-2 and 3-4 are isothermal while processes 2-3 and 4-1 are isentropic. We know that the Carnot cycle efficiency is given by the expression.
th = 1 -
T TL T =1- 4 =1- 3 TH T1 T2
Indian Institute of Technology Madras
Gas Power Cycles
Prof. U.S.P. Shet , Prof. T. Sundararajan and Prof. J.M . Mallikarjuna
TH TL
4 (a)
isothermal Turbine
1
heat in
Work out 2
Isentropic Turbine Work out
Work Work in in Isothermal heat out Compressor
Isentropic compressor
Fig.4.1. Steady flow Carnot engine
Indian Institute of Technology Madras
Gas Power Cycles
Prof. U.S.P. Shet , Prof. T. Sundararajan and Prof. J.M . Mallikarjuna
Process 1 Process 2 Process 3 Process 4
2: isothermal 3: isentropic 4: isothermal 1: isentropic
2 4 3 (a)
Piston displacement (b)
Fig.4.2. Reciprocating Carnot engine
Indian Institute of Technology Madras
Gas Power Cycles
Prof. U.S.P. Shet , Prof. T. Sundararajan and Prof. J.M . Mallikarjuna
3 4
2 1
Volume
T3=T4
T2=T1
Entropy
Fig.4.3. Carnot cycle on p-v and T-s diagrams
Indian Institute of Technology Madras
Gas Power Cycles
Prof. U.S.P. Shet , Prof. T. Sundararajan and Prof. J.M . Mallikarjuna
High temperature Source T3 K
Perfectly insulated walls
Piston
T1 K Low temperature Perfect insulator cum Sink Perfet conductor
Fig.4.4. Working of Carnot engine
Since the working fluid is an ideal gas with constant specific heats, we have, for the isentropic process,
T1 V4 = T4 V1
1
V T ; 2 = 3 T3 V3
Now, T1 = T2 and T4 = T3, therefore
v v4 = 3 = r = compression or expansion ratio v1 v2
Carnot cycle efficiency may be written as, th = 1 1 r - 1
From the above equation, it can be observed that the Carnot cycle efficiency increases as r increases. This implies that the high thermal efficiency of a Carnot cycle is
Indian Institute of Technology Madras
Gas Power Cycles
Prof. U.S.P. Shet , Prof. T. Sundararajan and Prof. J.M . Mallikarjuna
obtained at the expense of large piston displacement. Also, for isentropic processes we have,
p T1 = 1 T4 p4
1
p T and 2 = 2 T3 p3
Since, T1 = T2 and T4 = T3, we have
p1 p = 2 = rp = pressure ratio p4 p3
Therefore, Carnot cycle efficiency may be written as,
th = 1 rp 1
1
From the above equation, it can be observed that, the Carnot cycle efficiency can be increased by increasing the pressure ratio. This means that Carnot cycle should be operated at high peak pressure to obtain large efficiency.
Indian Institute of Technology Madras
You might also like
- Chemical Energy and Exergy: An Introduction to Chemical Thermodynamics for EngineersFrom EverandChemical Energy and Exergy: An Introduction to Chemical Thermodynamics for EngineersNo ratings yet
- Carnot CycleDocument6 pagesCarnot CycleyuvrajsinghvloggingNo ratings yet
- Phase Equilibrium in Mixtures: International Series of Monographs in Chemical EngineeringFrom EverandPhase Equilibrium in Mixtures: International Series of Monographs in Chemical EngineeringNo ratings yet
- ThermoDocument105 pagesThermoAcfMacNo ratings yet
- 8 Comparison of Otto, Diesel, Dual CyclesDocument7 pages8 Comparison of Otto, Diesel, Dual Cyclescaptainhass0% (1)
- Problems in Metallurgical Thermodynamics and Kinetics: International Series on Materials Science and TechnologyFrom EverandProblems in Metallurgical Thermodynamics and Kinetics: International Series on Materials Science and TechnologyRating: 4 out of 5 stars4/5 (5)
- 4 Ericsson CycleDocument2 pages4 Ericsson CyclecaptainhassNo ratings yet
- Gas Power Cycles IITDocument68 pagesGas Power Cycles IITAfsar HusainNo ratings yet
- CVPCDocument2 pagesCVPCapi-3830954No ratings yet
- Recommended Reference Materials for Realization of Physicochemical Properties: Pressure–Volume–Temperature RelationshipsFrom EverandRecommended Reference Materials for Realization of Physicochemical Properties: Pressure–Volume–Temperature RelationshipsE. F. G. HeringtonNo ratings yet
- 2 Carnot CycleDocument6 pages2 Carnot CycleSamujawal Kumar SumanNo ratings yet
- 4 Capacity of Steam Power PlantDocument4 pages4 Capacity of Steam Power PlantcaptainhassNo ratings yet
- COFSPPDocument4 pagesCOFSPPapi-3830954No ratings yet
- Transient Heat Conduction: Course ContentsDocument13 pagesTransient Heat Conduction: Course ContentsJainil GajjarNo ratings yet
- 6 Deviation of Actual Cycle From Ideal CycleDocument5 pages6 Deviation of Actual Cycle From Ideal CyclecaptainhassNo ratings yet
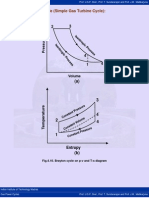
- 4.10 Brayton Cycle (Simple Gas Turbine Cycle) :: Fig.4.10. Brayton Cycle On P-V and T-S DiagramDocument3 pages4.10 Brayton Cycle (Simple Gas Turbine Cycle) :: Fig.4.10. Brayton Cycle On P-V and T-S DiagrammominjeelaniNo ratings yet
- 4.0 Nphys3518Document6 pages4.0 Nphys3518arun rajaramNo ratings yet
- 9 Atkinson CycleDocument3 pages9 Atkinson CyclecaptainhassNo ratings yet
- VTU E-Shikshana Programme 17ME43: Applied Thermodynamics E-NotesDocument37 pagesVTU E-Shikshana Programme 17ME43: Applied Thermodynamics E-NotesJakkula LavanyaNo ratings yet
- Basic Thermodynamic CyclesDocument4 pagesBasic Thermodynamic Cyclesapi-3830954No ratings yet
- Rankine CycleDocument4 pagesRankine CycleyuvrajsinghvloggingNo ratings yet
- ME495 BraytonCycleGasTurbine PDFDocument11 pagesME495 BraytonCycleGasTurbine PDFJordan SolisNo ratings yet
- 1 Prof - Sajeurkar /te (Mech) /nkocet SolapurDocument55 pages1 Prof - Sajeurkar /te (Mech) /nkocet Solapurravi00098No ratings yet
- 7 Limited Pressure CycleDocument4 pages7 Limited Pressure CyclecaptainhassNo ratings yet
- Experimental and Theoretical IR, R, and INS Spectra of 2,2,4,4-Tetramethyl-3-T-Butyl-Pentane-3-OlDocument9 pagesExperimental and Theoretical IR, R, and INS Spectra of 2,2,4,4-Tetramethyl-3-T-Butyl-Pentane-3-OlLupu ValentinNo ratings yet
- Tautomerism of 1,2,3 - and 1,2,4-Triazole in The Gas Phase and in Aqueous Solution - JPC 1990Document3 pagesTautomerism of 1,2,3 - and 1,2,4-Triazole in The Gas Phase and in Aqueous Solution - JPC 1990Fausto SalazarNo ratings yet
- Thermal C - 7 Part II (Student)Document24 pagesThermal C - 7 Part II (Student)pal148587No ratings yet
- Air Cycle RefrigerationDocument10 pagesAir Cycle RefrigerationashisNo ratings yet
- 2013 - AAPS - Electrostatic Charging Behavior of Gelatin and HPMC Hard CapsulesDocument1 page2013 - AAPS - Electrostatic Charging Behavior of Gelatin and HPMC Hard Capsulessubhash thakurNo ratings yet
- Adsfd PDFDocument1 pageAdsfd PDFsubhash thakurNo ratings yet
- Bell Coleman CycleDocument5 pagesBell Coleman CycleAshara Aakash100% (2)
- Air Standard Cycle Report 1Document17 pagesAir Standard Cycle Report 1prasad adsuleNo ratings yet
- Lecture 2 (Incomplete)Document27 pagesLecture 2 (Incomplete)CaptainPatsNo ratings yet
- Wed10 Exp E6Document11 pagesWed10 Exp E6shorya tamrakarNo ratings yet
- Lecture Notes OnDocument200 pagesLecture Notes Onananth k r100% (3)
- 400 623Document31 pages400 623SHIVAM JAT100% (1)
- Stefan BoltzmanDocument6 pagesStefan BoltzmanKylo RenNo ratings yet
- Smple Cycle Thermodynamics OverviewDocument31 pagesSmple Cycle Thermodynamics OverviewONURNo ratings yet
- Me F311 1267 PDFDocument3 pagesMe F311 1267 PDFBhaskar SharmaNo ratings yet
- Stirling and Ericsson Cycles 2016 PDFDocument13 pagesStirling and Ericsson Cycles 2016 PDFArvin Loui Bascon100% (1)
- NPG Phase TransformationDocument31 pagesNPG Phase Transformationraj_kr1078No ratings yet
- Thermal Physics - DPP 11 of (Lec 23) - Saakaar Batch For IIT JAM PhysicsDocument3 pagesThermal Physics - DPP 11 of (Lec 23) - Saakaar Batch For IIT JAM PhysicsAnkur TiwariNo ratings yet
- Required Reading:: Gas Power CyclesDocument32 pagesRequired Reading:: Gas Power CyclesZoraiz ButtNo ratings yet
- EtDocument12 pagesEtKotadia ShivamNo ratings yet
- BDA30403 Steam Power Plant - Rankine CycleDocument43 pagesBDA30403 Steam Power Plant - Rankine CycleShishajimoo100% (1)
- FlashKinetics - Spring 2014 UpdatedDocument9 pagesFlashKinetics - Spring 2014 Updatedpeterbishop89No ratings yet
- 1 Carnot VPC Iit LectureDocument2 pages1 Carnot VPC Iit LectureKapil GoyalNo ratings yet
- Decomposition of 1,3,5-Trioxane at 700-800 KDocument3 pagesDecomposition of 1,3,5-Trioxane at 700-800 KPilar MayaNo ratings yet
- Gas Turbin RevDocument11 pagesGas Turbin RevmnugroNo ratings yet
- FirstandSecond LawsAnalysisofanAir StandardDualCycleWithHeatLossConsiderationDocument19 pagesFirstandSecond LawsAnalysisofanAir StandardDualCycleWithHeatLossConsiderationpirate hereNo ratings yet
- Laboratory Assessment Form Mec 424 (Applied Mechanics Lab) : Name Group Student IdDocument24 pagesLaboratory Assessment Form Mec 424 (Applied Mechanics Lab) : Name Group Student Idshahril shaharoniNo ratings yet
- Applied Applied Applied Applied Physics Laboratory Manual Physics Laboratory Manual Physics Laboratory Manual Physics Laboratory ManualDocument26 pagesApplied Applied Applied Applied Physics Laboratory Manual Physics Laboratory Manual Physics Laboratory Manual Physics Laboratory ManualTony TheoNo ratings yet
- Swapnil Kumbhar Paper22222Document6 pagesSwapnil Kumbhar Paper22222Tech MitNo ratings yet
- SAT # 13 (Thermal Physics) - Paper (E+H) PDFDocument18 pagesSAT # 13 (Thermal Physics) - Paper (E+H) PDFTanvir NosaNo ratings yet
- Thorium Irradiation in Indian Phwrs-Applications and Limitations of Origen CodeDocument14 pagesThorium Irradiation in Indian Phwrs-Applications and Limitations of Origen CodePanchapakesan SrinivasanNo ratings yet
- Q FV V: 2.57 Nano-to-Macro Transport Processes Fall 2004Document7 pagesQ FV V: 2.57 Nano-to-Macro Transport Processes Fall 2004captainhassNo ratings yet
- 2.57 Nano-to-Macro Transport Processes Fall 2004 - Lecture 15 Guest Lecture by Prof. DresselhausDocument10 pages2.57 Nano-to-Macro Transport Processes Fall 2004 - Lecture 15 Guest Lecture by Prof. DresselhauscaptainhassNo ratings yet
- d dT J Lq L dx dx: q=J (Π - Π) q=J (Π - Π)Document6 pagesd dT J Lq L dx dx: q=J (Π - Π) q=J (Π - Π)captainhassNo ratings yet
- 2.57 Nano-to-Macro Transport Processes Fall 2004 Guest Lecture by Prof. Mildred S. DresselhausDocument10 pages2.57 Nano-to-Macro Transport Processes Fall 2004 Guest Lecture by Prof. Mildred S. DresselhauscaptainhassNo ratings yet
- Q FV V: 2.57 Nano-to-Macro Transport Processes Fall 2004Document5 pagesQ FV V: 2.57 Nano-to-Macro Transport Processes Fall 2004captainhassNo ratings yet
- 2.57 Nano-to-Macro Transport Processes Fall 2004Document9 pages2.57 Nano-to-Macro Transport Processes Fall 2004captainhassNo ratings yet
- 2.57 Nano-to-Macro Transport Processes Fall 2004: I T KXDocument8 pages2.57 Nano-to-Macro Transport Processes Fall 2004: I T KXcaptainhassNo ratings yet
- 2.57 Nano-to-Macro Transport Processes Fall 2004: F M F F FDocument5 pages2.57 Nano-to-Macro Transport Processes Fall 2004: F M F F FcaptainhassNo ratings yet
- 2.57 Nano-to-Macro Transport Processes Fall 2004:) N N N N (NDocument8 pages2.57 Nano-to-Macro Transport Processes Fall 2004:) N N N N (NcaptainhassNo ratings yet
- Lecture 18Document6 pagesLecture 18captainhassNo ratings yet
- 2.57 Nano-to-Macro Transport Processes Fall 2004Document7 pages2.57 Nano-to-Macro Transport Processes Fall 2004captainhassNo ratings yet
- 2.57 Nano-to-Macro Transport Processes Fall 2004: A Wigner-Seitz Primitive Unit CellDocument6 pages2.57 Nano-to-Macro Transport Processes Fall 2004: A Wigner-Seitz Primitive Unit CellcaptainhassNo ratings yet
- Lecture 14Document8 pagesLecture 14captainhassNo ratings yet
- 2.57 Nano-to-Macro Transport Processes Fall 2004: Ae Be CeDocument7 pages2.57 Nano-to-Macro Transport Processes Fall 2004: Ae Be CecaptainhassNo ratings yet
- 2.57 Nano-to-Macro Transport Processes Fall 2004: Ja (j-1) A (j+1) ADocument9 pages2.57 Nano-to-Macro Transport Processes Fall 2004: Ja (j-1) A (j+1) AcaptainhassNo ratings yet
- 2.57 Nano-to-Macro Transport Processes Fall 2004: Nlms 2Document7 pages2.57 Nano-to-Macro Transport Processes Fall 2004: Nlms 2captainhassNo ratings yet
- 2.57 Nano-to-Macro Transport Processes Fall 2004: V L KK VKK N VDocument7 pages2.57 Nano-to-Macro Transport Processes Fall 2004: V L KK VKK N VcaptainhassNo ratings yet
- N F T e U F T F TD D: 2.57 Nano-to-Macro Transport Processes Fall 2004Document6 pagesN F T e U F T F TD D: 2.57 Nano-to-Macro Transport Processes Fall 2004captainhassNo ratings yet
- 2.57 Nano-to-Macro Transport Processes Fall 2004Document7 pages2.57 Nano-to-Macro Transport Processes Fall 2004captainhassNo ratings yet
- 2.57 Nano-to-Macro Transport Processes Fall 2004: E HN MDocument10 pages2.57 Nano-to-Macro Transport Processes Fall 2004: E HN McaptainhassNo ratings yet
- 2.57 Nano-To-Macro Transport Processes Fall 2004: Ikn A IkxDocument7 pages2.57 Nano-To-Macro Transport Processes Fall 2004: Ikn A IkxcaptainhassNo ratings yet
- 2.57 Nano-to-Macro Transport Processes Fall 2004Document6 pages2.57 Nano-to-Macro Transport Processes Fall 2004captainhassNo ratings yet
- Problem Solving On D C Machines PDFDocument16 pagesProblem Solving On D C Machines PDFSelvaraj ParamasivanNo ratings yet
- 2.57 Nano-to-Macro Transport Processes Fall 2004: DT Q K T Q DX Q (W/M K (W/M-K) Is Thermal ConductivityDocument5 pages2.57 Nano-to-Macro Transport Processes Fall 2004: DT Q K T Q DX Q (W/M K (W/M-K) Is Thermal ConductivitycaptainhassNo ratings yet
- Three-Phase Induction Motor: Version 2 EE IIT, KharagpurDocument8 pagesThree-Phase Induction Motor: Version 2 EE IIT, KharagpurHarsh PatelNo ratings yet
- L-44 (GDR) (Et) ( (Ee) Nptel)Document15 pagesL-44 (GDR) (Et) ( (Ee) Nptel)yashaswiyellapragadaNo ratings yet
- L-05 (GDR) (Et) ( (Ee) Nptel)Document11 pagesL-05 (GDR) (Et) ( (Ee) Nptel)nvnmnitNo ratings yet
- 2.57 Nano-to-Macro Transport Processes Fall 2004Document8 pages2.57 Nano-to-Macro Transport Processes Fall 2004captainhassNo ratings yet
- L 07 (GDR) (Et) ( (Ee) Nptel)Document15 pagesL 07 (GDR) (Et) ( (Ee) Nptel)Aneurys DuranNo ratings yet
- 20110715122354-B SC - M SC - H S - Mathematics PDFDocument75 pages20110715122354-B SC - M SC - H S - Mathematics PDFn0% (1)
- MODULE-11 Human Person As Oriented Towards Their Impending DeathDocument18 pagesMODULE-11 Human Person As Oriented Towards Their Impending DeathRoyceNo ratings yet
- 7.proceeding Snib-Eng Dept, Pnp-IndonesiaDocument11 pages7.proceeding Snib-Eng Dept, Pnp-IndonesiamissfifitNo ratings yet
- Chapter 5.0.2018 - Retaining WallDocument36 pagesChapter 5.0.2018 - Retaining WallHawaiiChongNo ratings yet
- Lab 10 InterferenceDocument5 pagesLab 10 InterferenceSarin TumtongNo ratings yet
- Lec 5 (Welded Joint)Document38 pagesLec 5 (Welded Joint)Ahmed HassanNo ratings yet
- Music and IQDocument10 pagesMusic and IQh.kaviani88No ratings yet
- Engagement : Rebecca StobaughDocument188 pagesEngagement : Rebecca StobaughAli AhmadNo ratings yet
- Goulds Pumps: WE SeriesDocument4 pagesGoulds Pumps: WE SeriesJorge ManobandaNo ratings yet
- LR Chapter 5 - Input and OutputDocument6 pagesLR Chapter 5 - Input and OutputAakash KumarNo ratings yet
- Books Vs EbooksDocument9 pagesBooks Vs EbooksThe United IndianNo ratings yet
- TI C5510 Assembly ProgrammingDocument21 pagesTI C5510 Assembly ProgrammingWinsweptNo ratings yet
- Ms. Merrick's CSEC Geography Crash Course Booklet-1Document30 pagesMs. Merrick's CSEC Geography Crash Course Booklet-1shalini100% (4)
- Experion MX MD Controls R700.1 Traditional Control: Configuration and System Build ManualDocument62 pagesExperion MX MD Controls R700.1 Traditional Control: Configuration and System Build Manualdesaivilas60No ratings yet
- WellCAD Logs OverviewDocument9 pagesWellCAD Logs OverviewpankuNo ratings yet
- 1977 The Reactive Agility TestDocument6 pages1977 The Reactive Agility TestJessica Sepúlveda SalasNo ratings yet
- ROLAN BART Zadovoljstvo U TekstuDocument56 pagesROLAN BART Zadovoljstvo U Tekstujokokokl100% (18)
- Hamara FoundationDocument7 pagesHamara Foundationshruti chavanNo ratings yet
- Fundamental Neuroscience For Basic and Clinical Applications 5Th Edition Duane E Haines Full ChapterDocument67 pagesFundamental Neuroscience For Basic and Clinical Applications 5Th Edition Duane E Haines Full Chaptermaxine.ferrell318100% (8)
- Pakistan Academy School Al-Ahmadi Kuwait Monthly Test Schedule Class: AS-LEVELDocument9 pagesPakistan Academy School Al-Ahmadi Kuwait Monthly Test Schedule Class: AS-LEVELapi-126472277No ratings yet
- PHD Thesis Report Format Guidelines: S.No. Guidelines SpecificationsDocument10 pagesPHD Thesis Report Format Guidelines: S.No. Guidelines SpecificationsAr Ayoushika AbrolNo ratings yet
- Finite Element and Theoretical Investigations On PVC-CFRP Confined Concrete Columns Under Axial CompressionDocument42 pagesFinite Element and Theoretical Investigations On PVC-CFRP Confined Concrete Columns Under Axial CompressionShaker QaidiNo ratings yet
- Writing ProposalsDocument100 pagesWriting ProposalsSaad Motawéa50% (2)
- Be11 enDocument12 pagesBe11 enSaqlain Mir100% (1)
- Tests For Surface Wellheads and BOP Equipment Used in Operations On Land and Above Water LevelDocument6 pagesTests For Surface Wellheads and BOP Equipment Used in Operations On Land and Above Water Levelahmed121No ratings yet
- Introduction To Educational Research Connecting Methods To Practice 1st Edition Lochmiller Test Bank DownloadDocument12 pagesIntroduction To Educational Research Connecting Methods To Practice 1st Edition Lochmiller Test Bank DownloadDelores Cooper100% (20)
- The Self From Various PerspectivesDocument32 pagesThe Self From Various PerspectivesAbegail ArellanoNo ratings yet
- Window Pane Reflection ProblemDocument8 pagesWindow Pane Reflection ProblemLee GaoNo ratings yet
- Rocket PropulsionDocument29 pagesRocket PropulsionPrajwal Vemala JagadeeshwaraNo ratings yet
- Liberty Ships Brittle Fracture (Final)Document5 pagesLiberty Ships Brittle Fracture (Final)hsemargNo ratings yet
- Secrets of the Millionaire Mind: Mastering the Inner Game of WealthFrom EverandSecrets of the Millionaire Mind: Mastering the Inner Game of WealthRating: 4.5 out of 5 stars4.5/5 (197)
- The Game: Penetrating the Secret Society of Pickup ArtistsFrom EverandThe Game: Penetrating the Secret Society of Pickup ArtistsRating: 4 out of 5 stars4/5 (131)
- Aruba, Bonaire & Curacao Adventure GuideFrom EverandAruba, Bonaire & Curacao Adventure GuideRating: 5 out of 5 stars5/5 (2)
- Proof of Heaven: A Neurosurgeon's Journey into the AfterlifeFrom EverandProof of Heaven: A Neurosurgeon's Journey into the AfterlifeRating: 3.5 out of 5 stars3.5/5 (165)
- Coastal Alaska & the Inside Passage Adventure Travel GuideFrom EverandCoastal Alaska & the Inside Passage Adventure Travel GuideNo ratings yet
- Geneva, Lausanne, Fribourg & Western Switzerland Travel AdventuresFrom EverandGeneva, Lausanne, Fribourg & Western Switzerland Travel AdventuresNo ratings yet
- Hollywood & the Best of Los Angeles Travel GuideFrom EverandHollywood & the Best of Los Angeles Travel GuideRating: 4.5 out of 5 stars4.5/5 (2)
- Jamaica: A Guide to the Food & RestaurantsFrom EverandJamaica: A Guide to the Food & RestaurantsRating: 4 out of 5 stars4/5 (1)