Professional Documents
Culture Documents
NIDA Clinical Trial Network
Uploaded by
Nitesh Nishant0 ratings0% found this document useful (0 votes)
9 views1 pageThis certificate recognizes that Shalini Jha completed a six-hour Good Clinical Practice course through the NIDA Clinical Trials Network, passing modules on topics like informed consent, adverse events, the research protocol, and documentation. The course was completed on June 23, 2023 and the certification is valid through June 23, 2026.
Original Description:
Copyright
© © All Rights Reserved
Available Formats
PDF, TXT or read online from Scribd
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentThis certificate recognizes that Shalini Jha completed a six-hour Good Clinical Practice course through the NIDA Clinical Trials Network, passing modules on topics like informed consent, adverse events, the research protocol, and documentation. The course was completed on June 23, 2023 and the certification is valid through June 23, 2026.
Copyright:
© All Rights Reserved
Available Formats
Download as PDF, TXT or read online from Scribd
0 ratings0% found this document useful (0 votes)
9 views1 pageNIDA Clinical Trial Network
Uploaded by
Nitesh NishantThis certificate recognizes that Shalini Jha completed a six-hour Good Clinical Practice course through the NIDA Clinical Trials Network, passing modules on topics like informed consent, adverse events, the research protocol, and documentation. The course was completed on June 23, 2023 and the certification is valid through June 23, 2026.
Copyright:
© All Rights Reserved
Available Formats
Download as PDF, TXT or read online from Scribd
You are on page 1of 1
NIDA Clinical Trials Network
Certificate of Completion
is hereby granted to
Shalini Jha
to certify your completion of the six-hour required course on:
GOOD CLINICAL PRACTICE
MODULE: STATUS:
Introduction N/A
Institutional Review Boards Passed
Informed Consent Passed
Confidentiality & Privacy Passed
Participant Safety & Adverse Events Passed
Quality Assurance Passed
The Research Protocol Passed
Documentation & Record-Keeping Passed
Research Misconduct Passed
Roles & Responsibilities Passed
Recruitment & Retention Passed
Investigational New Drugs Passed
Course Completion Date: 23 June 2023
CTN Expiration Date: 23 June 2026
Eve Jelstrom, Principal Investigator
NDAT CTN Clinical Coordinating Center
Good Clinical Practice, Version 5, effective 03-Mar-2017
This training has been funded in whole or in part with Federal funds from the National Institute on Drug
Abuse, National Institutes of Health, Department of Health and Human Services, under Contract No.
HHSN27201201000024C.
You might also like
- GCP Certificate - Prof. Iliya LozevDocument1 pageGCP Certificate - Prof. Iliya LozevDSpirovaNo ratings yet
- NIDA Clinical Trials Network: Alton DsilvaDocument1 pageNIDA Clinical Trials Network: Alton DsilvaCRCE 9192 AltonNo ratings yet
- CertificateDocument1 pageCertificatembibaellen8No ratings yet
- NIDA CertDocument1 pageNIDA CertMargarita Limon BalunesNo ratings yet
- NIDA Clinical Trials Network: Rene Vanegas PoloDocument1 pageNIDA Clinical Trials Network: Rene Vanegas Polorene vanegas poloNo ratings yet
- Certificate DR DayatDocument1 pageCertificate DR DayatIndraSeptianNo ratings yet
- GCP Certificate 2017Document1 pageGCP Certificate 2017Mariana0% (1)
- Certificado Todo PDFDocument12 pagesCertificado Todo PDFLiss VillacresesNo ratings yet
- NIDA Clinical Trials Network: Niray MontoyaDocument1 pageNIDA Clinical Trials Network: Niray MontoyaJuan David MartinNo ratings yet
- Ethics CertificateDocument2 pagesEthics Certificateprajyotmagadum3304No ratings yet
- Good Clinical PracticeDocument2 pagesGood Clinical PracticeNicholeGarcesCisneros0% (3)
- Way To Quality: Accreditation Process: Dr. B.K. RanaDocument36 pagesWay To Quality: Accreditation Process: Dr. B.K. RanaThilinaAbhayarathneNo ratings yet
- Role of SponsorDocument30 pagesRole of SponsorYellow GutierrezNo ratings yet
- Outcomes of Ebp Process 2017Document3 pagesOutcomes of Ebp Process 2017api-272725467100% (1)
- Desarrollo de Base de Evidencia en Acreditación SaludDocument4 pagesDesarrollo de Base de Evidencia en Acreditación SaludNéstor Correa PreciadoNo ratings yet
- Form - LNRDocument6 pagesForm - LNRrobin.rjmNo ratings yet
- Quality Assurance in Qualitative ResearchDocument13 pagesQuality Assurance in Qualitative ResearchAndrew TandohNo ratings yet
- Cqi Pahrio March 27, 2023Document106 pagesCqi Pahrio March 27, 2023KUYA CHICO MORALESNo ratings yet
- OHST Complete GuideDocument24 pagesOHST Complete GuideFuzail Ayaz100% (1)
- Sample Presentation - Science InternshipDocument29 pagesSample Presentation - Science InternshipAmaayaNo ratings yet
- Introduction To Information Security Audits and Assessments - SparkDocument12 pagesIntroduction To Information Security Audits and Assessments - SparkCarlNo ratings yet
- Accreditation of Eye Hospitals - A Review: Nirmal Fredrick T, Sunitha NirmalDocument7 pagesAccreditation of Eye Hospitals - A Review: Nirmal Fredrick T, Sunitha Nirmalrgene bioscientificNo ratings yet
- Validation in Qualitative Research FinalDocument19 pagesValidation in Qualitative Research FinalChama K BenardNo ratings yet
- ISQua SurveyDocument14 pagesISQua SurveySuprapto DR, SPd,SKp,MMNo ratings yet
- ControllingDocument8 pagesControllingNonito Patrick GaleraNo ratings yet
- Inv Learner Guide v2.1Document10 pagesInv Learner Guide v2.1RamyNo ratings yet
- Performing The Engagement (Audit Evidence) : John Paolo T. JosonDocument12 pagesPerforming The Engagement (Audit Evidence) : John Paolo T. JosonJohn Paolo JosonNo ratings yet
- كورس الجودةDocument120 pagesكورس الجودةgaber 230No ratings yet
- The Team, The Procedures, The Monitor and The Sponsor: Lucy H H Parker Clinical Research Governance ManagerDocument20 pagesThe Team, The Procedures, The Monitor and The Sponsor: Lucy H H Parker Clinical Research Governance ManagerMohammed HammedNo ratings yet
- COMPASSIONATE 3A CLINPATH Activity 1 Comparison of AO 037 and ISO 15189Document17 pagesCOMPASSIONATE 3A CLINPATH Activity 1 Comparison of AO 037 and ISO 15189Adrian CaballesNo ratings yet
- Edit Module 2 Fall CRC 2022Document62 pagesEdit Module 2 Fall CRC 2022Raquel VargasNo ratings yet
- Maiga Ayub Hussein: The African Forum For Research and Education in Health (Afrehealth)Document7 pagesMaiga Ayub Hussein: The African Forum For Research and Education in Health (Afrehealth)Maiga Ayub HusseinNo ratings yet
- PML Company Profile - Clinical TrialDocument11 pagesPML Company Profile - Clinical Trialwisang geniNo ratings yet
- 3655 06 l6 Diploma Qualification HandbookDocument64 pages3655 06 l6 Diploma Qualification Handbooka.mdrashidNo ratings yet
- Ethical Issues in Clinical TrialsDocument16 pagesEthical Issues in Clinical TrialsPranjal KothaleNo ratings yet
- HLTWHS003 Cluster 7 Changqing GuanDocument38 pagesHLTWHS003 Cluster 7 Changqing GuanSujitha ReddyNo ratings yet
- Arens Aas17 PPT 06Document49 pagesArens Aas17 PPT 06a0939809094No ratings yet
- CSP Complete Guide PDFDocument32 pagesCSP Complete Guide PDFiqjreynaNo ratings yet
- J Pak Assoc Dermatol 2012Document5 pagesJ Pak Assoc Dermatol 2012elproedrosNo ratings yet
- CBIC 2020 Candidate Handbook CBIC 2020 Candidate HandbookDocument16 pagesCBIC 2020 Candidate Handbook CBIC 2020 Candidate HandbookAsif Iqbal100% (1)
- NapulanRM - CQI in Records and Clinical Documentation ImprovementDocument92 pagesNapulanRM - CQI in Records and Clinical Documentation ImprovementJeffreyReyesNo ratings yet
- Journal of Biosafety and Biosecurity: Research ArticleDocument4 pagesJournal of Biosafety and Biosecurity: Research ArticleDenise DeniseNo ratings yet
- CPHQ Detailed Content OutlineDocument8 pagesCPHQ Detailed Content Outlineempire707No ratings yet
- Clause 4.2 Interested Parties ListDocument3 pagesClause 4.2 Interested Parties Listwater labNo ratings yet
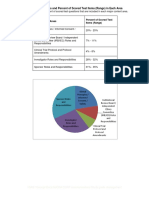
- Five Content Areas and Percent of Scored Test Items (Range) in Each AreaDocument5 pagesFive Content Areas and Percent of Scored Test Items (Range) in Each Areaসোমনাথ মহাপাত্রNo ratings yet
- Root Cause Analysis and Corrective ActionsTraining - OfficialDocument54 pagesRoot Cause Analysis and Corrective ActionsTraining - OfficialAbdunnajar MahamudNo ratings yet
- FDA - Clinical Trial Guidelines Consultation PDFDocument31 pagesFDA - Clinical Trial Guidelines Consultation PDFCzarPaguioNo ratings yet
- Study SetupDocument20 pagesStudy SetupROCKER GAMINGNo ratings yet
- Manpower Audit: Presented By: Saroj Somra Ruchika ChokhraDocument14 pagesManpower Audit: Presented By: Saroj Somra Ruchika ChokhraSaroj Rose75% (4)
- ICDIPDocument8 pagesICDIPyogurt pearsNo ratings yet
- Certification HandbookDocument28 pagesCertification HandbookRahul VashishtNo ratings yet
- Clinical Research Organization / Contract Research OrganizationDocument34 pagesClinical Research Organization / Contract Research OrganizationPHARMAIGNITENo ratings yet
- OSAC - Preferred Terms - 2021Document2 pagesOSAC - Preferred Terms - 2021ucac PFANo ratings yet
- The Complexity of Process ValidationDocument10 pagesThe Complexity of Process ValidationMiguel Angel Pacahuala CristobalNo ratings yet
- Gokula Metropolis Clinical Research Centre & Sir Ganga Ram HospitalDocument27 pagesGokula Metropolis Clinical Research Centre & Sir Ganga Ram HospitalSirgangaramhospital HospitalNo ratings yet
- Development of A Guideline - PITIkabi2017Document32 pagesDevelopment of A Guideline - PITIkabi2017Intan Eklesiana NapitupuluNo ratings yet
- ORTHO Clinical DiagnosticsDocument4 pagesORTHO Clinical DiagnosticsHuy Trần ThiệnNo ratings yet
- Clinical Research Associate - The Comprehensive Guide: Vanguard ProfessionalsFrom EverandClinical Research Associate - The Comprehensive Guide: Vanguard ProfessionalsNo ratings yet
- Medical Credentialing Specialist - The Comprehensive Guide: Vanguard ProfessionalsFrom EverandMedical Credentialing Specialist - The Comprehensive Guide: Vanguard ProfessionalsNo ratings yet
- MERN Stack Developer Resume of Tasneem Nur PDFDocument1 pageMERN Stack Developer Resume of Tasneem Nur PDFNitesh NishantNo ratings yet
- Archana NewDocument1 pageArchana NewNitesh NishantNo ratings yet
- Develop A Service OrientationDocument1 pageDevelop A Service OrientationNitesh NishantNo ratings yet
- Anchal BishtDocument2 pagesAnchal BishtNitesh NishantNo ratings yet
- Qrcode 1093491Document1 pageQrcode 1093491Nitesh NishantNo ratings yet
- PDF Resume NovDocument2 pagesPDF Resume NovNitesh NishantNo ratings yet
- Resume OverleafDocument1 pageResume OverleafNitesh NishantNo ratings yet
- Prachi Sonje (Resume)Document1 pagePrachi Sonje (Resume)Nitesh NishantNo ratings yet
- Disha CVDocument1 pageDisha CVNitesh NishantNo ratings yet
- Santosh PatilresumeDocument2 pagesSantosh PatilresumeNitesh NishantNo ratings yet
- Resume of Roli NayanDocument4 pagesResume of Roli NayanNitesh NishantNo ratings yet
- Tax Invoice: 1 Dwc7Dl7Gv Apple Iphone 11 (4 Gb/128 GB) Refurbished Good White Refurbished Mobiles 8517 1 28149.0 28149.0Document2 pagesTax Invoice: 1 Dwc7Dl7Gv Apple Iphone 11 (4 Gb/128 GB) Refurbished Good White Refurbished Mobiles 8517 1 28149.0 28149.0Nitesh NishantNo ratings yet