Professional Documents
Culture Documents
CT Advanded PDF
Uploaded by
adhad iofdshfOriginal Title
Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
CT Advanded PDF
Uploaded by
adhad iofdshfCopyright:
Available Formats
NUCLEON CHEMISTRY
CT for
CLASSS FOR IIT-JEE Target IIT-JEE 2016
Time : 1 hr Marks : 84
Atomic masses : [H = 1, D = 2, Li = 7, C = 12, N = 14, O = 16, F = 19, Na = 23, Mg = 24, Al = 27, Si = 28,
P = 31, S = 32, Cl = 35.5, K = 39, Ca = 40, Cr = 52, Mn = 55, Fe = 56, Cu = 63.5, Zn = 65, As = 75, Br = 80,
Ag = 108, I = 127, Ba = 137, Hg = 200, Pb = 207]
PAPER- (JEE-ADVANCED)
MCQ (9) [ 4 , –2]
1. 64 gm of an organic compound has 24 g carbon and rest hydrogen and oxygen. The empirical formula of the
compound is
(A) CH4O (B) CH2O (C) CH3OH (D) C2H8O2
2. When 2.86 g of a mixture of C4 H8 and C4 H10 was burnt in sufficient oxygen 4.14 g of water was formed. which
options are correct:
(A) 8.8 gm CO2 formed (B) 0.2 mole CO2 formed
(C) 1.12 g C4H8 in mixture (D) 0.03 mole C4H10
3. If 200 kg of 95% pure limestone (CaCO 3) is heated then
(A) mass of CaO obtained 106.4 kg. (B) mass of CaO obtained 212.8 kg.
(C) mass of CaO obtained 83.6 kg. (D) mass of CO 2 obtained 83.6 kg.
4. ‘When 100 ml of 80% (w/v) NaOH is mixed with certain amount of 40% (w/w) NaOH solution of density 1.2 gm/
ml, the percentage concentration of final solution becomes 60% (w/v). Which options are correct ?
(A) molarity = 10M (B) total volume of solution = 0.267M
(C) total moles of NaOH = 2 (D) molarity = 15M
5. 50 milliliters of CO is mixed with 20 mL of oxygen and sparked. After the reaction, the mixture is
treated with an aqueous KOH solution. Choose the correct option.
(A) The volume of the CO that reacts = 40 mL
(B) The volume of the CO 2 formed = 40 mL.
(C) The volume of the CO that remains after treatment with KOH = 10 mL
(D) The volume of the CO that remains after treatment with KOH = 20 mL
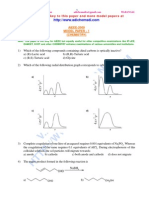
6. Which of the following is bondline structure of CH3 CH CH CH CH CH2
|
CH3
(A) (B)
(C) (D)
7. Select the correct options for molecular formula C2H2Cl2
(A) The total number of isomers is 4. (B) Only two compounds are geometrical isomers.
(C) All isomers have 5 bonds and one bond. (D) Its has linear shape.
Physical & Inorganic By Organic Chemistry By
NV Sir A-479 Indra vihar, kota
VKP Sir Page No. # 1
B.Tech. IIT Delhi
Ph. - 9982433693 (NV Sir) 9462729791(VKP Sir)
M.Sc. IT-BHU
8. The possible structures of molecular formula C4H6O2
O OH
O O O
(A) (B) (C) (D)
O HO
O
9. Which of the following statement is/are correct ?
(A) Isomers showing metamerism will have same functional group.
(B) All compounds having C = C bond exhibit geometrical isomerism.
(C) Stereoisomers can never be structural isomers.
(D) All structural isomers have different boiling point.
COMPREHENSION (1 × 2) [ 4 , –2]
Comprehension #
A hydrocarbon ‘X’ has 2 geometrical isomers. It has only one bond in its molecule along with 11
bonds.
10. Which of the following can be the structural isomer of the hydrocarbon ‘X’.
CH2 CH CH2 CH3 CH3 C CH2
(I ) |
CH3
( II)
(A) III, IV (B) I, II (C) II, IV (D) I, III
11. The ring chain functional isomer of hydrocarbon ‘X’ is / are
(A) I, II (B) II, III (C) III, IV (D) I, IV
INTEGER TYPE (8) [ 4, 0 ]
12. 264 g CO2 is completely converted into glucose C6H12O6 so that all carbon atoms are conserved. Find
minimum moles of O2 required for completely combustion of glucose :
13. 60 g C and 80 g O2 are mixed if percentage yield of reaction is 60% then how many moles of Fe2O3 can be
reduced in Fe from CO produced in above reaction :
14. The hydrated salt Na2SO 4.nH2O undergoes 50.3% loss in weight on heating and become anhydrous.
The value of 'n' will be :
15. What weight(in Kg) of CaCO3 must be decomposed to produce the sufficient quantity of carbondioxide to
convert 2.12 kg of Na2SO3 completely in to NaHCO3. [Atomic mass of Na = 23, Ca = 40]
CaCO3 CaO + CO2
Na2 CO3 + CO2 + H2O 2NaHCO3
Physical & Inorganic By Organic Chemistry By
NV Sir A-479 Indra vihar, kota
VKP Sir Page No. # 2
B.Tech. IIT Delhi
Ph. - 9982433693 (NV Sir) 9462729791(VKP Sir)
M.Sc. IT-BHU
16. 10 ml of sulphuric acid solution (sp. gr. = 1.84) contains 98% by weight of pure acid. Calculate the molarity
of NaOH solution required to completely neutralize the acid if volume of NaOH used is 184 ml.
17.
Degree of unsaturation of the above compound ?
18. The compound (X) has molecular formula C4H7Cl. Find out the number of its cyclic isomers (structural and
geometrical only excluding optical isomers).
19. The number of geometrical isomer possible for given compound
is
MTC (1) [ 2 × 4, -1 ]
Marking scheme For MTC
For each entry in Column-I, +2. If only the bubble(s) cooresponding to all the correct match(es) is(are)
darkned, 0 If none of the bubbles is darkned, –1 in all other cases.
20. Match the following :
Column-I Column-II
(A) 1 M glucose solution (p) 1 mol solute per litre solution
(B) 3 M urea solution (q) 180 g solute per litre solution
(C) 3 M CH3COOH solution (r) % w/v = 18% (solution)
(D) 1 M H2SO4 solution (s) % w/v = 9.8% (solution)
ANSWER KEY
1. (AC) 2. (ABCD) 3. (AD) 4. (BCD) 5. (ABC) 6. (BD) 7. (BC)
8. (BCD) 9. (ACD) 10. (B) 11. (D) 12. 6 13. 1 14. 8
15. 2 16. 2 17. 9 18. 5. 19. 8
20. A- p, q, r ; B- q, r ; C -q,r ; D- p, s
Physical & Inorganic By Organic Chemistry By
NV Sir A-479 Indra vihar, kota
VKP Sir Page No. # 3
B.Tech. IIT Delhi
Ph. - 9982433693 (NV Sir) 9462729791(VKP Sir)
M.Sc. IT-BHU
You might also like
- Straight Objective Type: Part-IDocument4 pagesStraight Objective Type: Part-Iaditya aryaNo ratings yet
- Fiitjee Coimbatore Centre: 10 - Chemistry - CPMDocument2 pagesFiitjee Coimbatore Centre: 10 - Chemistry - CPMC.M.M GAMINGNo ratings yet
- Paper I 11Document5 pagesPaper I 11master aexpeckNo ratings yet
- Chemistry Sbcic 50Document4 pagesChemistry Sbcic 50Sagar AnawadeNo ratings yet
- Some Basic Concept of Chemistry: Chapter - 01Document7 pagesSome Basic Concept of Chemistry: Chapter - 01MidhunNo ratings yet
- MCQ Module V Btech 2nd SemDocument6 pagesMCQ Module V Btech 2nd SemUttam NeelapureddyNo ratings yet
- Mole and Stoichiometric CalculationDocument18 pagesMole and Stoichiometric CalculationSajjad MiraniNo ratings yet
- Chapter 1 3 Class Xi MCQDocument11 pagesChapter 1 3 Class Xi MCQZaid KhanNo ratings yet
- Midterm Preparation DocumentDocument15 pagesMidterm Preparation DocumentbettieboomNo ratings yet
- Test 6Document11 pagesTest 6Prakash KapadiaNo ratings yet
- Time: 3.00 Hours) /maximum Marks: 100: This Question Paper Contains 8 Printed PagesDocument8 pagesTime: 3.00 Hours) /maximum Marks: 100: This Question Paper Contains 8 Printed PagesOmpratapNo ratings yet
- Question Bank-Chemistry (043) - Xi Annual FinalDocument32 pagesQuestion Bank-Chemistry (043) - Xi Annual Finalsushobhanmahapatra19No ratings yet
- SS3 Chemistry First TermDocument5 pagesSS3 Chemistry First TermKel FelixNo ratings yet
- CHEM101 172 Final SolvedDocument12 pagesCHEM101 172 Final SolvedTorong VNo ratings yet
- Chemistry: PlaneDocument3 pagesChemistry: PlanegamerwizcastNo ratings yet
- Chemistry: Crash Course For JEE Main 2020Document14 pagesChemistry: Crash Course For JEE Main 2020johnNo ratings yet
- Matrix Science Academy: Chemistry MHT CET L2 2022-23Document4 pagesMatrix Science Academy: Chemistry MHT CET L2 2022-23Light MayNo ratings yet
- Arjuna JEE Main Test 01 - ChemistryDocument6 pagesArjuna JEE Main Test 01 - Chemistrymriitian56No ratings yet
- Coordination Compounds 109 QuestionsDocument19 pagesCoordination Compounds 109 QuestionsAnkit kumarNo ratings yet
- Chemistry Term 1 MCQ 2021Document29 pagesChemistry Term 1 MCQ 2021manish dagarNo ratings yet
- IX Chem Mole Concept Kailash Khatwani FinalDocument8 pagesIX Chem Mole Concept Kailash Khatwani FinalAditya ParuiNo ratings yet
- 1-Mole & RedoxDocument3 pages1-Mole & RedoxArnab DasNo ratings yet
- Quice Review Center: C) The Total Mass of The Atom A) RBDocument5 pagesQuice Review Center: C) The Total Mass of The Atom A) RBMary Francia RicoNo ratings yet
- Test Paper - 1 (Arihant)Document7 pagesTest Paper - 1 (Arihant)kumarsanu2905No ratings yet
- Mole CPP Combine PDFDocument13 pagesMole CPP Combine PDFPiyushNo ratings yet
- C1 Home AssignmentDocument2 pagesC1 Home AssignmentMemoona GullNo ratings yet
- C1 Home AssignmentDocument2 pagesC1 Home AssignmentMemoona GullNo ratings yet
- C1 Home AssignmentDocument2 pagesC1 Home AssignmentMemoona GullNo ratings yet
- C1 Home AssignmentDocument2 pagesC1 Home AssignmentMemoona GullNo ratings yet
- Stepchem 11 PDFDocument13 pagesStepchem 11 PDFSyeda Fatima ZahraNo ratings yet
- Chemitry (Comprehensive Exam)Document3 pagesChemitry (Comprehensive Exam)sehar aliNo ratings yet
- AL Chemistry 1996 Paper 1+2Document12 pagesAL Chemistry 1996 Paper 1+2api-3734333No ratings yet
- Practice Sheet 1.2Document7 pagesPractice Sheet 1.2daya nandNo ratings yet
- Pre Board Chemsirty 11thDocument2 pagesPre Board Chemsirty 11thSyed Raza Hassan GardeziNo ratings yet
- CHE 110 Exam 2 F'13 v1Document6 pagesCHE 110 Exam 2 F'13 v1MicahNo ratings yet
- TS - X Chemistry All DCEB Papers Chapter Wise Academic Standard Wise Prefinal - I & 2 QuestionsDocument40 pagesTS - X Chemistry All DCEB Papers Chapter Wise Academic Standard Wise Prefinal - I & 2 Questionsc18180707No ratings yet
- 17 Chemistry1Document3 pages17 Chemistry1Abdul MuneerNo ratings yet
- Class XiDocument7 pagesClass XiBenson BennyNo ratings yet
- Aieee Assign Chemistry 01Document3 pagesAieee Assign Chemistry 01rocklandgetNo ratings yet
- Ch. 1,2,3,4Document29 pagesCh. 1,2,3,4Kankana ChoudhuryNo ratings yet
- Mole & Eqt. ConceptDocument10 pagesMole & Eqt. ConceptthilaivananNo ratings yet
- 2018 Chemistry Standardised Test For Science Stream (SPM)Document7 pages2018 Chemistry Standardised Test For Science Stream (SPM)carnationNo ratings yet
- DPT-2 Chem & Zoo Neet 29.12.2023Document8 pagesDPT-2 Chem & Zoo Neet 29.12.2023pinnaacleclasses salemNo ratings yet
- 01) Xii Theory Paper 24-01-24Document3 pages01) Xii Theory Paper 24-01-24bbfnpsy2cdNo ratings yet
- Mole ConceptDocument7 pagesMole ConceptTahir Raj BhasinNo ratings yet
- Chemistry Live Quiz-1 QuestionsDocument2 pagesChemistry Live Quiz-1 Questionslavya guptaNo ratings yet
- In 2012 E.C Rehoboth Academy Chemistry Worksheet For Grade 8 Students Unit 4 Environmental ChemistryDocument4 pagesIn 2012 E.C Rehoboth Academy Chemistry Worksheet For Grade 8 Students Unit 4 Environmental ChemistryWedaje AlemayehuNo ratings yet
- كيمياء انجليزي12-3Document17 pagesكيمياء انجليزي12-3Ahmed BasemNo ratings yet
- 04 ChemistryDocument4 pages04 ChemistryAgneebha GhoshNo ratings yet
- XI - Chemistry Model Question PaperDocument16 pagesXI - Chemistry Model Question PaperLakshmi SinghNo ratings yet
- Prepared by v. Aditya VardhanDocument6 pagesPrepared by v. Aditya Vardhankrishna kanthNo ratings yet
- 11 Chemistry23 24sp 01Document13 pages11 Chemistry23 24sp 01AbhishekNo ratings yet
- Chemistry 100 Days Challenge CPPDocument141 pagesChemistry 100 Days Challenge CPPFardeen MalickNo ratings yet
- 2000-2019 Nesa Chemistry Advanced Level-1Document269 pages2000-2019 Nesa Chemistry Advanced Level-1Jeff AlbaNo ratings yet
- IB Stiochiometry QuestionDocument10 pagesIB Stiochiometry QuestionPakorn Winayanuwattikun0% (1)
- Latihan Ulangkaji Chapter 3: Chemical Formula and EquationDocument15 pagesLatihan Ulangkaji Chapter 3: Chemical Formula and EquationIzz KhawarizmiNo ratings yet
- ARC CatalogDocument416 pagesARC Catalogdomitian88No ratings yet
- Group 15 Elements: - D (Pi) BondDocument7 pagesGroup 15 Elements: - D (Pi) BondSanju PatelNo ratings yet
- PPT-SpecKare? GDL (Gluconolactone) Photo-Sensitive & Photo-Aging Repair & Protection EN 20210202Document29 pagesPPT-SpecKare? GDL (Gluconolactone) Photo-Sensitive & Photo-Aging Repair & Protection EN 20210202Kenneth CfkNo ratings yet
- Antioxidants Feb 2016 enDocument6 pagesAntioxidants Feb 2016 enPhilip Blair OngNo ratings yet
- 16 Ganesh DDocument16 pages16 Ganesh DGanesh DhobaleNo ratings yet
- Bridge ElementsDocument28 pagesBridge ElementsBea Lha Zandra BesingaNo ratings yet
- Luliconazole Specs and STP - From DMF - Nosch LabsDocument22 pagesLuliconazole Specs and STP - From DMF - Nosch Labsshakt londaNo ratings yet
- AP Chemistry Mini-LabsDocument1 pageAP Chemistry Mini-LabsNori HeplerNo ratings yet
- HUG y LEUPIN 2003Document9 pagesHUG y LEUPIN 2003Juan Ignacio Gonzalez CabreraNo ratings yet
- PKa Table of AcidsDocument1 pagePKa Table of AcidsGuery SaenzNo ratings yet
- Kalkulator AB Mix 1Document82 pagesKalkulator AB Mix 1Erwin100% (1)
- Membrane Cleaning and Support ChemicalsDocument5 pagesMembrane Cleaning and Support ChemicalsMohsin AliNo ratings yet
- Screening of Litsea SalicifoliaDocument21 pagesScreening of Litsea SalicifoliaMarmish DebbarmaNo ratings yet
- Practice Packet: Chapter 8:organicchemistryDocument45 pagesPractice Packet: Chapter 8:organicchemistryKerala MekuriyaNo ratings yet
- CHEM 15 - Lec 04 - StoichiometryDocument27 pagesCHEM 15 - Lec 04 - StoichiometryLetecia DughoNo ratings yet
- Manual For Determination of Carotenoids in Deferent Food ItemsDocument24 pagesManual For Determination of Carotenoids in Deferent Food ItemsTadesse Gebregiyorgis TakeleNo ratings yet
- Table of ContentsDocument34 pagesTable of ContentslaraNo ratings yet
- 86 TricksDocument164 pages86 Trickslozerface8863% (8)
- Moles Moles H O Moles Moles H O H O HC H O: Stoichiometry of SolutionsDocument3 pagesMoles Moles H O Moles Moles H O H O HC H O: Stoichiometry of SolutionsElle KateNo ratings yet
- Citric AcidDocument2 pagesCitric AcidIqbal batchaNo ratings yet
- Class 10 Science Chapter NotesDocument192 pagesClass 10 Science Chapter NotesTarunNo ratings yet
- Volatile Chemicals From Thermal Degradation of Less Volatile Coffee ComponentsDocument7 pagesVolatile Chemicals From Thermal Degradation of Less Volatile Coffee ComponentsemiliaNo ratings yet
- Reaksi PA Dan ButanolDocument5 pagesReaksi PA Dan ButanolUnlucky 2019No ratings yet
- 3.1a PRACTICE EXERCISE (Dela Vega)Document8 pages3.1a PRACTICE EXERCISE (Dela Vega)Aivan NovillaNo ratings yet
- Experimental Dipoles - Dipole Moments in DebyeDocument82 pagesExperimental Dipoles - Dipole Moments in DebyeYourMotherNo ratings yet
- STP 742-1981Document238 pagesSTP 742-1981Feliandro FirmansyahNo ratings yet
- Astm D 2036-98 CianuroDocument20 pagesAstm D 2036-98 CianuroGianinaRoncalChávezNo ratings yet
- Chapter 62 Structured QuestionsDocument23 pagesChapter 62 Structured QuestionsytNo ratings yet
- HardnessDocument3 pagesHardnessVara PrasadNo ratings yet