Professional Documents
Culture Documents
Endocrine ' Endocrine ' Section Iii: Endocrine-Physiology Endocrine-Physiology
Uploaded by
Luis Jose VelazquezOriginal Title
Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
Endocrine ' Endocrine ' Section Iii: Endocrine-Physiology Endocrine-Physiology
Uploaded by
Luis Jose VelazquezCopyright:
Available Formats
340 SEC TION III Endocrine
endocrine—physiology
Cortisol
SOURCE Adrenal zona fasciculata. Bound to corticosteroid-binding globulin.
FUNCTION Appetite Cortisol is A BIG FIB.
Blood pressure: Exogenous glucocorticoids can cause
Upregulates α1-receptors on arterioles reactivation of TB and candidiasis (blocks IL-2
sensitivity to norepinephrine and production).
epinephrine (permissive action)
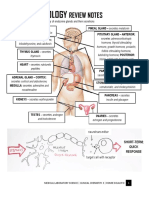
Stress Hypothalamus
At high concentrations, can bind to Circadian rhythm
mineralocorticoid (aldosterone) receptors
CRH
Insulin resistance (diabetogenic)
Gluconeogenesis, lipolysis, and proteolysis
( glucose utilization)
Fibroblast activity (poor wound healing, Anterior
collagen synthesis, striae) pituitary
Inflammatory and Immune responses: Endorphins
MSH
Inhibits production of leukotrienes and
Proopiomelanocortin ACTH
prostaglandins
Inhibits WBC adhesion neutrophilia
Blocks histamine release from mast cells
Cortisol
Eosinopenia, lymphopenia
Blocks IL-2 production
Bone formation ( osteoblast activity)
Downstream cortisol
function
REGULATION CRH (hypothalamus) stimulates ACTH release Chronic stress may induce prolonged cortisol
(pituitary) cortisol production in adrenal secretion, cortisol resistance, impaired
zona fasciculata. Excess cortisol CRH, immunocompetency, and dysregulation of
ACTH, and cortisol secretion. HPA axis.
Appetite regulation
Ghrelin Stimulates hunger (orexigenic effect) and GH release (via GH secretagog receptor). Produced by
stomach. Sleep deprivation, fasting, or Prader-Willi syndrome ghrelin production.
Ghrelin makes you ghrow hunghry. Acts on lateral area of hypothalamus (hunger center) to
appetite.
Leptin Satiety hormone. Produced by adipose tissue. Mutation of leptin gene severe obesity. Obese
people have leptin due to adipose tissue but are tolerant or resistant to leptin’s anorexigenic
effect. Sleep deprivation or starvation leptin production.
Leptin keeps you thin. Acts on ventromedial area of hypothalamus (satiety center) to appetite.
Endocannabinoids Act at cannabinoid receptors in hypothalamus and nucleus accumbens, two key brain areas for the
homeostatic and hedonic control of food intake appetite.
Exogenous cannabinoids cause “the munchies.”
FAS1_2023_08-Endocrine.indd 340 11/17/22 8:57 PM
Endocrine
endocrine—physiology SEC TION III 341
Signaling pathways of endocrine hormones
cAMP FSH, LH, ACTH, TSH, CRH, hCG, ADH (V2- FLAT ChAMPs CHuGG
receptor), MSH, PTH, Calcitonin, Histamine
(H2-receptor), Glucagon, GHRH
cGMP BNP, ANP, EDRF (NO) BAD GraMPa
Think vasodilation and diuresis
IP3 GnRH, Oxytocin, ADH (V1-receptor), TRH, GOAT HAG
Histamine (H1-receptor), Angiotensin II,
Gastrin
Intracellular receptor Progesterone, Estrogen, Testosterone, Cortisol, PET CAT in TV
Aldosterone, T3/T4, Vitamin D
Receptor tyrosine IGF-1, FGF, PDGF, EGF, Insulin MAP kinase pathway
kinase Get Found In the MAP
Nonreceptor tyrosine G-CSF, Erythropoietin, Thrombopoietin JAK/STAT pathway
kinase Prolactin, Immunomodulators (eg, cytokines Think acidophils and cytokines
IL-2, IL-6, IFN), GH GET a JAKed PIG
Signaling pathways of steroid hormones
Binding to receptor Steroid hormones are lipophilic and therefore
located in nucleus
or in cytoplasm
H Hormone must circulate bound to specific binding
globulins, which their solubility.
In males, sex hormone–binding
Transformation of R Receptor
receptor to expose globulin (SHBG) lowers free testosterone
DNA-binding protein gynecomastia.
In females, SHBG raises free testosterone
Binding to hirsutism.
enhancer-like H Cytoplasm
element in DNA R
Gene
estrogen (eg, OCPs, pregnancy) SHBG.
Intron Exon
Pre-mRNA
mRNA Nucleus
Protein
Ribosome
Response
FAS1_2023_08-Endocrine.indd 341 11/17/22 8:57 PM
342 SEC TION III Endocrine
endocrine—Pathology
ENDOCRINE—PATHOLOGY
`
Syndrome of inappropriate Characterized by excessive free water retention,
antidiuretic hormone secretion euvolemic hyponatremia with continued
urinary Na+ excretion, urine osmolality >
serum osmolality.
serum serum osmolality
osmolality Hypothalamus
serum volume
Body responds to water retention with
aldosterone and ANP and BNP urinary
Na+ secretion normalization of extracellular
urine osmolality
Central DI ADH
urine volume fluid volume euvolemic hyponatremia.
Treatment: fluid restriction (first line), salt
SIADH
tablets, IV hypertonic saline, diuretics,
Posterior Medullary collecting duct
Aquaporin channels
ADH antagonists (eg, conivaptan, tolvaptan,
pituitary
ADH
(storage) demeclocycline).
ADH antagonists
SIADH causes include (HEELD-up water):
Lithium Head trauma/CNS disorders
Nephrogenic DI
Ectopic ADH (eg, small cell lung cancer)
Exogenous hormones (eg, vasopressin,
desmopressin, oxytocin)
Lung disease
Drugs (eg, SSRIs, carbamazepine,
cyclophosphamide)
Primary polydipsia and Characterized by the production of large amounts of dilute urine +/– thirst. Urine specific gravity
diabetes insipidus < 1.006. Urine osmolality usually < 300 mOsm/kg. Central DI may be transient if damage is
below hypothalamic median eminence or in the posterior pituitary (ADH in hypothalamus can
still be secreted systemically via portal capillaries in median eminence).
Primary polydipsia Central DI Nephrogenic DI
DEFINITION Excessive water intake ADH release ADH resistance
CAUSES Psychiatric illnesses, Idiopathic, brain injury Hereditary (ADH receptor
hypothalamic lesions (trauma, hypoxia, tumor, mutation), drugs (eg,
affecting thirst center surgery, infiltrative diseases) lithium, demeclocycline),
hypercalcemia, hypokalemia
SERUM OSMOLALITY
ADH LEVEL or normal Normal or
WATER RESTRICTION a
Significant in urine No change or slight in urine No change or slight in urine
osmolality (> 700 mOsm/kg) osmolality osmolality
DESMOPRESSIN ADMINISTRATIONb — Significant in urine Minimal change in urine
osmolality (> 50%) osmolality
TREATMENT Water restriction Desmopressin (DDAVP) Manage the underlying cause;
low-solute diet, HCTZ,
amiloride, indomethacin
a
No water intake for 2–3 hours followed by hourly measurements of urine volume and osmolality as well as plasma Na+
concentration and osmolality.
b
Desmopressin (ADH analog) is administered if serum osmolality > 295–300 mOsm/kg, plasma Na+ ≥ 145 mEq/L, or urine
osmolality does not increase despite plasma osmolality.
FAS1_2023_08-Endocrine.indd 342 11/17/22 8:57 PM
Endocrine
endocrine—Pathology SEC TION III 343
Hypopituitarism Undersecretion of pituitary hormones due to
Nonsecreting pituitary adenoma, craniopharyngioma
Sheehan syndrome—ischemic infarct of pituitary following severe postpartum hemorrhage;
pregnancy-induced pituitary growth susceptibility to hypoperfusion. Usually presents with
failure to lactate, amenorrhea, cold intolerance (anterior pituitary hormones mainly affected).
Empty sella syndrome—atrophy or compression of pituitary (which lies in the sella turcica),
often idiopathic, common in obese females; associated with idiopathic intracranial hypertension
Pituitary apoplexy—sudden hemorrhage of pituitary gland, often in the presence of an existing
pituitary adenoma. Usually presents with sudden onset severe headache, visual impairment (eg,
bitemporal hemianopia, diplopia due to CN III palsy), and features of hypopituitarism
Brain injury
Radiation
Treatment: hormone replacement therapy (glucocorticoids, thyroxine, sex steroids, human growth
hormone)
Acromegaly Excess GH in adults. Typically caused by pituitary adenoma.
FINDINGS Large tongue with deep furrows, frontal GH in children gigantism ( linear bone
bossing, coarsening of facial features with growth due to unfused epiphysis).
aging A , deep voice, diaphoresis (excessive
A
sweating), hypertrophic arthropathy, impaired
glucose tolerance (insulin resistance), HTN,
LVH, HFpEF (most common cause of death).
DIAGNOSIS serum IGF-1; failure to suppress serum GH
following oral glucose tolerance test; pituitary
mass seen on brain MRI.
TREATMENT Pituitary adenoma resection. If not cured, Baseline
treat with octreotide (somatostatin analog),
pegvisomant (GH receptor antagonist), or
dopamine agonists (eg, cabergoline).
FAS1_2023_08-Endocrine.indd 343 11/17/22 8:57 PM
344 SEC TION III Endocrine
endocrine—Pathology
Hypothyroidism vs hyperthyroidism
Hypothyroidism Hyperthyroidism
METABOLIC Cold intolerance, sweating, weight gain Heat intolerance, sweating, weight loss
( basal metabolic rate calorigenesis), ( synthesis of Na+/K+-ATPase basal
hyponatremia ( free water clearance) metabolic rate calorigenesis)
SKIN/HAIR Dry, cool skin (due to blood flow); coarse, Warm, moist skin (due to vasodilation); fine hair;
brittle hair; diffuse alopecia; brittle nails; onycholysis ( A ); pretibial myxedema in Graves
puffy facies and generalized nonpitting edema disease B
(myxedema) due to GAGs in interstitial
spaces osmotic pressure water retention
OCULAR Periorbital edema C Ophthalmopathy in Graves disease (including
periorbital edema, exophthalmos), lid lag/
retraction ( sympathetic stimulation of
superior tarsal muscle)
GASTROINTESTINAL Constipation ( GI motility), appetite Hyperdefecation/diarrhea ( GI motility),
appetite
MUSCULOSKELETAL Hypothyroid myopathy (proximal weakness, Thyrotoxic myopathy (proximal weakness,
CK), carpal tunnel syndrome, myoedema normal CK), osteoporosis/ fracture rate (T3
(small lump rising on the surface of a muscle directly stimulates bone resorption)
when struck with a hammer)
REPRODUCTIVE Abnormal uterine bleeding, libido, infertility Abnormal uterine bleeding, gynecomastia,
libido, infertility
NEUROPSYCHIATRIC Hypoactivity, lethargy, fatigue, weakness, Hyperactivity, restlessness, anxiety, insomnia,
depressed mood, reflexes (delayed/slow fine tremors (due to β-adrenergic activity),
relaxing) reflexes (brisk)
CARDIOVASCULAR Bradycardia, dyspnea on exertion ( cardiac Tachycardia, palpitations, dyspnea, arrhythmias
output) (eg, atrial fibrillation), chest pain and systolic
HTN due to number and sensitivity of
β-adrenergic receptors, expression of cardiac
sarcolemmal ATPase and expression of
phospholamban
LABS TSH (if 1°) TSH (if 1°)
free T3 and T4 free T3 and T4
Hypercholesterolemia (due to LDL receptor LDL, HDL, and total cholesterol
expression)
A B C
FAS1_2023_08-Endocrine.indd 344 11/17/22 8:57 PM
Endocrine
endocrine—Pathology SEC TION III 345
Hypothyroidism
Hashimoto thyroiditis Also called chronic autoimmune thyroiditis. Most common cause of hypothyroidism in iodine-
sufficient regions. Associated with HLA-DR3 (differs by ethnicity), risk of primary thyroid
lymphoma (typically diffuse large B-cell lymphoma).
Findings: moderately enlarged, nontender thyroid. May be preceded by transient hyperthyroid
state (“Hashitoxicosis”) due to follicular rupture and thyroid hormone release.
Serology: ⊕ antithyroid peroxidase (antimicrosomal) and antithyroglobulin antibodies.
Histology: Hürthle cells A , lymphoid aggregates with germinal centers B .
Postpartum thyroiditis—mild, self-limited variant of Hashimoto thyroiditis arising < 1 year after
delivery.
Subacute Also called de Quervain thyroiditis. Usually, a self-limited disease. Natural history: transient
granulomatous hyperthyroidism euthyroid state hypothyroidism euthyroid state. Often preceded by viral
thyroiditis infection.
Findings: ESR, jaw pain, very tender thyroid (de Quervain is associated with pain).
Histology: granulomatous inflammation C .
Riedel thyroiditis Also called invasive fibrous thyroiditis. May occur as part of IgG4-related disease spectrum (eg,
autoimmune pancreatitis, retroperitoneal fibrosis, noninfectious aortitis). Hypothyroidism occurs
in 1⁄3 of patients. Fibrosis may extend to local structures (eg, trachea, esophagus), mimicking
anaplastic carcinoma.
Findings: slowly enlarging, hard (rocklike), fixed, nontender thyroid.
Histology: thyroid replaced by fibrous tissue and inflammatory infiltrate D .
Congenital Formerly called cretinism. Most commonly caused by thyroid dysgenesis (abnormal thyroid gland
hypothyroidism development; eg, agenesis, hypoplasia, ectopy) or dyshormonogenesis (abnormal thyroid hormone
synthesis; eg, mutations in thyroid peroxidase) in iodine-sufficient regions.
Findings (6 P’s): pot-bellied, pale, puffy-faced child E with protruding umbilicus, protuberant
tongue F , and poor brain development.
Other causes Iodine deficiency (most common cause worldwide; typically presents with goiter G ), iodine excess
(Wolff-Chaikoff effect), drugs (eg, amiodarone, lithium), nonthyroidal illness syndrome (also
called euthyroid sick syndrome; T3 with normal/ T4 and TSH in critically ill patients).
A B C D
E F G
Before treatment After treatment
FAS1_2023_08-Endocrine.indd 345 11/17/22 8:57 PM
346 SEC TION III Endocrine
endocrine—Pathology
Hyperthyroidism
Graves disease Most common cause of hyperthyroidism. Thyroid-stimulating immunoglobulin (IgG, can cause
A
transient neonatal hyperthyroidism; type II hypersensitivity) stimulates TSH receptors on thyroid
(hyperthyroidism, diffuse goiter), dermal fibroblasts (pretibial myxedema), and orbital fibroblasts
(Graves orbitopathy). Activation of T-cells lymphocytic infiltration of retroorbital space
cytokines (eg, TNF-α, IFN-γ) fibroblast secretion of hydrophilic GAGs osmotic
muscle swelling, muscle inflammation, and adipocyte count exophthalmos A . Often presents
during stress (eg, pregnancy). Associated with HLA-DR3 and HLA-B8.
Histology: tall, crowded follicular epithelial cells; scalloped colloid.
Toxic multinodular Focal patches of hyperfunctioning follicular cells distended with colloid working independently
goiter of TSH (due to TSH receptor mutations in 60% of cases). release of T3 and T4. Hot nodules
(hyperfunctioning nodules visualized on radioactive iodine scan) are rarely malignant.
Thyroid storm Uncommon but serious complication that occurs when hyperthyroidism is incompletely treated/
untreated and then significantly worsens in the setting of acute stress such as infection, trauma,
surgery. Presents with agitation, delirium, fever, diarrhea, coma, and tachyarrhythmia (cause
of death). May see LFTs. Treat with the 4 P’s: β-blockers (eg, propranolol), propylthiouracil,
glucocorticoids (eg, prednisolone), potassium iodide (Lugol iodine). Iodide load T4 synthesis
Wolff-Chaikoff effect.
Jod-Basedow Iodine-induced hyperthyroidism. Occurs when a patient with iodine deficiency and partially
phenomenon autonomous thyroid tissue (eg, autonomous nodule) is made iodine replete. Can happen after iodine
IV contrast or amiodarone use. Opposite to Wolff-Chaikoff effect.
Causes of goiter Smooth/diffuse: Graves disease, Hashimoto thyroiditis, iodine deficiency, TSH-secreting pituitary
adenoma.
Nodular: toxic multinodular goiter, thyroid adenoma, thyroid cancer, thyroid cyst.
Thyroid adenoma Benign solitary growth of the thyroid. Most are nonfunctional (“cold” on radioactive iodine scan),
A
can rarely cause hyperthyroidism via autonomous thyroid hormone production (“hot” or “toxic”).
Most common histology is follicular (arrows in A ); absence of capsular or vascular invasion
(unlike follicular carcinoma).
FAS1_2023_08-Endocrine.indd 346 11/17/22 8:59 PM
Endocrine
endocrine—Pathology SEC TION III 347
Thyroid cancer Typically diagnosed with fine needle aspiration; treated with thyroidectomy. Complications of
surgery include hypocalcemia (due to removal of parathyroid glands), transection of recurrent
laryngeal nerve during ligation of inferior thyroid artery (leads to dysphagia and dysphonia
[hoarseness]), and injury to the external branch of the superior laryngeal nerve during ligation of
superior thyroid vascular pedicle (may lead to loss of tenor usually noticeable in professional voice
users).
Papillary carcinoma Most common. Empty-appearing nuclei with central clearing (“Orphan Annie” eyes) A ,
A
psamMoma bodies, nuclear grooves (Papi and Moma adopted Orphan Annie). risk with RET/
PTC rearrangements and BRAF mutations, childhood irradiation.
Papillary carcinoma: most prevalent, palpable lymph nodes. Good prognosis.
Follicular carcinoma Good prognosis. Invades thyroid capsule and vasculature (unlike follicular adenoma), uniform
follicles; hematogenous spread is common. Associated with RAS mutation and PAX8-PPAR-γ
translocations. Fine needle aspiration cytology may not be able to distinguish between follicular
adenoma and carcinoma.
Medullary carcinoma From parafollicular “C cells”; produces calcitonin, sheets of polygonal cells in an amyloid stroma
B (stains with Congo red). Associated with MEN 2A and 2B (RET mutations).
B
Undifferentiated/ Older patients; presents with rapidly enlarging neck mass compressive symptoms (eg, dyspnea,
anaplastic carcinoma dysphagia, hoarseness); very poor prognosis. Associated with TP53 mutation.
FAS1_2023_08-Endocrine.indd 347 11/17/22 8:59 PM
348 SEC TION III Endocrine
endocrine—Pathology
Diagnosing
parathyroid disease
250
3° hyperparathyroidism
2° hyperparathyroidism (chronic kidney disease)
(vitamin D deficiency, ↓ Ca2+ intake,
chronic kidney disease)
50 1° hyperparathyroidism
PTH (pg/mL)
(hyperplasia, adenoma,
carcinoma)
Normal
10
Hypoparathyroidism PTH-independent
(surgical resection, hypercalcemia
autoimmune) (excess Ca2+ intake, cancer,
↑ vitamin D)
2
4 6 8 10 12 14 16 18 20
2+
Ca (mg/dL)
Hypoparathyroidism Due to injury to parathyroid glands or their blood supply (usually during thyroid surgery),
A autoimmune destruction, or DiGeorge syndrome. Findings: tetany, hypocalcemia,
hyperphosphatemia.
Chvostek sign—tapping of facial nerve (tap the Cheek) contraction of facial muscles.
Trousseau sign—occlusion of brachial artery with BP cuff (cuff the Triceps) carpal spasm.
Pseudohypoparathyroidism type 1A—autosomal dominant, maternally transmitted mutations
(imprinted GNAS gene). GNAS1-inactivating mutation (coupled to PTH receptor) that encodes
the Gs protein α subunit inactivation of adenylate cyclase when PTH binds to its receptor
end-organ resistance (kidney and bone) to PTH.
Physical findings: Albright hereditary osteodystrophy (shortened 4th/5th digits A , short stature,
round face, subcutaneous calcifications, developmental delay).
Labs: PTH, Ca2+, PO43–.
Pseudopseudohypoparathyroidism—autosomal dominant, paternally transmitted mutations
(imprinted GNAS gene) but without end-organ resistance to PTH due to normal maternal allele
maintaining renal responsiveness to PTH.
Physical findings: same as Albright hereditary osteodystrophy.
Labs: normal PTH, Ca2+, PO43–.
Lab values in hypocalcemic disorders
DISORDER Ca2+ PO 43– PTH ALP 25(OH) VITAMIN D 1,25(OH)2 VITAMIN D
Vitamin D deficiency —/ —/ —/
2° hyperpara- —
thyroidism (CKD)
Hypoparathyroidism — — —/
Pseudohypo- — —/
parathyroidism
FAS1_2023_08-Endocrine.indd 348 11/17/22 9:00 PM
Endocrine
endocrine—Pathology SEC TION III 349
Hyperparathyroidism
Primary Usually due to parathyroid adenoma or Osteitis fibrosa cystica—cystic bone spaces
hyperparathyroidism hyperplasia. Hypercalcemia, hypercalciuria filled with brown fibrous tissue A (“brown
A
(renal stones), polyuria (thrones), tumor” consisting of osteoclasts and deposited
hypophosphatemia, PTH, ALP, urinary hemosiderin from hemorrhages; causes
cAMP. Most often asymptomatic. May present bone pain). Due to PTH, classically
with bone pain, weakness, constipation associated with 1° (but also seen with 2°)
(“groans”), abdominal/flank pain (kidney hyperparathyroidism.
stones, acute pancreatitis), neuropsychiatric “Stones, thrones, bones, groans, and
disturbances (“psychiatric overtones”). psychiatric overtones.”
Secondary 2° hyperplasia due to Ca2+ absorption Renal osteodystrophy—renal disease 2° and
hyperparathyroidism and/or PO43−, most often in chronic 3° hyperparathyroidism bone lesions.
kidney disease (causes hypovitaminosis D
and hyperphosphatemia Ca2+).
Hypocalcemia, hyperphosphatemia in
chronic kidney disease (vs hypophosphatemia
with most other causes), ALP, PTH.
Tertiary Refractory (autonomous) hyperparathyroidism
hyperparathyroidism resulting from chronic kidney disease.
PTH, Ca2+.
Familial hypocalciuric Autosomal dominant. Defective G-coupled Ca2+-sensing receptors in multiple tissues (eg,
hypercalcemia parathyroids, kidneys). Higher than normal Ca2+ levels required to suppress PTH. Excessive renal
Ca2+ reabsorption mild hypercalcemia and hypocalciuria with normal to PTH levels.
FAS1_2023_08-Endocrine.indd 349 11/17/22 9:00 PM
350 SEC TION III Endocrine
endocrine—Pathology
Diabetes mellitus
ACUTE MANIFESTATIONS Polydipsia, polyuria, polyphagia (3 P’s), weight loss, DKA (type 1), hyperosmolar hyperglycemic
state (type 2).
Rarely, can be caused by unopposed secretion of GH and epinephrine. Also seen in patients on
glucocorticoid therapy (steroid diabetes).
CHRONIC COMPLICATIONS Nonenzymatic glycation:
Small vessel disease (hyaline arteriolosclerosis) retinopathy, neuropathy, nephropathy.
Large vessel disease (atherosclerosis) CAD, cerebrovascular disease, peripheral vascular
disease. MI is the most common cause of death.
Osmotic damage (sorbitol accumulation in organs with aldose reductase and or absent sorbitol
dehydrogenase):
Neuropathy: motor, sensory (glove and stocking distribution), autonomic degeneration (eg,
GERD, gastroparesis, diabetic diarrhea).
Cataracts.
DIAGNOSIS TEST DIAGNOSTIC CUTOFF NOTES
HbA1c ≥ 6.5% Reflects average blood glucose
over prior 3 months (influenced
by RBC turnover)
Fasting plasma glucose ≥ 126 mg/dL Fasting for > 8 hours
2-hour oral glucose tolerance test ≥ 200 mg/dL 2 hours after consumption of 75 g
of glucose in water
Random plasma glucose ≥ 200 mg/dL Presence of hyperglycemic
symptoms is required
available insulin
lipolysis proteolysis gluconeogenesis glycogenolysis tissue glucose uptake
plasma muscle mass,
free fatty acids weight loss Hypoglycemia,
glycosuria
Osmotic diuresis plasma osmolality
ketogenesis, Loss of water,
Vomiting thirst
ketonemia, ketonuria Na, and K
Anion gap Hyperventilation,
Hypovolemia
metabolic acidosis Kussmaul respiration
Circulation failure,
tissue perfusion
FAS1_2023_08-Endocrine.indd 350 11/17/22 9:00 PM
Endocrine
endocrine—Pathology SEC TION III 351
Type 1 vs type 2 diabetes mellitus
Type 1 Type 2
1° DEFECT Autoimmune T-cell–mediated destruction of resistance to insulin, progressive pancreatic
β cells β-cell failure
INSULIN NECESSARY IN TREATMENT Always Sometimes
AGE (EXCEPTIONS COMMON) < 30 yr > 40 yr
ASSOCIATION WITH OBESITY No Yes
GENETIC PREDISPOSITION Relatively weak (50% concordance in identical Relatively strong (90% concordance in identical
twins), polygenic twins), polygenic
ASSOCIATION WITH HLA SYSTEM Yes, HLA-DR4 and -DR3 (4 – 3 = type 1) No
GLUCOSE INTOLERANCE Severe Mild to moderate
INSULIN SENSITIVITY High Low
KETOACIDOSIS Common Rare
β-CELL NUMBERS IN THE ISLETS Variable (with amyloid deposits)
SERUM INSULIN LEVEL initially, but in advanced disease
CLASSIC SYMPTOMS OF POLYURIA, Common Sometimes
POLYDIPSIA, POLYPHAGIA, WEIGHT
LOSS
HISTOLOGY Islet leukocytic infiltrate Islet amyloid polypeptide deposits
Hyperglycemic emergencies
Diabetic ketoacidosis Hyperosmolar hyperglycemic state
PATHOGENESIS Insulin noncompliance or requirements Profound hyperglycemia excessive osmotic
due to stress (eg, infection) lipolysis and diuresis dehydration and serum osmolality
oxidation of free fatty acids ketone bodies HHS. Classically seen in older patients with
(β-hydroxybutyrate > acetoacetate). type 2 DM and limited ability to drink.
Insulin deficient, ketones present. Insulin present, ketones deficient.
SIGNS/SYMPTOMS DKA is Deadly: Delirium/psychosis, Kussmaul Thirst, polyuria, lethargy, focal neurologic
respirations (rapid, deep breathing), Abdominal deficits, seizures.
pain/nausea/vomiting, Dehydration. Fruity
breath odor due to exhaled acetone.
LABS Hyperglycemia, H+, HCO3 – ( anion gap Hyperglycemia (often > 600 mg/dL), serum
metabolic acidosis), urine and blood ketone osmolality (> 320 mOsm/kg), normal pH (no
levels, leukocytosis. Normal/ serum K+, but acidosis), no ketones. Normal/ serum K+,
depleted intracellular K+ due to transcellular intracellular K+.
shift from insulin and acidosis. Osmotic
diuresis K+ loss in urine total body
K+ depletion.
COMPLICATIONS Life-threatening mucormycosis, cerebral Can progress to coma and death if untreated.
edema, cardiac arrhythmias.
TREATMENT IV fluids, IV insulin, and K+ (to replete intracellular stores). Glucose may be required to prevent
hypoglycemia from insulin therapy.
FAS1_2023_08-Endocrine.indd 351 11/17/22 9:00 PM
352 SEC TION III Endocrine
endocrine—Pathology
Hypoglycemia in Usually occurs in patients treated with insulin or insulin secretagogues (eg, sulfonylureas,
diabetes mellitus meglitinides) in the setting of high-dose treatment, inadequate food intake, and/or exercise.
Neurogenic (autonomic) symptoms: diaphoresis, tachycardia, tremor, anxiety, hunger. Allow
perception of glucose (hypoglycemia awareness).
Neuroglycopenic symptoms: altered mental status, seizures, death due to insufficient glucose in
CNS. May occur in the absence of preceding neurogenic symptoms in patients with attenuated
autonomic response (hypoglycemia unawareness).
Treatment: simple carbohydrates (eg, glucose tablets, fruit juice), IM glucagon, IV dextrose.
Cushing syndrome
ETIOLOGY cortisol due to a variety of causes:
Exogenous glucocorticoids ACTH bilateral adrenal atrophy. Most common cause.
Primary adrenal adenoma, hyperplasia, or carcinoma ACTH atrophy of uninvolved
adrenal gland.
ACTH-secreting pituitary adenoma (Cushing disease); paraneoplastic ACTH secretion (eg,
small cell lung cancer, bronchial carcinoids) bilateral adrenal hyperplasia. Cushing disease is
responsible for the majority of endogenous cases of Cushing syndrome.
FINDINGS MOON FACIES: Metabolic syndrome (hypertension, hyperglycemia, hyperlipidemia), Obesity
(truncal weight gain with wasting of extremities, round “moon” facies A , dorsocervical fat pad
“buffalo hump”), Osteoporosis, Neuropsychiatric (depression, anxiety, irritability), Facial plethora,
Androgen excess (acne, hirsutism), Cataract, Immunosuppression, Ecchymoses (easy bruising),
Skin changes (thinning, striae B , hyperpigmentation).
DIAGNOSIS Screening tests include: free cortisol on 24-hr urinalysis, late night salivary cortisol, and no
suppression with overnight low-dose dexamethasone test.
A 24-hr urine free cortisol, late night salivary
cortisol, and/or inadequate suppression on
1 mg overnight dexamethasone test
Measure serum ACTH
Suppressed Elevated
ACTH-independent ACTH-dependent
Cushing syndrome Cushing syndrome
B
Exogenous glucocorticoids High-dose dexamethasone CRH stimulation test
or adrenal tumor suppression test
No ACTH, cortisol ACTH, cortisol No
Consider adrenal CT Ectopic ACTH Ectopic ACTH
to confirm secretion Cushing disease secretion
MRI of the pituitary
(to identify adenoma)
CT of the chest/abdomen/pelvis
(to identify tumor)
FAS1_2023_08-Endocrine.indd 352 11/17/22 9:00 PM
Endocrine
endocrine—Pathology SEC TION III 353
Nelson syndrome Enlargement of pre-existing ACTH–secreting pituitary adenoma after bilateral adrenalectomy for
refractory Cushing disease ACTH (hyperpigmentation), mass effect (headaches, bitemporal
hemianopia).
Treatment: transsphenoidal resection, postoperative pituitary irradiation for residual tumor.
Adrenal insufficiency Inability of adrenal glands to generate enough glucocorticoids +/− mineralocorticoids for the body’s
needs. Can be acute or chronic. Symptoms include weakness, fatigue, orthostatic hypotension,
muscle aches, weight loss, GI disturbances, sugar and/or salt cravings.
Treatment: glucocorticoid +/− mineralocorticoid replacement.
Primary adrenal gland function cortisol, aldosterone hypotension (hyponatremic volume contraction),
insufficiency hyperkalemia, metabolic acidosis, skin/mucosal hyperpigmentation A ( melanin synthesis due to
A MSH, a byproduct of POMC cleavage). Primary pigments the skin/mucosa.
Addison disease—chronic 1° adrenal insufficiency; caused by adrenal atrophy or destruction. Most
commonly due to autoimmune adrenalitis (high-income countries) or TB (low-income countries).
Secondary and pituitary ACTH secretion (secondary) or hypothalamic CRH secretion (tertiary). No
tertiary adrenal hyperkalemia (aldosterone synthesis preserved due to functioning adrenal gland, intact RAAS), no
insufficiency hyperpigmentation.
2° adrenal insufficiency is due to pituitary pathologies, 3° adrenal insufficiency is most commonly
due to abrupt cessation of chronic glucocorticoid therapy (HPA suppression). Tertiary from
treatment.
Acute adrenal Also called adrenal (addisonian) crisis; often precipitated by acute stressors that glucocorticoid
insufficiency requirements (eg, infection) in patients with pre-existing adrenal insufficiency or on
glucocorticoid therapy. May present with acute abdominal pain, nausea, vomiting, altered mental
status, shock.
Waterhouse-Friderichsen syndrome—bilateral adrenal hemorrhage in the setting of sepsis
(eg, meningococcemia) acute 1° adrenal insufficiency.
Concern for adrenal
insufficiency
Check AM cortisol or
ACTH stimulation
↓ AM cortisol
and/or
↓ peak cortisol on
stimulation test
Measure random
serum ACTH
↓ or inappropriately
normal ACTH ↑ ACTH
2°/3° adrenal 1° adrenal
insufficiency insufficiency
FAS1_2023_08-Endocrine.indd 353 11/17/22 9:00 PM
354 SEC TION III Endocrine
endocrine—Pathology
Hyperaldosteronism Increased secretion of aldosterone from adrenal gland. Clinical features include hypertension,
or normal K+, metabolic alkalosis. 1° hyperaldosteronism does not directly cause edema due
to aldosterone escape mechanism. However, certain 2° causes of hyperaldosteronism (eg, heart
failure) impair the aldosterone escape mechanism, leading to worsening of edema.
Primary Seen in patients with bilateral adrenal hyperplasia or adrenal adenoma (Conn syndrome).
hyperaldosteronism aldosterone, renin. Leads to treatment-resistant hypertension.
Secondary Seen in patients with renovascular hypertension, juxtaglomerular cell tumors (renin-producing),
hyperaldosteronism and edema (eg, cirrhosis, heart failure, nephrotic syndrome).
Neuroendocrine Heterogeneous group of neoplasms originating from neuroendocrine cells (which have traits similar
tumors to nerve cells and hormone-producing cells).
Most neoplasms occur in the GI system (eg, carcinoid, gastrinoma), pancreas (eg, insulinoma,
glucagonoma), and lungs (eg, small cell carcinoma). Also in thyroid (eg, medullary carcinoma)
and adrenals (eg, pheochromocytoma).
Neuroendocrine cells (eg, pancreatic β cells, enterochromaffin cells) share a common biologic
function through amine precursor uptake decarboxylase (APUD) despite differences in
embryologic origin, anatomic site, and secretory products (eg, chromogranin A, neuron-specific
enolase [NSE], synaptophysin, serotonin, histamine, calcitonin). Treatment: surgical resection,
somatostatin analogs.
Neuroblastoma Most common tumor of the adrenal medulla in children, usually < 4 years old. Originates from
A
neural crest cells. Occurs anywhere along the sympathetic chain.
Most common presentation is abdominal distension and a firm, irregular mass that can cross the
midline (vs Wilms tumor, which is smooth and unilateral). Less likely to develop hypertension
than with pheochromocytoma (neuroblastoma is normotensive). Can also present with
opsoclonus-myoclonus syndrome (“dancing eyes-dancing feet”).
HVA and VMA (catecholamine metabolites) in urine. Homer-Wright rosettes (neuroblasts
surrounding a central area of neuropil A ) characteristic of neuroblastoma and medulloblastoma.
Bombesin and NSE ⊕. Associated with amplification of N-myc oncogene.
FAS1_2023_08-Endocrine.indd 354 11/17/22 9:00 PM
Endocrine
endocrine—Pathology SEC TION III 355
Pheochromocytoma
ETIOLOGY Most common tumor of the adrenal medulla Rule of 10’s:
in adults (black arrow in A ; red arrow points 10% malignant
A
to bone metastases). Derived from chromaffin 10% bilateral
cells (arise from neural crest). 10% extra-adrenal (eg, bladder wall, organ of
May be associated with germline mutations (eg, Zuckerkandl)
NF-1, VHL, RET [MEN 2A, 2B]). 10% calcify
10% kids
SYMPTOMS Most tumors secrete epinephrine, Episodic hyperadrenergic symptoms (5 P’s):
norepinephrine, and dopamine, which can Pressure ( BP)
cause episodic hypertension. May also secrete Pain (headache)
EPO polycythemia. Perspiration
Symptoms occur in “spells”—relapse and remit. Palpitations (tachycardia)
Pallor
FINDINGS catecholamines and metanephrines (eg, Chromogranin, synaptophysin and NSE ⊕.
homovanillic acid, vanillylmandelic acid) in
urine and plasma.
TREATMENT Irreversible α-antagonists (eg, Phenoxybenzamine for pheochromocytoma.
phenoxybenzamine) followed by β-blockers
prior to tumor resection. α-blockade must be
achieved before giving β-blockers to avoid a
hypertensive crisis. A before B.
FAS1_2023_08-Endocrine.indd 355 11/17/22 9:00 PM
356 SEC TION III Endocrine
endocrine—Pathology
Multiple endocrine All MEN syndromes have autosomal dominant inheritance.
neoplasias The X-MEN are dominant over villains.
SUBTYPE CHARACTERISTICS
MEN1 Pituitary tumors (prolactin or GH)
Pancreatic endocrine tumors—Zollinger-Ellison syndrome, insulinomas, VIPomas, glucagonomas
(rare)
Parathyroid adenomas
Associated with mutation of MEN1 (tumor suppressor, codes for menin, chromosome 11),
angiofibromas, collagenomas, meningiomas
MEN2A Parathyroid hyperplasia
Medullary thyroid carcinoma—neoplasm of parafollicular C cells; secretes calcitonin; prophylactic
thyroidectomy required
Pheochromocytoma (secretes catecholamines)
Associated with mutation in RET (protooncogene, codes for receptor tyrosine kinase, chromosome 10)
MEN2B Medullary thyroid carcinoma
A
Pheochromocytoma
Mucosal neuromas A (oral/intestinal ganglioneuromatosis)
Associated with marfanoid habitus; mutation in RET gene
MEN1 = 3 P’s MEN2A = 2 P’s, 1 M MEN2B = 1 P, 2 M’s
Medullary
thyroid carcinoma
Mucosal
Pituitary Parathyroid neuromas
Pheochromocytoma
Pancreas
FAS1_2023_08-Endocrine.indd 356 11/17/22 9:00 PM
Endocrine
endocrine—Pathology SEC TION III 357
Pancreatic islet cell tumors
Insulinoma Tumor of pancreatic β cells overproduction of insulin hypoglycemia.
May see Whipple triad: low blood glucose, symptoms of hypoglycemia (eg, lethargy, syncope,
diplopia), and resolution of symptoms after normalization of plasma glucose levels. Symptomatic
patients have blood glucose and C-peptide levels (vs exogenous insulin use). ∼ 10% of cases
associated with MEN1 syndrome.
Treatment: surgical resection.
Glucagonoma Tumor of pancreatic α cells overproduction of glucagon.
Presents with 6 D’s: dermatitis (necrolytic migratory erythema), diabetes (hyperglycemia), DVT,
declining weight, depression, diarrhea.
Treatment: octreotide, surgical resection.
Somatostatinoma Tumor of pancreatic δ cells overproduction of somatostatin secretion of secretin,
cholecystokinin, glucagon, insulin, gastrin, gastric inhibitory peptide (GIP).
May present with diabetes/glucose intolerance, steatorrhea, gallstones, achlorhydria.
Treatment: surgical resection; somatostatin analogs (eg, octreotide) for symptom control.
Carcinoid tumors Carcinoid tumors arise from neuroendocrine cells, most commonly in the intestine or lung.
A
Neuroendocrine cells secrete 5-HT, which undergoes hepatic first-pass metabolism and
enzymatic breakdown by MAO in the lung. If 5-HT reaches the systemic circulation (eg, after
liver metastasis), carcinoid tumor may present with carcinoid syndrome—episodic flushing,
diarrhea, wheezing, right-sided valvular heart disease (eg, tricuspid regurgitation, pulmonic
stenosis), niacin deficiency (pellagra), urinary 5-HIAA.
Histology: rosettes A , chromogranin A ⊕, synaptophysin ⊕.
Treatment: surgical resection, somatostatin analog (eg, octreotide) or tryptophan hydroxylase
inhibitor (eg, telotristat) for symptom control.
Rule of thirds:
1/3 metastasize
1/3 present with 2nd malignancy
1/3 are multiple
Zollinger-Ellison Gastrin-secreting tumor (gastrinoma) of duodenum or pancreas. Acid hypersecretion causes
syndrome recurrent ulcers in duodenum and jejunum. Presents with abdominal pain (peptic ulcer disease,
distal ulcers), diarrhea (malabsorption). Positive secretin stimulation test: gastrin levels after
administration of secretin, which normally inhibits gastrin release. May be associated with
MEN1.
FAS1_2023_08-Endocrine.indd 357 11/17/22 9:00 PM
358 SEC TION III Endocrine
endocrine—Pharmacology
ENDOCRINE—PHARMACOLOGY
`
Diabetes mellitus All patients with diabetes mellitus should receive education on diet, exercise, blood glucose
therapy monitoring, and complication management. Treatment differs based on the type of diabetes and
glycemic control:
Type 1 DM—insulin replacement
Type 2 DM—oral agents (metformin is first line), non-insulin injectables, insulin replacement;
weight loss particularly helpful in lowering blood glucose
Gestational DM—insulin replacement if nutrition therapy and exercise alone fail
Regular (short-acting) insulin is preferred for DKA (IV), hyperkalemia (+ glucose), stress
hyperglycemia.
Metformin,
pioglitazone
insulin sensitivity
Adipose tissue
Skeletal muscle
GLP-1 analogs, DPP-4
glucose production inhibitors, amylin analogs
Liver
glucagon release
SGLT2 inhibitors Pancreas (α cells)
glucose reabsorption gastric emptying
Kidney Stomach
Sulfonylureas, meglitinides,
GLP-1 analogs, DPP-4
α-Glucosidase inhibitors inhibitors
glucose absorption insulin secretion
Intestine Pancreas (β cells)
DRUG MECHANISM ADVERSE EFFECTS
Insulin preparations
Rapid acting (no lag): Bind insulin receptor (tyrosine kinase activity) Hypoglycemia, lipodystrophy, hypersensitivity
lispro, aspart, glulisine Liver: glucose storage as glycogen reactions (rare), weight gain
Short acting: Muscle: glycogen, protein synthesis
regular Fat: TG storage Lispro, aspart, glulisine
Plasma insulin level
Intermediate acting: Cell membrane: K+ uptake Regular
NPH NPH
Long acting: Detemir
Glargine
detemir, glargine
Very long acting:
degludec 0 2 4 6 8 10 12 14 16 18
Hours
FAS1_2023_08-Endocrine.indd 358 11/17/22 9:00 PM
Endocrine
endocrine—Pharmacology SEC TION III 359
Diabetes mellitus therapy (continued)
DRUG MECHANISM ADVERSE EFFECTS
Increase insulin sensitivity
Metformin Inhibits mitochondrial glycerol-3-phosphate GI upset, lactic acidosis (use with caution in
dehydrogenase (mGPD) inhibition of renal insufficiency), vitamin B12 deficiency.
hepatic gluconeogenesis and the action of Weight loss (often desired).
glucagon.
glycolysis, peripheral glucose uptake ( insulin
sensitivity).
Pioglitazone Activate PPAR-γ (a nuclear receptor) insulin Weight gain, edema, HF, risk of fractures.
sensitivity and levels of adiponectin Delayed onset of action (several weeks).
regulation of glucose metabolism and fatty
acid storage.
Increase insulin secretion
Sulfonylureas (1st gen)
Chlorpropamide,
tolbutamide Disulfiram-like reaction with first-generation
Sulfonylureas (2nd gen) Close K+ channels in pancreatic B cell
membrane cell depolarizes insulin sulfonylureas only (rarely used).
Glipizide, glyburide Hypoglycemia ( risk in renal insufficiency),
release via Ca2+ influx.
Meglitinides weight gain.
Nateglinide,
repaglinide
Increase glucose-induced insulin secretion
GLP-1 analogs glucagon release, gastric emptying, Nausea, vomiting, pancreatitis. Weight loss
Exenatide, liraglutide, glucose-dependent insulin release. (often desired).
semaglutide satiety (often desired).
DPP-4 inhibitors Inhibit DPP-4 enzyme that deactivates GLP-1 Respiratory and urinary infections, weight
Linagliptin, saxagliptin, glucagon release, gastric emptying. neutral.
sitagliptin glucose-dependent insulin release. satiety (often desired).
Decrease glucose absorption
Sodium-glucose Block reabsorption of glucose in proximal Glucosuria (UTIs, vulvovaginal candidiasis),
co-transporter 2 convoluted tubule. dehydration (orthostatic hypotension), weight
inhibitors loss. Glucose flows in urine.
Canagliflozin, Use with caution in renal insufficiency
dapagliflozin, ( efficacy with GFR).
empagliflozin
α-glucosidase Inhibit intestinal brush-border α-glucosidases GI upset, bloating.
inhibitors delayed carbohydrate hydrolysis and glucose Not recommended in renal insufficiency.
Acarbose, miglitol absorption postprandial hyperglycemia.
Others
Amylin analogs glucagon release, gastric emptying. Hypoglycemia, nausea. satiety (often desired).
Pramlintide
FAS1_2023_08-Endocrine.indd 359 11/17/22 9:00 PM
You might also like
- Systemic responses to injury: an overview of hormonal regulationDocument56 pagesSystemic responses to injury: an overview of hormonal regulationFalling HateNo ratings yet
- Endo Other Hormones ElhDocument12 pagesEndo Other Hormones Elhodiodi57No ratings yet
- Dermatologic manifestations of hypopituitarismDocument10 pagesDermatologic manifestations of hypopituitarismWPRN RanasingheNo ratings yet
- Endocrinology TableDocument3 pagesEndocrinology TableWilliam PartonNo ratings yet
- Endocrine Glands and Hormone SignallingDocument12 pagesEndocrine Glands and Hormone SignallingMohammed EljackNo ratings yet
- endorine systemDocument27 pagesendorine systemBenjamin YickNo ratings yet
- Diseases of the Pituitary GlandDocument8 pagesDiseases of the Pituitary GlandSheryl Layne Lao-SebrioNo ratings yet
- Diseases of Glucocorticoid Excess & Deficiency - Cushing & Addison SyndromesDocument3 pagesDiseases of Glucocorticoid Excess & Deficiency - Cushing & Addison SyndromesKeneyziaNo ratings yet
- Biosynthesis of HistamineDocument11 pagesBiosynthesis of HistamineAnaliza Kitongan LantayanNo ratings yet
- Systemic Response To InjuryDocument58 pagesSystemic Response To InjuryJustinNo ratings yet
- Endocrine Day 1 TemplateDocument17 pagesEndocrine Day 1 TemplateMikeNo ratings yet
- Adrenal Control Within The Body: Presented byDocument20 pagesAdrenal Control Within The Body: Presented byhumnaNo ratings yet
- Adrenal Gland Physiology (DRDocument77 pagesAdrenal Gland Physiology (DRapi-3769252100% (4)
- UP Manila Disorders of Adrenal CortexDocument3 pagesUP Manila Disorders of Adrenal CortexGwendareign ElizanNo ratings yet
- FOUR MAIN GROUPS OF IMMUNOSUPPRESSANT DRUGSDocument52 pagesFOUR MAIN GROUPS OF IMMUNOSUPPRESSANT DRUGSFransiscus RivaldyNo ratings yet
- Endocrine 2Document13 pagesEndocrine 2Erika Mae Sta. MariaNo ratings yet
- Gangguan Kel. AdrenalDocument16 pagesGangguan Kel. AdrenalInna Nur Fitri DahliyantiNo ratings yet
- Adenohypophysis: HormonesDocument4 pagesAdenohypophysis: HormonesTomilynjan GarpaNo ratings yet
- Adrenal Glands Clinical Chemistry (Laboratory) : Lesson 7Document5 pagesAdrenal Glands Clinical Chemistry (Laboratory) : Lesson 7Cherry Ann ColechaNo ratings yet
- CorticosteroidsDocument42 pagesCorticosteroidsejiNo ratings yet
- Clinical Chemistry 3: EndocrinologyDocument21 pagesClinical Chemistry 3: EndocrinologyRomie Solacito100% (3)
- Ana Upward Ballein ThrowDocument16 pagesAna Upward Ballein ThrowJohnny eawNo ratings yet
- How to Safely Stop Glucocorticoid TherapyDocument5 pagesHow to Safely Stop Glucocorticoid TherapysimonchikNo ratings yet
- Endocrine Drugs by JIS DumbriqueDocument10 pagesEndocrine Drugs by JIS DumbriqueYangyang R. AzradNo ratings yet
- Disorders of The Anterior Pituitary and HypothalamusDocument121 pagesDisorders of The Anterior Pituitary and Hypothalamusbiniam MesfinNo ratings yet
- Adrnl StressDocument43 pagesAdrnl StressPraveen ChowdharyNo ratings yet
- Hormonal Control (Endocrine GlandsDocument7 pagesHormonal Control (Endocrine GlandsShahzaib AhmedNo ratings yet
- Cushing Syndrome Addisons 1Document6 pagesCushing Syndrome Addisons 1Czarena Ysabelle PayotNo ratings yet
- Pathophysiology of Type 2 Diabetes MellitusDocument5 pagesPathophysiology of Type 2 Diabetes MellitusPearl JuntillaNo ratings yet
- BasicsDocument120 pagesBasicsbiniam MesfinNo ratings yet
- Hormones List - NavyaDocument4 pagesHormones List - NavyaNavya GuptaNo ratings yet
- The Endocrine System: Properties: Mechanisms of Hormone ActionDocument5 pagesThe Endocrine System: Properties: Mechanisms of Hormone ActionaudreyNo ratings yet
- 1.2 CorticosteroidsDocument9 pages1.2 Corticosteroidsua091031No ratings yet
- Dr. Dwi Indria Anggraini, MSC., Spkk. Faculty of Medicine Lampung UniversityDocument36 pagesDr. Dwi Indria Anggraini, MSC., Spkk. Faculty of Medicine Lampung UniversityAnfasha_26No ratings yet
- Adrenal Cortex - Corticosteroids Corticosteroids: GonadotrocorticoidsDocument13 pagesAdrenal Cortex - Corticosteroids Corticosteroids: GonadotrocorticoidsThierd Cañete IIINo ratings yet
- Articulos de HipertensionDocument18 pagesArticulos de HipertensionUlises RaceNo ratings yet
- The Role of Hormones in Regulating Physiological ProcessesDocument26 pagesThe Role of Hormones in Regulating Physiological ProcessesFadli IlhamNo ratings yet
- Endocrine Disorders Cont... (For ADRENAL GLAND)Document4 pagesEndocrine Disorders Cont... (For ADRENAL GLAND)Ernie G. Bautista II, RN, MD100% (3)
- Adrenocortical SteroidsDocument63 pagesAdrenocortical SteroidsmisssarjeeNo ratings yet
- Endocrine System: Harliansyah Dept Biochemistry University of YARSIDocument26 pagesEndocrine System: Harliansyah Dept Biochemistry University of YARSIAnonymous 7r6o3g4jNo ratings yet
- Endocrine & Metabolic Response to Injury ExplainedDocument29 pagesEndocrine & Metabolic Response to Injury ExplainedArWiny Wulandari HipiNo ratings yet
- Adrenocortical Hormones: Learning ObjectivesDocument3 pagesAdrenocortical Hormones: Learning ObjectivesMarlin Berliannanda TawayNo ratings yet
- Intercellular CommunicationDocument8 pagesIntercellular CommunicationDina MaulidaNo ratings yet
- Intercellular Communication Mechanisms and DiseasesDocument8 pagesIntercellular Communication Mechanisms and DiseasesDina MaulidaNo ratings yet
- Congenital Adrenal Hyperplasia:: Diagnosis, Evaluation, and ManagementDocument11 pagesCongenital Adrenal Hyperplasia:: Diagnosis, Evaluation, and Managementsabachannel456No ratings yet
- Corticosteriod Hormones Role Imbalance and Treatment Lec 29Document14 pagesCorticosteriod Hormones Role Imbalance and Treatment Lec 29abdullah808No ratings yet
- Endocrine 1.2 CushingDocument7 pagesEndocrine 1.2 CushingJem QuintoNo ratings yet
- General Biology Lesson 13Document13 pagesGeneral Biology Lesson 13GUCOR, LOVELY SHANE C.No ratings yet
- 3 - Steroid and Gonadal DrugsDocument21 pages3 - Steroid and Gonadal DrugsSoc Gerren TuasonNo ratings yet
- CH 16 Endocrine TableDocument9 pagesCH 16 Endocrine TablebetazenNo ratings yet
- Metabolic response to injuryDocument5 pagesMetabolic response to injuryZllison Mae Teodoro Mangabat100% (1)
- CLINICAL CHEMISTRYDocument11 pagesCLINICAL CHEMISTRYangelwithoutwings321No ratings yet
- Anatomy and Physiology of The Endocrine SystemDocument41 pagesAnatomy and Physiology of The Endocrine SystemJessica Glitter100% (2)
- Gland Stimulation of Hormones Produced Action Gland: Fsh/Lh-Gonads ActhDocument2 pagesGland Stimulation of Hormones Produced Action Gland: Fsh/Lh-Gonads ActhshirleyNo ratings yet
- Pharmacology of Nonsteroidal Anti-Inflammatory Drugs and GlucocorticoidsDocument56 pagesPharmacology of Nonsteroidal Anti-Inflammatory Drugs and GlucocorticoidsRidhaNo ratings yet
- Glucocorticoids & Their EffectsDocument5 pagesGlucocorticoids & Their EffectsJENICCA GONo ratings yet
- Metabolic Trauma: (Stress ResponseDocument42 pagesMetabolic Trauma: (Stress ResponsevisagaNo ratings yet
- Endo ReviewerDocument5 pagesEndo ReviewerZIAN LABADIANo ratings yet
- Histamine and AntihistaminesDocument26 pagesHistamine and Antihistaminesgloria anyebeNo ratings yet
- Immunology ' Immunology ' Section Ii: Immunology-Cellular Components Immunology-Cellular ComponentsDocument20 pagesImmunology ' Immunology ' Section Ii: Immunology-Cellular Components Immunology-Cellular ComponentsLuis Jose VelazquezNo ratings yet
- Biochemistry ' Biochemistry ' Section Ii: Biochemistry-Genetics Biochemistry-GeneticsDocument20 pagesBiochemistry ' Biochemistry ' Section Ii: Biochemistry-Genetics Biochemistry-GeneticsLuis Jose VelazquezNo ratings yet
- Neurology and Special Senses ' Neurology and Special Senses ' Section IiiDocument20 pagesNeurology and Special Senses ' Neurology and Special Senses ' Section IiiLuis Jose VelazquezNo ratings yet
- Biochemistry ' Biochemistry ' Section Ii: Biochemistry-Metabolism Biochemistry-MetabolismDocument20 pagesBiochemistry ' Biochemistry ' Section Ii: Biochemistry-Metabolism Biochemistry-MetabolismLuis Jose VelazquezNo ratings yet
- Endocrine ' Endocrine ' Section Iii: Endocrine-Pharmacology Endocrine-PharmacologyDocument20 pagesEndocrine ' Endocrine ' Section Iii: Endocrine-Pharmacology Endocrine-PharmacologyLuis Jose VelazquezNo ratings yet
- Psychiatry ' Psychiatry ' Section Iii: Psychiatry-Pathology Psychiatry-PathologyDocument20 pagesPsychiatry ' Psychiatry ' Section Iii: Psychiatry-Pathology Psychiatry-PathologyLuis Jose VelazquezNo ratings yet
- Image Acknowledgments Image Acknowledgments Section Iv: Clin Med. 2015 May 4 (5) :884-917. DOI: 10.3390/jcm4050884Document20 pagesImage Acknowledgments Image Acknowledgments Section Iv: Clin Med. 2015 May 4 (5) :884-917. DOI: 10.3390/jcm4050884Luis Jose VelazquezNo ratings yet
- Index Index: SMN1 Mutation, 546Document9 pagesIndex Index: SMN1 Mutation, 546Luis Jose VelazquezNo ratings yet
- Index Index: Streptococcus PneumoniaeDocument20 pagesIndex Index: Streptococcus PneumoniaeLuis Jose VelazquezNo ratings yet
- Rapid Review ' Section Iii: Easily Confused MedicationsDocument20 pagesRapid Review ' Section Iii: Easily Confused MedicationsLuis Jose VelazquezNo ratings yet
- (First Name) SdadsDocument1 page(First Name) SdadsLuis Jose VelazquezNo ratings yet
- Index Index: MUTYH-associated PolyposisDocument20 pagesIndex Index: MUTYH-associated PolyposisLuis Jose VelazquezNo ratings yet
- ZXZXCZCDocument1 pageZXZXCZCLuis Jose VelazquezNo ratings yet
- Cool Cover Letter 5Document1 pageCool Cover Letter 5Ahmed Asif IqbalNo ratings yet
- First NameDocument1 pageFirst Namesanjay kashyapNo ratings yet
- First NameDocument1 pageFirst Namesanjay kashyapNo ratings yet
- A319/A320/A321 Technical Training Manual Mechanics / Electrics & Avionics Course 33 LightsDocument224 pagesA319/A320/A321 Technical Training Manual Mechanics / Electrics & Avionics Course 33 LightsAhmedHamdyElsaidy100% (3)
- 7 Ways of Looking at Grammar China EditDocument20 pages7 Ways of Looking at Grammar China EditAshraf MousaNo ratings yet
- Microsoft Security Product Roadmap Brief All Invitations-2023 AprilDocument5 pagesMicrosoft Security Product Roadmap Brief All Invitations-2023 Apriltsai wen yenNo ratings yet
- TC-21FJ30LA: Service ManualDocument33 pagesTC-21FJ30LA: Service ManualRajo Peto alamNo ratings yet
- Get TRDocDocument209 pagesGet TRDoc10131No ratings yet
- Laser Beam Machining (LBM)Document10 pagesLaser Beam Machining (LBM)RAMAKANT RANANo ratings yet
- Assignement 4Document6 pagesAssignement 4sam khanNo ratings yet
- Cellular Tower: Bayombong, Nueva VizcayaDocument17 pagesCellular Tower: Bayombong, Nueva VizcayaMonster PockyNo ratings yet
- Drainage Manual: State of Florida Department of TransportationDocument78 pagesDrainage Manual: State of Florida Department of TransportationghoyarbideNo ratings yet
- SW - Simulation - Product MatrixDocument2 pagesSW - Simulation - Product Matrixsamar2011No ratings yet
- NIJ Sawmark Analysis Manual for Criminal MutilationDocument49 pagesNIJ Sawmark Analysis Manual for Criminal MutilationAntonio jose Garrido carvajalinoNo ratings yet
- Rectangle StabbingDocument49 pagesRectangle StabbingApurba DasNo ratings yet
- Connect 4 UML Class DiagramDocument1 pageConnect 4 UML Class DiagramDuy Nguyễn Văn NhậtNo ratings yet
- TaxonomyDocument56 pagesTaxonomyKrezia Mae SolomonNo ratings yet
- Workbook. Unit 3. Exercises 5 To 9. RESPUESTASDocument3 pagesWorkbook. Unit 3. Exercises 5 To 9. RESPUESTASRosani GeraldoNo ratings yet
- Timesheet 25Document1 pageTimesheet 25Gwenzieh TugadeNo ratings yet
- Asian Paint FMVRDocument20 pagesAsian Paint FMVRdeepaksg787No ratings yet
- An Improvement in Endodontic Therapy You Will AppreciateDocument2 pagesAn Improvement in Endodontic Therapy You Will AppreciateIs MNo ratings yet
- The History of Coins and Banknotes in Mexico: September 2012Document35 pagesThe History of Coins and Banknotes in Mexico: September 2012Mladen VidovicNo ratings yet
- WhatsoldDocument141 pagesWhatsoldLuciana KarajalloNo ratings yet
- Problem solving and decision making in nutrition postgraduate studiesDocument55 pagesProblem solving and decision making in nutrition postgraduate studiesteklayNo ratings yet
- SAP MM ReportsDocument59 pagesSAP MM Reportssaprajpal95% (21)
- Vertical Transportation SystemDocument15 pagesVertical Transportation SystemAnupama MorankarNo ratings yet
- The Facility Manager's HandbookDocument362 pagesThe Facility Manager's HandbookLuân Châu100% (14)
- Adafruit Color SensorDocument25 pagesAdafruit Color Sensorarijit_ghosh_18No ratings yet
- 41-How To Calculate Air Temp in Unconditioned SpacesDocument3 pages41-How To Calculate Air Temp in Unconditioned Spacesalmig200No ratings yet
- Introduction To Human Anatomy & Physiology: Prepared by Mr. Abhay Shripad Joshi Yash Institute of Pharmacy AurangabadDocument18 pagesIntroduction To Human Anatomy & Physiology: Prepared by Mr. Abhay Shripad Joshi Yash Institute of Pharmacy AurangabadMourian AmanNo ratings yet
- English 9Document26 pagesEnglish 9Joann Gaan YanocNo ratings yet
- Python operators and data types quizDocument34 pagesPython operators and data types quizATUL SHARMANo ratings yet
- Aimt ProspectusDocument40 pagesAimt ProspectusdustydiamondNo ratings yet