Professional Documents
Culture Documents
N Factor
Uploaded by
zahir khan0 ratings0% found this document useful (0 votes)
12 views2 pagesRedox reactions involve the transfer of electrons between reactants. Oxidation is the loss of electrons while reduction is the gain of electrons. Common redox reactions include combustion reactions where a fuel is oxidized and an oxidant like oxygen is reduced, as well as corrosion where a metal oxidizes as it reacts with oxygen and water.
Original Description:
Original Title
n-Factor
Copyright
© © All Rights Reserved
Available Formats
PDF, TXT or read online from Scribd
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentRedox reactions involve the transfer of electrons between reactants. Oxidation is the loss of electrons while reduction is the gain of electrons. Common redox reactions include combustion reactions where a fuel is oxidized and an oxidant like oxygen is reduced, as well as corrosion where a metal oxidizes as it reacts with oxygen and water.
Copyright:
© All Rights Reserved
Available Formats
Download as PDF, TXT or read online from Scribd
0 ratings0% found this document useful (0 votes)
12 views2 pagesN Factor
Uploaded by
zahir khanRedox reactions involve the transfer of electrons between reactants. Oxidation is the loss of electrons while reduction is the gain of electrons. Common redox reactions include combustion reactions where a fuel is oxidized and an oxidant like oxygen is reduced, as well as corrosion where a metal oxidizes as it reacts with oxygen and water.
Copyright:
© All Rights Reserved
Available Formats
Download as PDF, TXT or read online from Scribd
You are on page 1of 2
28 October 2020 19:23
Redox reaction VFSC1 Page 1
Redox reaction VFSC1 Page 2
You might also like
- Bill Akhir Tn. Antonius LieDocument2 pagesBill Akhir Tn. Antonius LieoktavianuspandianganNo ratings yet
- LESSONDocument9 pagesLESSONYUFENYUY SEVERINE W.No ratings yet
- Tentative and Assessment Form 4 PDFDocument1 pageTentative and Assessment Form 4 PDFMoises CentenoNo ratings yet
- Note # 25: May Be ApprovedDocument3 pagesNote # 25: May Be ApprovedSanttosh Kummar AgathamudiNo ratings yet
- Emirates Skycargo Flight Schedule: Flight Number Route Type Origin Destination STD Date STD Sta Date Sta Day of WeekDocument11 pagesEmirates Skycargo Flight Schedule: Flight Number Route Type Origin Destination STD Date STD Sta Date Sta Day of WeekRajNo ratings yet
- MMH 582 Ul Series My99 m17 Ed0 Rev1Document304 pagesMMH 582 Ul Series My99 m17 Ed0 Rev1diegoNo ratings yet
- Week 23 PR Residence: ExpensesDocument1 pageWeek 23 PR Residence: ExpensesMayieNo ratings yet
- OH Weekly Report 02 Oktober - 08 Oktober 2017 ALL FARDocument76 pagesOH Weekly Report 02 Oktober - 08 Oktober 2017 ALL FARFahroni ErlianurNo ratings yet
- GEP June 2020 Chapter1 Box2Document59 pagesGEP June 2020 Chapter1 Box2Ronald Pally OrtizNo ratings yet
- Rapat Hasil PTS Ganjil T.P 2020-2021Document2 pagesRapat Hasil PTS Ganjil T.P 2020-2021Qosim PrasetyoNo ratings yet
- Ilovepdf MergedDocument132 pagesIlovepdf Mergedalialqadrie8No ratings yet
- Communication System Modulation TechniquesDocument82 pagesCommunication System Modulation TechniquesragNo ratings yet
- الوحدة 01 الحصة 03Document18 pagesالوحدة 01 الحصة 03Mehdi Houari ZaidNo ratings yet
- Lifesaving SafetyDocument3 pagesLifesaving SafetyЕвгений ШматковNo ratings yet
- Rubavu - Waste Management Plan - WMP-Rev 2.1.docx - 20200827Document70 pagesRubavu - Waste Management Plan - WMP-Rev 2.1.docx - 20200827HoussemNo ratings yet
- Invoice 46F7CFF3 0004Document1 pageInvoice 46F7CFF3 0004pravasunfilteredNo ratings yet
- Procurement: Dana Procurement All Cash Book: IDR - 45,600.00Document2 pagesProcurement: Dana Procurement All Cash Book: IDR - 45,600.00limpbickitNo ratings yet
- Performance Tasks: What Subject Description Due Status QuarterDocument1 pagePerformance Tasks: What Subject Description Due Status QuarterMyka ZoldyckNo ratings yet
- Lompoc FCC Class Action Faustino BernadettDocument168 pagesLompoc FCC Class Action Faustino BernadettDave MinskyNo ratings yet
- Dailysunspot 2Document1 pageDailysunspot 2samer saeedNo ratings yet
- ISO 27001 A.8.1.1 - Inventory of Assets-2020-07-21 18 - 17 - 52.595Document1 pageISO 27001 A.8.1.1 - Inventory of Assets-2020-07-21 18 - 17 - 52.595Diego minerNo ratings yet
- Buku Utang: Status Tanggal Jatuh Tempo Klien Deskripsi SaldoDocument1 pageBuku Utang: Status Tanggal Jatuh Tempo Klien Deskripsi SaldoOva FirmansyahNo ratings yet
- Pemakaian Sarung TanganDocument1 pagePemakaian Sarung TanganAbdulNo ratings yet
- 7 Solid RigidDocument29 pages7 Solid RigidTomás de JorgeNo ratings yet
- Emirates Skycargo Flight Schedule: Flight Number Route Type Origin Destination STD Date STD Sta Date Sta Day of WeekDocument26 pagesEmirates Skycargo Flight Schedule: Flight Number Route Type Origin Destination STD Date STD Sta Date Sta Day of WeekRajNo ratings yet
- Ciat-Lonbarde - MUFF WIGGLERDocument5 pagesCiat-Lonbarde - MUFF WIGGLERMsuttonNo ratings yet
- Pengangkutan Limbah B3 Mei 2022Document2 pagesPengangkutan Limbah B3 Mei 2022Melinda Agustia PratiwiNo ratings yet
- Invoice: Description Quantity Unit Price CostDocument1 pageInvoice: Description Quantity Unit Price CostMessiasestumano ficialNo ratings yet
- Dld-I: App. No App. Type App. Date Duedate Plot Block Scheme Fileno Sr. No Sector S.Dir Date/Time P.Dir RemarksDocument6 pagesDld-I: App. No App. Type App. Date Duedate Plot Block Scheme Fileno Sr. No Sector S.Dir Date/Time P.Dir RemarksQasim AliNo ratings yet
- Linki Do Zajęć On Line WF/ Zdrowy Kręgosłup: WtorekDocument8 pagesLinki Do Zajęć On Line WF/ Zdrowy Kręgosłup: Wtorekschagerlc330No ratings yet
- Patent Application Publication (10) Pub - No .: US 2022/0341698 A1Document17 pagesPatent Application Publication (10) Pub - No .: US 2022/0341698 A1kimba worthNo ratings yet
- DAFTAR OBAT Januari - Juli 2021 Sadewa - 2Document17 pagesDAFTAR OBAT Januari - Juli 2021 Sadewa - 2Julius TambaNo ratings yet
- Infinite Computer Solutions (India) Limited: IT Declaration For The Period April 2022 To March 2023Document1 pageInfinite Computer Solutions (India) Limited: IT Declaration For The Period April 2022 To March 2023VENKATESH NVSNo ratings yet
- Penghitungan HemaDocument26 pagesPenghitungan HemaNatalia putri widati handayaniNo ratings yet
- Name Group Section First Scan First Temp. First Reader/ Form Last Scan Last Temp. Last Reader/ FormDocument14 pagesName Group Section First Scan First Temp. First Reader/ Form Last Scan Last Temp. Last Reader/ FormKu Ahmad FatakhsyaNo ratings yet
- Design Status Summary 30-Jun-20Document1 pageDesign Status Summary 30-Jun-20Pranshu SinglaNo ratings yet
- Sakania Pour Tr8Document18 pagesSakania Pour Tr8Nickson Kyanho officielNo ratings yet
- Invoice # 1Document1 pageInvoice # 1miftakhurrosyidin smpNo ratings yet
- Subcontractors Weekly HSE Statictic Report: Lagging Indicators - Up To DateDocument1 pageSubcontractors Weekly HSE Statictic Report: Lagging Indicators - Up To Dateindra setiawanNo ratings yet
- Winco 29 November 2022Document1 pageWinco 29 November 2022babar azamNo ratings yet
- PTR Trippings From April 2020 To March2021Document4 pagesPTR Trippings From April 2020 To March2021ae.m.nagarkurnool aeNo ratings yet
- Post Mortem EditDocument22 pagesPost Mortem Editnanda lisisinaNo ratings yet
- EE205 Lecture - Section 3Document26 pagesEE205 Lecture - Section 3Atharva GuptaNo ratings yet
- Badac DAPCDocument1 pageBadac DAPCCentro UnoNo ratings yet
- Alfa ResultClinicalGroupDocument1 pageAlfa ResultClinicalGroupayaabouaufNo ratings yet
- Monitoring GambarDocument31 pagesMonitoring GambarPurnomo LintangNo ratings yet
- G-20 (GS) Physical Geo ModuleDocument2 pagesG-20 (GS) Physical Geo ModuleSanjayNo ratings yet
- BAC Resolution A 2020 014 First Failure of Bidding For FOCDocument2 pagesBAC Resolution A 2020 014 First Failure of Bidding For FOCEm FernandezNo ratings yet
- Shall Declare The Bidding A Failure When: XXX C) All Prospective Bidders Are Declared Ineligible "Document2 pagesShall Declare The Bidding A Failure When: XXX C) All Prospective Bidders Are Declared Ineligible "Em FernandezNo ratings yet
- RDInstallmentReport25 10 2020 PDFDocument2 pagesRDInstallmentReport25 10 2020 PDFChiranjeevi GunturuNo ratings yet
- Nolcom Budget Request08182020Document3 pagesNolcom Budget Request08182020Marvin MartinezNo ratings yet
- Invoice: Mol ProductionsDocument2 pagesInvoice: Mol ProductionsEsther EkeomaNo ratings yet
- Physics DiscussionDocument38 pagesPhysics DiscussionMisashi KishimotoNo ratings yet
- Project: Proposed Residential Bungalow House Location: Brgy. # 12 Malasin, Puyupuyan, Pasuquin, Ilocos NorteDocument1 pageProject: Proposed Residential Bungalow House Location: Brgy. # 12 Malasin, Puyupuyan, Pasuquin, Ilocos Nortejay-ar barangayNo ratings yet
- Qop Steelmark 71122Document1 pageQop Steelmark 71122Kawsar AlamNo ratings yet
- 9th 10th Physics Syllabus DivisionDocument6 pages9th 10th Physics Syllabus DivisionMZeeshanAkramNo ratings yet
- Emirates Skycargo Flight Schedule: Flight Number Route Type Origin Destination STD Date STD Sta Date Sta Day of WeekDocument12 pagesEmirates Skycargo Flight Schedule: Flight Number Route Type Origin Destination STD Date STD Sta Date Sta Day of WeekRajNo ratings yet
- Dian Gustina, Skom, Mmsi Teori Bahasa & Otomata (2 SKS) : Foto NIM Nama Tgl. Download Materi File TugasDocument46 pagesDian Gustina, Skom, Mmsi Teori Bahasa & Otomata (2 SKS) : Foto NIM Nama Tgl. Download Materi File Tugaspratiwi susantiNo ratings yet
- Daily DerivativesDocument5 pagesDaily DerivativesRanjan BeheraNo ratings yet
- Market Trends - Materials - L Jacquemin S2 2022Document19 pagesMarket Trends - Materials - L Jacquemin S2 2022LaurentNo ratings yet
- Sample Paper For Class 12 Boards Ip ExamDocument7 pagesSample Paper For Class 12 Boards Ip Examzahir khanNo ratings yet
- Unique Practice Questions - ChemistryDocument53 pagesUnique Practice Questions - Chemistryzahir khanNo ratings yet
- FTS Revised Test Planar Version 6.0Document4 pagesFTS Revised Test Planar Version 6.0zahir khanNo ratings yet
- Final Test Series (Online) For JEE (Main / Advanced) - 2022 Phase - IDocument3 pagesFinal Test Series (Online) For JEE (Main / Advanced) - 2022 Phase - Izahir khanNo ratings yet
- Revision Point - DataframeDocument11 pagesRevision Point - Dataframezahir khanNo ratings yet
- Jee Previous Year Analysis - Graph (2010-2018)Document3 pagesJee Previous Year Analysis - Graph (2010-2018)zahir khanNo ratings yet
- Structure of atom-FINALDocument47 pagesStructure of atom-FINALzahir khanNo ratings yet
- FTS Revised Test Planar Version 6.0Document4 pagesFTS Revised Test Planar Version 6.0zahir khanNo ratings yet
- CE&PPDocument12 pagesCE&PPzahir khanNo ratings yet
- Final Test Series-1Document74 pagesFinal Test Series-1zahir khanNo ratings yet
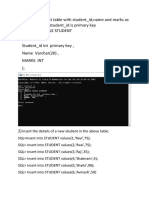
- 1) Create A Student Table With Student - Id, Name and Marks As Attributes Where Student - Id Is Primary Key SQL Create Table StudentDocument5 pages1) Create A Student Table With Student - Id, Name and Marks As Attributes Where Student - Id Is Primary Key SQL Create Table Studentzahir khan100% (1)
- Previous Year JEE - Main Chemistry Analysis-2019Document2 pagesPrevious Year JEE - Main Chemistry Analysis-2019zahir khanNo ratings yet
- Physics: Unique Practice QuestionsDocument77 pagesPhysics: Unique Practice Questionszahir khanNo ratings yet
- Unique Practise Question - MathematicsDocument109 pagesUnique Practise Question - Mathematicszahir khanNo ratings yet
- XII W.S. PPT of INVITATION AND REPLIESDocument20 pagesXII W.S. PPT of INVITATION AND REPLIESzahir khanNo ratings yet
- Revision Point - SeriesDocument5 pagesRevision Point - Serieszahir khanNo ratings yet
- Vehicle Management SystemDocument9 pagesVehicle Management Systemzahir khan100% (1)
- Quick Revision NetworkingDocument7 pagesQuick Revision Networkingzahir khanNo ratings yet
- Chem Group Project ManitDocument13 pagesChem Group Project Manitzahir khanNo ratings yet
- IP Practicals TERM2Document8 pagesIP Practicals TERM2zahir khanNo ratings yet
- Previous Year JEE - Main Analysis (2021) - ChemistryDocument1 pagePrevious Year JEE - Main Analysis (2021) - Chemistryzahir khanNo ratings yet
- MYSQL NewDocument5 pagesMYSQL Newzahir khanNo ratings yet
- Answer Keys: Chemistry Daily Test 21 For - Class - 11 - Engg: You Scored 4 of 4Document7 pagesAnswer Keys: Chemistry Daily Test 21 For - Class - 11 - Engg: You Scored 4 of 4zahir khanNo ratings yet
- PN Junction DiodeDocument6 pagesPN Junction Diodezahir khanNo ratings yet
- Spirit UntamedDocument13 pagesSpirit Untamedzahir khanNo ratings yet
- CHEMISTRY INVESTIGATORY Group PROJECTDocument12 pagesCHEMISTRY INVESTIGATORY Group PROJECTzahir khanNo ratings yet
- Print: You Scored 4 of 4Document5 pagesPrint: You Scored 4 of 4zahir khanNo ratings yet
- PROJECTDocument13 pagesPROJECTzahir khanNo ratings yet
- Oliver TwistDocument11 pagesOliver Twistzahir khanNo ratings yet
- Options:: You Scored 0 of 4Document13 pagesOptions:: You Scored 0 of 4zahir khanNo ratings yet