Professional Documents
Culture Documents
Chemistry 12 Worksheet 4-6 Anhydrides, Acid Rain and Titrations
Uploaded by
Bekki VanderlendeOriginal Description:
Original Title
Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
Chemistry 12 Worksheet 4-6 Anhydrides, Acid Rain and Titrations
Uploaded by
Bekki VanderlendeCopyright:
Available Formats
Chemistry 12 Worksheet 4-6—Anhydrides , Acid Rain and Titrations
Chemistry 12
Worksheet 4-6
Anhydrides, Acid Rain and Titrations
Name __________________________
71
Date Due _______________________
Correct & Hand In by ____________________
This worksheet covers material from class notes and Student Workbook pages 154-158,
and pages 186-188.
1. State whether the following compounds will act as acids (A) or bases (B) when
added to water. (10 marks)
a) ClO 2 ...... ___________ f) CO2 ...... ___________
b) SrO...... ___________ g) Cs2O ... ___________
c) Cr2O3 ... ___________ h) BaO ..... ___________
d) SeO 2 .... ___________ i) SO3 ...... ___________
e) NO2 ..... ___________ j) Ag2O ..... ___________
2. a) Define a basic anhydride (1 mark) -
b) Give three examples of basic anhydrides (make sure formulas are correct!) (3 marks)
_______________ _______________ _______________
3. a) Define an acidic anhydride (1 mark) -
b) Give three examples of acidic anhydrides (make sure formulas are correct!) (3 marks)
_______________ _______________ _______________
18
Chemistry 12 Worksheet 4-6—Anhydrides , Acid Rain and Titrations Page 1 of 6 Pages
Chemistry 12 Worksheet 4-6—Anhydrides , Acid Rain and Titrations
4. Give three examples of amphoteric (or amphiprotic) anhydrides.(3 marks)
_______________ _______________ _______________
The element other than oxygen in these is found near the ________________________
of the periodic table.(1 mark)
5. Normal (unpolluted) rain water usually has a pH of about _________. This is
less than 7 due to __________gas dissolving in the air.(2 marks)
6. “Acid rain” could be defined as rain having a pH below ___________. (1 mark)
7. Fossil fuels containing the element ________ are a major cause of acid rain. (1 mark)
8. Give two formula equations which show how sulphurous acid can be produced
starting with sulphur. (2 marks)
1.
2.
9. Give three formula equations which show how sulphuric acid can be produced
starting with sulphur. (3 marks)
1.
2.
3.
10. The element _________ reacts with oxygen in the cylinders of automobile motors
to produce various nitrogen oxides. (1 mark)
11. Give the balanced equation for the formation of NO from it’s elements. (1 mark)
__________________________________________________________________
15
Chemistry 12 Worksheet 4-6—Anhydrides , Acid Rain and Titrations Page 2 of 6 Pages
Chemistry 12 Worksheet 4-6—Anhydrides , Acid Rain and Titrations
12. Give the balanced equation for the formation of NO2 from it’s elements. (1 mark)
_________________________________________________________________
13. Give the balanced equation for the formation of nitrous and nitric acid from NO2
dissolving in rain water. (1 mark)
_________________________________________________________________
14. Other than cars and fossil fuel burning power plants, what are some other sources
of acid rain? (2 marks)
_________________________________________________________________
15. Explain why acid rain falling on one area may do less damage than the same acid rain
falling in a different area. (1 mark)
16. Give four major unfavourable effects of acid rain. (4 marks)
1.
2.
3.
4.
17. Give four ways in which acid rain and the problems associated with it could be solved or
at least helped (4 marks)
1.
2.
3.
4.
13
Chemistry 12 Worksheet 4-6—Anhydrides , Acid Rain and Titrations Page 3 of 6 Pages
Chemistry 12 Worksheet 4-6—Anhydrides , Acid Rain and Titrations
18. 13.45 mL of 0.200 M NaOH is required to titrate 25.0 mL of a solution which is
known to have HCl. Calculate the original [HCl]. Show all your steps. (3 marks)
19. 13.45 mL of 0.200 M HCl is required to titrate 25.0 mL of a solution which is
known to have Ba(OH)2. Calculate the original [Ba(OH)2]. Show all your steps.
(3 marks)
20. 13.45 mL of 0.200 M Sr(OH)2 is required to titrate 25.0 mL of a solution which is
known to have HNO3. Calculate the original [HNO3]. Show all your steps. (3 marks)
Chemistry 12 Worksheet 4-6—Anhydrides , Acid Rain and Titrations Page 4 of 6 Pages
Chemistry 12 Worksheet 4-6—Anhydrides , Acid Rain and Titrations
21. What volume of 0.100 M NaOH would be required to titrate 35.0 mL of a
0.231M solution of H2C2O4. Show all your steps. (3 marks)
22. Consider the following 0.100 M solutions:
I. HF II. HBr III. H2SO4
The equivalence point is reached when 10.00 mL of 0.100 M NaOH has been
added to 10.00 mL of solutions (1 mark)
A. II only B. I and II only C. II and III only D. I, II and III
23. a) Write the balanced formula equation for the titration between sulphurous acid
and potassium hydroxide. (1 mark)
__________________________________________________________
b) Write the balanced net-ionic equation for the titration between sulphurous acid
and potassium hydroxide. (1 mark)
__________________________________________________________
24. Given the following data table:
Beaker Volume Contents
1 10.0 mL 0.1 M Ba(OH)2
2 15.0 mL 0.2 M NH3
3 20.0 mL 0.05 M KOH
4 50.0 mL 0.2 M NaOH
a) Which beaker would require the greatest volume of 0.1M HCl for complete
neutralization? ________________ (1 mark)
Chemistry 12 Worksheet 4-6—Anhydrides , Acid Rain and Titrations Page 5 of 6 Pages
Chemistry 12 Worksheet 4-6—Anhydrides , Acid Rain and Titrations
b) What volume of 0.1M HCl would be needed for the neutralization in (a)?
(2 marks)
Answer ___________mL
c) Which beaker would require the least volume of 0.1M HCl for complete
neutralization? _______________ (1 mark)
d) What volume of 0.1M HCl would be needed for the neutralization in (c)?
(2 marks)
Answer ___________mL
25. Calculate the mass of NaOH which is required to neutralize 15.00 mL of
0.350 M H2SO4? (3 marks)
Answer __________grams
26. When a 0.1 M strong base titrates a 0.1 M weak monoprotic acid, it takes
(less/more/the same)_____________ volume of the base as it would to titrate a
0.1 M strong monoprotic acid. (1 mark)
Chemistry 12 Worksheet 4-6—Anhydrides , Acid Rain and Titrations Page 6 of 6 Pages
You might also like
- CSEC Chemistry January 2011 P2Document18 pagesCSEC Chemistry January 2011 P2AshleyNo ratings yet
- Daily Paragraph Editing 6Document2 pagesDaily Paragraph Editing 6Bekki Vanderlende100% (1)
- Chemistry 20 Unit C Solutions Quiz QuestionsDocument7 pagesChemistry 20 Unit C Solutions Quiz Questionsapi-265758110No ratings yet
- Precipitation ReactionsDocument3 pagesPrecipitation ReactionsborgiamatriceNo ratings yet
- Options To Treat Refractory OresDocument35 pagesOptions To Treat Refractory OresAlyssa Mae BolangaNo ratings yet
- Ajinomoto FormulationguideDocument17 pagesAjinomoto FormulationguidelalitNo ratings yet
- (S11ES-Ib-5) : I. Content II. Learning ResourcesDocument3 pages(S11ES-Ib-5) : I. Content II. Learning ResourcesCE YM100% (1)
- IB Chemistry Worksheet 1: Significant Figures and Dimensional AnalysisDocument17 pagesIB Chemistry Worksheet 1: Significant Figures and Dimensional AnalysisFinn Bruchet100% (1)
- B. Gas AnalysisDocument5 pagesB. Gas AnalysisSoma GhoshNo ratings yet
- First Day - GR 11 Review IIDocument4 pagesFirst Day - GR 11 Review IIAmana AlamNo ratings yet
- ATAR Chemistry Year 12 Asc 2017 (WA)Document12 pagesATAR Chemistry Year 12 Asc 2017 (WA)Raghav GanaNo ratings yet
- Quantititive Chemistry - Paper 1 TES - 4Document37 pagesQuantititive Chemistry - Paper 1 TES - 4KshitijNo ratings yet
- Weird Chem FinalDocument9 pagesWeird Chem Finalalludra_spicaNo ratings yet
- Exercise: HCL + H O (Aq) + CL (Aq)Document3 pagesExercise: HCL + H O (Aq) + CL (Aq)baskieNo ratings yet
- Exam 2Document11 pagesExam 2honkytonkzNo ratings yet
- Chemistry Test Review - Mar 2023Document4 pagesChemistry Test Review - Mar 2023rblxproNo ratings yet
- Subject: Chemistry Chapter #: 6: Quantitative Chemistry - How Much? (Moles)Document26 pagesSubject: Chemistry Chapter #: 6: Quantitative Chemistry - How Much? (Moles)Haider Ali100% (2)
- CHE 110 Exam 2 F'13 v1Document6 pagesCHE 110 Exam 2 F'13 v1MicahNo ratings yet
- Test 2-P2Document8 pagesTest 2-P2Salman Ul MoazzamNo ratings yet
- Neutralisation reactions and acid-base titrationsDocument8 pagesNeutralisation reactions and acid-base titrationsWong Hui XinNo ratings yet
- Stoichiometry Introduction Worksheet - Chem1Document2 pagesStoichiometry Introduction Worksheet - Chem1Riley SharkNo ratings yet
- 2 U2 and U3 Review 1Document7 pages2 U2 and U3 Review 1angle5858zhaoNo ratings yet
- May 2014Document52 pagesMay 2014Dylan EllulNo ratings yet
- Acid Base SAC RevisionDocument5 pagesAcid Base SAC RevisionAntony ThabesanNo ratings yet
- ShapesofcomplexionsDocument41 pagesShapesofcomplexions/ “Nu” /No ratings yet
- Practice Questions Unit 2Document13 pagesPractice Questions Unit 2Kaitlyn CarrNo ratings yet
- Std. X Chemistry PAPER IV ExtendedDocument8 pagesStd. X Chemistry PAPER IV ExtendedYashodhaNo ratings yet
- Theory Worksheet: Acids, Bases and SaltsDocument4 pagesTheory Worksheet: Acids, Bases and Saltsخانزاده بلال احمدخان لودہیNo ratings yet
- Sep 2017Document32 pagesSep 2017Dylan EllulNo ratings yet
- ChemistryDocument12 pagesChemistryW. Joseph the chemistNo ratings yet
- 2012 June Exam Y10 Chemistry BDF New CourseDocument6 pages2012 June Exam Y10 Chemistry BDF New CourseLeilaNo ratings yet
- Formative Test of Form 4 ChemistryDocument5 pagesFormative Test of Form 4 ChemistryMarinda YieNo ratings yet
- Introduction to Chemical Processes Exam 1 ProblemsDocument7 pagesIntroduction to Chemical Processes Exam 1 ProblemsDaniela Andrea Vargas MedinaNo ratings yet
- Acids and Bases QuestionsDocument14 pagesAcids and Bases Questionsmariam saikNo ratings yet
- Rate of Reaction FactorsDocument8 pagesRate of Reaction FactorsAmirah Noor AffandiNo ratings yet
- Acidsandbases AllDocument95 pagesAcidsandbases AllarindamNo ratings yet
- AP CH 4 ReviewDocument3 pagesAP CH 4 ReviewAmanda ClayNo ratings yet
- Ka & Kb worksheetDocument4 pagesKa & Kb worksheetHumphrey LolozhiNo ratings yet
- Chem 1a Turnin WkshopsDocument45 pagesChem 1a Turnin WkshopsELLEN CERNANo ratings yet
- Year 11 Chemistry Time: 2 HoursDocument12 pagesYear 11 Chemistry Time: 2 HoursAdrianHedleyNo ratings yet
- Year 7 A-B Chemistry Term 1Document45 pagesYear 7 A-B Chemistry Term 1H ChowdreyNo ratings yet
- Year 11 Chemistry Time: 2 HoursDocument11 pagesYear 11 Chemistry Time: 2 HoursAdrianHedleyNo ratings yet
- MolesDocument16 pagesMolesChaudhryAbdullahNo ratings yet
- Hydrolysis Worksheet Chemistry Unit 4.13Document3 pagesHydrolysis Worksheet Chemistry Unit 4.13Gideon CavidaNo ratings yet
- 3na CHEM End-Of-year 09Document13 pages3na CHEM End-Of-year 09Francis Ho HoNo ratings yet
- Alkane and Alkene Review Question MSDocument19 pagesAlkane and Alkene Review Question MSjw wNo ratings yet
- Spring Review Chem 2013Document14 pagesSpring Review Chem 2013Halee RobinsonNo ratings yet
- Old Final Fall 2015Document18 pagesOld Final Fall 2015JessicaNo ratings yet
- Chem1 Problem Set 1 StoichiometryDocument2 pagesChem1 Problem Set 1 StoichiometrynicolassarragaNo ratings yet
- carbonyls ppqs NO ANSWERSDocument9 pagescarbonyls ppqs NO ANSWERSmariam saikNo ratings yet
- Acids and Bases: Titration #1 Determination of (Naoh) by Microtitration With HCL of Known ConcentrationDocument90 pagesAcids and Bases: Titration #1 Determination of (Naoh) by Microtitration With HCL of Known ConcentrationKim Shyen BontuyanNo ratings yet
- MODUL ENDEAVOUR CHEMISTRY A+ REVIEWDocument18 pagesMODUL ENDEAVOUR CHEMISTRY A+ REVIEWMuhammad Syafiq AsyrafNo ratings yet
- Molarity 2 HW: Show All Work Using Dimensional Analysis!Document1 pageMolarity 2 HW: Show All Work Using Dimensional Analysis!Anthony WangNo ratings yet
- Chemistry - Ordinary Level: An Roinn Oideachais Agus EolaíochtaDocument7 pagesChemistry - Ordinary Level: An Roinn Oideachais Agus Eolaíochta4162897039No ratings yet
- Worksheets12 KDAV Worksheet Solution - Session 2019-2020-DPDocument1 pageWorksheets12 KDAV Worksheet Solution - Session 2019-2020-DPKashyap PatelNo ratings yet
- Carbon Compound Isomers TestDocument4 pagesCarbon Compound Isomers TestKeman MjNo ratings yet
- L13 1e-Calculating The RFM and Using Moles WiDocument7 pagesL13 1e-Calculating The RFM and Using Moles WiOMarNo ratings yet
- Honors Chemistry EquationsDocument2 pagesHonors Chemistry EquationsShadae ClarkeNo ratings yet
- 4 Chemistry Assignemnt-1 HHWDocument2 pages4 Chemistry Assignemnt-1 HHWFINE staffNo ratings yet
- Year 10 Chemistry Time: 2 HoursDocument12 pagesYear 10 Chemistry Time: 2 HoursAdrianHedleyNo ratings yet
- May 2017Document52 pagesMay 2017Dylan EllulNo ratings yet
- C Annual 2023Document12 pagesC Annual 2023AdrianHedleyNo ratings yet
- Placing decimals with multiplicationDocument1 pagePlacing decimals with multiplicationBekki Vanderlende0% (1)
- A Study of Tribal Composition and Conflict in Darfur: Gao YifeiDocument4 pagesA Study of Tribal Composition and Conflict in Darfur: Gao YifeiBekki VanderlendeNo ratings yet
- Chemistry 12 Notes on Unit 4 TitrationDocument24 pagesChemistry 12 Notes on Unit 4 TitrationBekki VanderlendeNo ratings yet
- Vernier Act9 Evaporation of AlcoholsDocument8 pagesVernier Act9 Evaporation of AlcoholsBerra SirmaliNo ratings yet
- One StepDocument4 pagesOne StepAnitha NathenNo ratings yet
- Chemistry 12 Notes on Unit 4 TitrationDocument24 pagesChemistry 12 Notes on Unit 4 TitrationBekki VanderlendeNo ratings yet
- Mult Decimals 2Document1 pageMult Decimals 2Bekki VanderlendeNo ratings yet
- Perimeter Triangles 1Document1 pagePerimeter Triangles 1api-262413275100% (1)
- Perimeter Triangles 1Document1 pagePerimeter Triangles 1api-262413275100% (1)
- Perimeter Triangles 1Document1 pagePerimeter Triangles 1api-262413275100% (1)
- Perimeter Triangles 1Document1 pagePerimeter Triangles 1api-262413275100% (1)
- One StepDocument4 pagesOne StepAnitha NathenNo ratings yet
- Perimeter Triangles 1Document1 pagePerimeter Triangles 1api-262413275100% (1)
- Mult Decimals 2Document1 pageMult Decimals 2Bekki VanderlendeNo ratings yet
- Chemistry 12 Notes on Unit 4 TitrationDocument24 pagesChemistry 12 Notes on Unit 4 TitrationBekki VanderlendeNo ratings yet
- Placing decimals with multiplicationDocument1 pagePlacing decimals with multiplicationBekki Vanderlende0% (1)
- Perimeter Triangles 1Document1 pagePerimeter Triangles 1api-262413275100% (1)
- Chemistry 12 Worksheet 4-6 Anhydrides, Acid Rain and TitrationsDocument6 pagesChemistry 12 Worksheet 4-6 Anhydrides, Acid Rain and TitrationsBekki VanderlendeNo ratings yet
- One StepDocument4 pagesOne StepAnitha NathenNo ratings yet
- Operations With Fractions (A) : Calculate The Answer To Each QuestionDocument2 pagesOperations With Fractions (A) : Calculate The Answer To Each QuestionBekki VanderlendeNo ratings yet
- Placing decimals with multiplicationDocument1 pagePlacing decimals with multiplicationBekki Vanderlende0% (1)
- Multiplying With Decimals: 1) 2) 3) Solve Each ProblemDocument1 pageMultiplying With Decimals: 1) 2) 3) Solve Each ProblemBekki VanderlendeNo ratings yet
- Rutherford Model Virtual Inquiry (40mDocument4 pagesRutherford Model Virtual Inquiry (40mBekki VanderlendeNo ratings yet
- Function Machines 1Document2 pagesFunction Machines 1Bekki VanderlendeNo ratings yet
- Finite Geometric SeriesDocument4 pagesFinite Geometric SeriesGeorge HouNo ratings yet
- Function MachinesDocument1 pageFunction MachinesBekki VanderlendeNo ratings yet
- Arithmetic Series: Evaluate The Related Series of Each SequenceDocument4 pagesArithmetic Series: Evaluate The Related Series of Each SequenceBekki VanderlendeNo ratings yet
- Function MachinesDocument1 pageFunction MachinesBekki VanderlendeNo ratings yet
- Operations With Fractions (A) : Calculate The Answer To Each QuestionDocument2 pagesOperations With Fractions (A) : Calculate The Answer To Each QuestionBekki VanderlendeNo ratings yet
- Essential & Trace Elements: Iron, Copper, Zinc CompoundsDocument38 pagesEssential & Trace Elements: Iron, Copper, Zinc CompoundsVinod MuleNo ratings yet
- Summary of Chapter 7 HaloalkanesDocument3 pagesSummary of Chapter 7 Haloalkanesfaris zainuddinNo ratings yet
- Iones ComplejosDocument18 pagesIones ComplejosSantiago AlexanderNo ratings yet
- Molecules 12 01796Document9 pagesMolecules 12 01796Kalpesh PatelNo ratings yet
- Excellent Extended Essay (Chemistry)Document38 pagesExcellent Extended Essay (Chemistry)Aqeel JariwallaNo ratings yet
- Ammonium Copper (II) Sulphate PreparationDocument5 pagesAmmonium Copper (II) Sulphate PreparationMpilo ManyoniNo ratings yet
- Art Integrated Learning Project (Ritik Kumar)Document49 pagesArt Integrated Learning Project (Ritik Kumar)abhiabhishek6073No ratings yet
- Cambridge IGCSE: CHEMISTRY 0620/63Document12 pagesCambridge IGCSE: CHEMISTRY 0620/63lovely12807No ratings yet
- ZircadyneDocument18 pagesZircadyneClaudia MmsNo ratings yet
- Toward Industrial Catalysis of Zeolite For Linear Alkylbenzene Synthesis, A Mini ReviewDocument8 pagesToward Industrial Catalysis of Zeolite For Linear Alkylbenzene Synthesis, A Mini ReviewIrpan RomadonaNo ratings yet
- Experiment No. 7 Alcohols and Phenols Part A. AlcoholsDocument8 pagesExperiment No. 7 Alcohols and Phenols Part A. Alcoholsgeyb away0% (1)
- ColourDocument21 pagesColourSreedevi KrishnakumarNo ratings yet
- UCAR™ Latex DL 450 - MielcoDocument5 pagesUCAR™ Latex DL 450 - MielcoLong An DoNo ratings yet
- LPP BiomoleculesDocument6 pagesLPP BiomoleculesAashiNo ratings yet
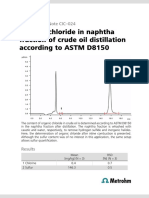
- Organic Chloride in Naphtha Fraction of Crude Oil Distillation According To ASTM D8150Document2 pagesOrganic Chloride in Naphtha Fraction of Crude Oil Distillation According To ASTM D8150HUYQUYENNo ratings yet
- Assumptions For VER EstimationDocument17 pagesAssumptions For VER EstimationPukhraj SethiyaNo ratings yet
- 化學奧林匹亞冬令營 分析3 2018Document76 pages化學奧林匹亞冬令營 分析3 2018楊泰萱No ratings yet
- School Project On Soft DrinksDocument14 pagesSchool Project On Soft DrinksBala Sundaram100% (1)
- Phenobarbital PDFDocument12 pagesPhenobarbital PDFunikwlnNo ratings yet
- Class 12 Chemistry Syllabus 2022-23 by Physics LinxDocument18 pagesClass 12 Chemistry Syllabus 2022-23 by Physics LinxRudar MalviyaNo ratings yet
- Volumetric Analysis: Lab ReportDocument6 pagesVolumetric Analysis: Lab ReportJeff LimNo ratings yet
- Acidity in TeaDocument17 pagesAcidity in TeaRukmani 1011No ratings yet
- Buffers: Maintaining pH EquilibriumDocument62 pagesBuffers: Maintaining pH EquilibriumSara FatimaNo ratings yet
- BIOCHEM FR 2 Qualitative Color ReactionsDocument6 pagesBIOCHEM FR 2 Qualitative Color ReactionsJNo ratings yet
- Oil Analysis Report: PT Petrolab ServicesDocument1 pageOil Analysis Report: PT Petrolab ServicesJon HermanNo ratings yet
- My TestDocument3 pagesMy TestMeatball BananaNo ratings yet