Professional Documents
Culture Documents
Name of Drug Mechanism of Action Contraindication Side Effects Adverse Effects Nursing Responsibilities
Uploaded by
Michelle Dona Miralles0 ratings0% found this document useful (0 votes)
8 views2 pagesTetanus toxoid vaccine is indicated for booster injections only in people 7 years and older against tetanus. The mechanism of action is that tetanus toxin is transported to the central nervous system where it blocks the release of inhibitory neurotransmitters, leading to muscle spasms. Contraindications include hypersensitivity to any vaccine component or a history of allergic reactions to previous doses. Potential side effects include difficulty breathing, seizures, fever, headache, and swelling at the injection site. Nurses are responsible for assessing patients, monitoring for adverse effects, properly administering the vaccine, and educating patients.
Original Description:
Original Title
a3c3175a8e834f3087cd0779f3975ffe
Copyright
© © All Rights Reserved
Available Formats
DOCX, PDF, TXT or read online from Scribd
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentTetanus toxoid vaccine is indicated for booster injections only in people 7 years and older against tetanus. The mechanism of action is that tetanus toxin is transported to the central nervous system where it blocks the release of inhibitory neurotransmitters, leading to muscle spasms. Contraindications include hypersensitivity to any vaccine component or a history of allergic reactions to previous doses. Potential side effects include difficulty breathing, seizures, fever, headache, and swelling at the injection site. Nurses are responsible for assessing patients, monitoring for adverse effects, properly administering the vaccine, and educating patients.
Copyright:
© All Rights Reserved
Available Formats
Download as DOCX, PDF, TXT or read online from Scribd
0 ratings0% found this document useful (0 votes)
8 views2 pagesName of Drug Mechanism of Action Contraindication Side Effects Adverse Effects Nursing Responsibilities
Uploaded by
Michelle Dona MirallesTetanus toxoid vaccine is indicated for booster injections only in people 7 years and older against tetanus. The mechanism of action is that tetanus toxin is transported to the central nervous system where it blocks the release of inhibitory neurotransmitters, leading to muscle spasms. Contraindications include hypersensitivity to any vaccine component or a history of allergic reactions to previous doses. Potential side effects include difficulty breathing, seizures, fever, headache, and swelling at the injection site. Nurses are responsible for assessing patients, monitoring for adverse effects, properly administering the vaccine, and educating patients.
Copyright:
© All Rights Reserved
Available Formats
Download as DOCX, PDF, TXT or read online from Scribd
You are on page 1of 2
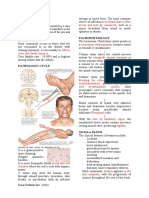
NAME OF DRUG MECHANISM OF CONTRAINDICATION SIDE EFFECTS ADVERSE NURSING
ACTION EFFECTS RESPONSIBILITIES
GENERIC NAME: Tetanus toxin is Hypersensitivity to Difficulty Confusion. Assessment and
Tetanus Toxoid apparently any component of the in breathing Monitoring
transported vaccine, or Convulsion 12 rights in
BRAND NAME: retrogradely to including Thimerosal, swallowing. s (seizures). giving the
Tetanus the CNS where it a mercury derivative, Hives. medication.
exerts its is Fever over
CLASSIFICATION: primary action, a contraindication for Itching, 103 °F Vital signs.
Vaccines, Inactivated, namely, to block further use of this especially (39.4 °C).
Bacterial the release of the vaccine. of feet or
inhibitory amino hands. Headache Intake and
INDICATION: acid transmitters A history Reddening (severe or output.
Tetanus (tetanus gamma- of systemic allergic of skin, continuing).
toxoid) Toxoid is aminobutyric or neurologic especially
indicated for booster acid (GABA) reactions following a around Sleepiness Client Education
injection only for and glycine, previous dose ears. (excessive). Educate the
persons 7 years of age or leading of Tetanus (tetanus patient that
ultimately to a toxoid). Swelling of
older against tetanus Swelling, pain and
spastic form of eyes, face,
(tetanus toxoid). This blistering, tenderness at
paralysis. or inside of
vaccine is NOT or pain at injection site
nose.
indicated A minor afebrile place of may occur.
for primary immunizati Human illness such as a Unusual injection
on. clostridium mild upper tiredness or (severe or Educate the
tetani toxoid respiratory weakness continuing). patient to
DOSAGE/FREQUENCY: immune infection should not (sudden and increase fluid
Parenteral drug products globulin prevent preclude severe). Swelling of intake.
should be inspected visually for s tetanus toxoid immunization. glands in
extraneous particulate matter from damaging armpit.
and/or discoloration prior to tissue and
administration whenever producing the Unusual
solution and container permit. symptoms irritability.
The vaccine should not be used associated with
if particulate matter or tetanus
Vomiting
discoloration is found. (severe or
continuing).
The immune
Primary Immunization for globulin binds to
Persons 7 Years of Age and tetanus toxiod,
Older interfering with
the normal
A series of three doses of interaction of the
0.5 mL each, of Tetanus toxoid with
Toxoid Adsorbed vaccine human tissue.
should be given
intramuscularly
Routine Booster Injections
a booster dose of 0.5 mL
of Td (For Adult Use)
SUBMITTED BY: MENDOZA, ROVIC U. SUBMITTED TO: JOHN EMAR DE GUZMAN
YEAR & BLOCK: UP-FA1-BSN2-09 (Clinical Instrucator)
You might also like
- Share DRUGSTUDYDocument4 pagesShare DRUGSTUDYTyronne JingcoNo ratings yet
- Drug Study Tetanus ToxoidDocument1 pageDrug Study Tetanus ToxoidBunnie Alpha100% (3)
- Cephalexin Drug StudyDocument1 pageCephalexin Drug StudyAubrey SungaNo ratings yet
- PHINMA Education NetworkDocument7 pagesPHINMA Education NetworkMichelle Dona MirallesNo ratings yet
- Urn Uvci 01 Ro M4q6dre9p31zge888lnv705oygk8w2#xDocument2 pagesUrn Uvci 01 Ro M4q6dre9p31zge888lnv705oygk8w2#xFlorian BodnariuNo ratings yet
- Polytechnic College of Davao Del Sur, Inc.: Drug StudyDocument3 pagesPolytechnic College of Davao Del Sur, Inc.: Drug StudyDwight DiazNo ratings yet
- Tetanus Toxoid Body System As A WholeDocument3 pagesTetanus Toxoid Body System As A WholeMaxenia FaboresNo ratings yet
- Drug Name Drug Action Client Indication Contraindications Adverse Effects Nursing Considerations Date OrderedDocument2 pagesDrug Name Drug Action Client Indication Contraindications Adverse Effects Nursing Considerations Date OrderedyehosuaNo ratings yet
- Drug StudyDocument2 pagesDrug StudyZek ComidoyNo ratings yet
- Tetanus Toxoid: CNS: Slight Fever Headache, SeizuresDocument3 pagesTetanus Toxoid: CNS: Slight Fever Headache, Seizuresronan_2905No ratings yet
- TetanustoxoidDocument2 pagesTetanustoxoidShantal AbelloNo ratings yet
- Penicillin G Drug StudyDocument2 pagesPenicillin G Drug StudyRussel Kate SulangNo ratings yet
- Penicillin G Benza Drug StudyDocument2 pagesPenicillin G Benza Drug StudyRussel Kate SulangNo ratings yet
- Case-Scenario-Oxygen Therapy-BuenconsejoDocument6 pagesCase-Scenario-Oxygen Therapy-BuenconsejoCarna BuenconsejoNo ratings yet
- Tetanu S: By: Reno WaisyahDocument18 pagesTetanu S: By: Reno WaisyahReno WaisyahNo ratings yet
- Metronidazole 500mg/tab 1 Tab 3xadayDocument4 pagesMetronidazole 500mg/tab 1 Tab 3xadayCrisyl LipawenNo ratings yet
- Tetanus: Andrew Michael Taylor FRCADocument4 pagesTetanus: Andrew Michael Taylor FRCAILHAM AMIENNo ratings yet
- Micro paraDocument2 pagesMicro parafeminaNo ratings yet
- Tetanus (Harrison's Principle of Internal Medicine)Document4 pagesTetanus (Harrison's Principle of Internal Medicine)Renz Mervin Rivera100% (1)
- DIPTHERIADocument1 pageDIPTHERIAGrace StephanieNo ratings yet
- 4 DS Case StudyDocument4 pages4 DS Case StudyIvan VillapandoNo ratings yet
- Drugstudy 20Document9 pagesDrugstudy 20MahledJoy EnriquezNo ratings yet
- Amikacin Drug StudyDocument2 pagesAmikacin Drug StudyRussel Kate SulangNo ratings yet
- 0.5 ML Repeat at 4-8weeks After First Dose and at 6-12 Months After Second DoseDocument2 pages0.5 ML Repeat at 4-8weeks After First Dose and at 6-12 Months After Second DoseLenneth Ann M. LozadaNo ratings yet
- Group 2 TetanusDocument43 pagesGroup 2 TetanusDaymon, Ma. TeresaNo ratings yet
- Terrorism Information ChartDocument15 pagesTerrorism Information ChartAsad Imran100% (1)
- TDAPDocument1 pageTDAPJEVEE JEAN BETTERNo ratings yet
- Amoxicillin Drug StudyDocument1 pageAmoxicillin Drug StudyAngela CancinoNo ratings yet
- Drug StudyDocument6 pagesDrug StudyJanzelvine Lee MontenegroNo ratings yet
- Complication CL OsceDocument6 pagesComplication CL OsceAdam EdzelNo ratings yet
- Drug Study With NCPDocument4 pagesDrug Study With NCPJoanne Kathleen SantolicesNo ratings yet
- Vaccine (Infant, Child)Document5 pagesVaccine (Infant, Child)Nursing LectureNo ratings yet
- Tetanus: DR - Vemuri ChaitanyaDocument49 pagesTetanus: DR - Vemuri ChaitanyaalifNo ratings yet
- Tetanus ToxoidDocument2 pagesTetanus Toxoidunkown userNo ratings yet
- TetanusDocument4 pagesTetanuseagame gamersNo ratings yet
- Drug StudyDocument3 pagesDrug StudyRainny CommsNo ratings yet
- Patient 1 Drug Study 1Document1 pagePatient 1 Drug Study 1Memcom SolutionNo ratings yet
- Drug Study - CiprofloxacinDocument2 pagesDrug Study - CiprofloxacinryanNo ratings yet
- Cefuroxime Drug StudyDocument1 pageCefuroxime Drug StudyDUMANGENG ELLAINE D.No ratings yet
- Tetanus: F de Assis Aquino Gondim, Faculdade de Medicina Christus, Fortaleza, BrazilDocument2 pagesTetanus: F de Assis Aquino Gondim, Faculdade de Medicina Christus, Fortaleza, BrazilNurzulifa 15 104No ratings yet
- Case Study 4 CVD Infarct CAP MR HUD RDU DementiaDocument21 pagesCase Study 4 CVD Infarct CAP MR HUD RDU DementiaVictoria Mae Irong CabahugNo ratings yet
- Cefu, Metro, KetoDocument4 pagesCefu, Metro, KetoSethlyn_Gomez_5337No ratings yet
- DRUG STUDY (Ceftriaxone)Document2 pagesDRUG STUDY (Ceftriaxone)Avianna CalliopeNo ratings yet
- RMC CiprofloxacinDocument3 pagesRMC Ciprofloxacinshendae cosmianoNo ratings yet
- Drug Study Format and SampleDocument6 pagesDrug Study Format and SampleA.No ratings yet
- TetanusDocument4 pagesTetanusDiana Fadhilah SariNo ratings yet
- Drug StudyyyyDocument3 pagesDrug StudyyyyNathalie kate petallarNo ratings yet
- CefuroximeDocument1 pageCefuroximeAbijah Leris SarmientoNo ratings yet
- Permethrin 5% Cream: Medication MOA Instruction CommentsDocument1 pagePermethrin 5% Cream: Medication MOA Instruction CommentsChron MedNo ratings yet
- Full Details of Pathophysiology of Upper Tract Respiratory DiseaseDocument11 pagesFull Details of Pathophysiology of Upper Tract Respiratory DiseaseSittie Aliah SALAHUDINNo ratings yet
- Clortrimazole Drug StudyDocument3 pagesClortrimazole Drug StudyJasmin NatocNo ratings yet
- Drug StudyDocument10 pagesDrug StudySofia CartallaNo ratings yet
- Drug Study - Penicillin GDocument1 pageDrug Study - Penicillin GAngela CancinoNo ratings yet
- Poison TableDocument12 pagesPoison TableKirk NeneriaNo ratings yet
- Arellano, Ma. Frances Therese D. BSN Ii-A Drug/S Generic/Brand Name Uses Adverse Effect Drug Interaction Nursing ResponsibilitiesDocument6 pagesArellano, Ma. Frances Therese D. BSN Ii-A Drug/S Generic/Brand Name Uses Adverse Effect Drug Interaction Nursing ResponsibilitiesTherese ArellanoNo ratings yet
- Emergency DrugsDocument11 pagesEmergency DrugsJade Hemmings100% (1)
- Lunar Drug Study Optha Last RotationDocument4 pagesLunar Drug Study Optha Last RotationGlizzel Jade GumadeNo ratings yet
- MCN2 WK13Document7 pagesMCN2 WK13Cecilia Micole M. De GulaNo ratings yet
- Tetanus and Tetanus Prone WoundsDocument5 pagesTetanus and Tetanus Prone WoundsANKITA RCHAWLANo ratings yet
- CefuroximeDocument2 pagesCefuroximeJon Corpuz AggasidNo ratings yet
- Labs and Meds Ch19Document2 pagesLabs and Meds Ch19USC Upstate Nursing CoachesNo ratings yet
- Toxic Epidermal Necrolysis, A Simple Guide To The Condition, Diagnosis, Treatment And Related ConditionsFrom EverandToxic Epidermal Necrolysis, A Simple Guide To The Condition, Diagnosis, Treatment And Related ConditionsNo ratings yet
- Fluid and Electrolytes BalanceDocument6 pagesFluid and Electrolytes BalanceMichelle Dona MirallesNo ratings yet
- 9 Hypertension UpdatedDocument23 pages9 Hypertension UpdatedMichelle Dona MirallesNo ratings yet
- Session 13 Psychiatric Emergencies - Rape, ChildDocument1 pageSession 13 Psychiatric Emergencies - Rape, ChildMichelle Dona MirallesNo ratings yet
- PHINMA Education NetworkDocument3 pagesPHINMA Education NetworkMichelle Dona MirallesNo ratings yet
- Rev Enrol ProcDocument2 pagesRev Enrol ProcMichelle Dona MirallesNo ratings yet
- Terazosin Drug Study Group 4Document3 pagesTerazosin Drug Study Group 4Michelle Dona MirallesNo ratings yet
- Drug Study For CefuroximeDocument4 pagesDrug Study For CefuroximeMichelle Dona MirallesNo ratings yet
- Term 2 UtsDocument5 pagesTerm 2 UtsMichelle Dona MirallesNo ratings yet
- Assessment Diagnosis Inference Planning Intervention Rationale EvaluationDocument2 pagesAssessment Diagnosis Inference Planning Intervention Rationale EvaluationMichelle Dona MirallesNo ratings yet
- Biochem Term 2Document28 pagesBiochem Term 2Michelle Dona MirallesNo ratings yet
- Stone Age), This Era Is Marked by The Use of Tools by Our Early Human Ancestors (Who Evolved Around 300,000 B.C.)Document7 pagesStone Age), This Era Is Marked by The Use of Tools by Our Early Human Ancestors (Who Evolved Around 300,000 B.C.)Michelle Dona MirallesNo ratings yet
- Uts Reviewer FinalDocument21 pagesUts Reviewer FinalMichelle Dona MirallesNo ratings yet
- Gen005 Sas 1 6Document15 pagesGen005 Sas 1 6Michelle Dona MirallesNo ratings yet
- CDC Streamlines COVID-19 GuidanceDocument9 pagesCDC Streamlines COVID-19 GuidanceJessica A. BotelhoNo ratings yet
- Tetanus: Dr. Zubaida AimalDocument31 pagesTetanus: Dr. Zubaida AimalkhanmoinNo ratings yet
- Universal Immunization ProgrammeDocument8 pagesUniversal Immunization ProgrammeNagaraj ReddyNo ratings yet
- Urn Uvci 01 Ro O0el5r27d48j5197y9nx3vwpgq96km#bDocument2 pagesUrn Uvci 01 Ro O0el5r27d48j5197y9nx3vwpgq96km#bgrigore sinteanuNo ratings yet
- Vaccine Card - 20231120 - 175739 - 0000Document2 pagesVaccine Card - 20231120 - 175739 - 0000MarilynNo ratings yet
- Health at A Glance Mexico ENDocument2 pagesHealth at A Glance Mexico ENluis sanchezNo ratings yet
- EY Techathon 2021: GamificationDocument2 pagesEY Techathon 2021: GamificationHARIHARAN V PRK19ISD020No ratings yet
- Maklumat Vaksinasi: Vaccination DetailsDocument2 pagesMaklumat Vaksinasi: Vaccination DetailsMohd Faizal Bin Omar HathabNo ratings yet
- Uji Efek Antipiretik Ekstrak Etanol Daun Bayam MERAH (Amaranthus Tricolor L.) PADA TIKUS PUTIH (Rattus Norvegicus) JANTAN GALUR WISTARDocument9 pagesUji Efek Antipiretik Ekstrak Etanol Daun Bayam MERAH (Amaranthus Tricolor L.) PADA TIKUS PUTIH (Rattus Norvegicus) JANTAN GALUR WISTARWIDIA PUSPITA FAJRINo ratings yet
- Listening Skills Practice: How False Information Spreads - ExercisesDocument2 pagesListening Skills Practice: How False Information Spreads - ExercisesLeo MerlinoNo ratings yet
- Second Covid-19 Vaccine Boosters, ExplainedDocument10 pagesSecond Covid-19 Vaccine Boosters, ExplainedsiesmannNo ratings yet
- Passenger Locator Form: Non-Red ListDocument4 pagesPassenger Locator Form: Non-Red Listbriga456No ratings yet
- 1 Model Analyze Driversofvaccineconfidence 22 March PDFDocument5 pages1 Model Analyze Driversofvaccineconfidence 22 March PDFAsep KusnaliNo ratings yet
- Immunization Outreach Form 2023Document1 pageImmunization Outreach Form 2023Ignatius KashumeNo ratings yet
- Homologous or Heterologous Booster of Inactivated Vaccine Reduces Sars-Cov-2 Omicron Variant Escape From Neutralizing AntibodiesDocument12 pagesHomologous or Heterologous Booster of Inactivated Vaccine Reduces Sars-Cov-2 Omicron Variant Escape From Neutralizing AntibodiesSariSyahruniNo ratings yet
- DR Mercola - Thousands of Deaths and Adverse Reactions Deleted From VAERS (20220521)Document9 pagesDR Mercola - Thousands of Deaths and Adverse Reactions Deleted From VAERS (20220521)guy777No ratings yet
- Collective Evolution 1Document11 pagesCollective Evolution 1Timefor WaspsNo ratings yet
- Truechemie: Widal (Antigen Agglutination) Test KitDocument1 pageTruechemie: Widal (Antigen Agglutination) Test Kitfitriadin damayantiNo ratings yet
- Who Afro Draft Guidelines For Integrating Child Survival Interventions Into Routine ImmunizationDocument20 pagesWho Afro Draft Guidelines For Integrating Child Survival Interventions Into Routine ImmunizationtpartapNo ratings yet
- CertificateDocument1 pageCertificatemahesh yadavNo ratings yet
- 2021-11-04 - Affidavit No 1 of Kip Warner (Injection Passport)Document162 pages2021-11-04 - Affidavit No 1 of Kip Warner (Injection Passport)Canadian Society for the Advancement of Science in Public Policy100% (3)
- What Else? 2 SentencesDocument3 pagesWhat Else? 2 SentencesMarvin Medem LaquidanNo ratings yet
- An Analytical Essay On Sinovac VaccineDocument3 pagesAn Analytical Essay On Sinovac VaccineShanya Lameyna VillanuevaNo ratings yet
- COVID-19 Vaccines - Monovalent Vs Bivalent: DOH 825-082 December 2022Document1 pageCOVID-19 Vaccines - Monovalent Vs Bivalent: DOH 825-082 December 2022Eunike KaramoyNo ratings yet
- EUA Fact Sheet For Recipients - 12 Yoa and Older - Pfizer-BioNTech COVID-19 Vaccine - 1.3.2022 FinalDocument9 pagesEUA Fact Sheet For Recipients - 12 Yoa and Older - Pfizer-BioNTech COVID-19 Vaccine - 1.3.2022 FinalMaurA DowlingNo ratings yet
- Ashotinthearmora Hard Pill To Swallow?: Comparing Vaccine FormulationsDocument3 pagesAshotinthearmora Hard Pill To Swallow?: Comparing Vaccine FormulationsKaren SinorNo ratings yet
- Hamlet Act 3 Vocabulary Sheet PDFDocument2 pagesHamlet Act 3 Vocabulary Sheet PDFLuisNo ratings yet
- Drug Study Hepa BDocument3 pagesDrug Study Hepa BKwebblekop JordiNo ratings yet
- Pasteur's Experiment: FermentationDocument2 pagesPasteur's Experiment: FermentationNikki RomeroNo ratings yet