Professional Documents
Culture Documents
Guided Plan-7 (E)
Uploaded by
abhiraw30062005Original Title
Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
Guided Plan-7 (E)
Uploaded by
abhiraw30062005Copyright:
Available Formats
JEE (MAIN + ADVANCED) 2023
GUIDED REVISION JEE (Main + Advanced) 2023
LEADER + ENTHUSIAST COURSE
LEADER + ENTHUSIAST COURSE
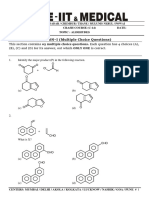
ORGANIC CHEMISTRY GR # 07 GUIDED REVISION PLAN
SECTION–I : (i) Only One option correct Type
This section contains 05 multiple choice questions. Each question has four choices (A), (B), (C) and
(D) out of which ONLY ONE is correct. 3(–1)
1. The principle functional group in following compound for IUPAC naming :
O
N H
OH O
(A) Aldehyde (B) Ketone (C) Amine (D) Amide
2. Which of the following represent aromatic compound :
O O O
(A) (B) (C) (D)
O
+
O
NO2
(i )CH3I,K 2CO3
3. HO
(ii) H2SO4 ,HNO3
Major product of the reaction is :
NO2 NO2
H3C
(A) HO (B) MeO
NO2 NO2
NO2 NO2
O2N O2N
(C) HO (D) MeO
CH3
ORGANIC CHEMISTRY / GR # 07 (Set-3) E-1/7
JEE (MAIN + ADVANCED) 2023
GUIDED REVISION LEADER + ENTHUSIAST COURSE
4. In the following reaction the major product [X] is :
O
Me
(i )SeO2 / HOAc
[X]
(ii )NaOH
(iii )H3O
O HOOC OH COOH
COOH Me COOH
Me OH (C) Me
(A) (B) (D)
5. Major product 'Y' in following reaction sequence is :
–
O3 OH
O Zn X Y
C–H O O
C–H O
(A) (B) (C) (D)
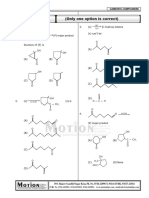
(ii) One or more options correct Type
This section contains 05 multiple choice questions. Each question has four choices (A), (B), (C) and
(D) out of which ONE or MORE are correct. 4(–1)
6. Identify correct reaction set(s) ?
Cl O -
O + O
+ Br N Br
N -
O HS S
(A) + Base
Cl Cl
Cl
(B) MeOH MeO
O O
H3C CO2H H3C CO2H
(C) OH-,Ag+
Br OH
O
O
(D) Base
Cl
O O
ORGANIC CHEMISTRY / GR # 07 (Set-3) E-2/7
JEE (MAIN + ADVANCED) 2023
GUIDED REVISION LEADER + ENTHUSIAST COURSE
7. To prepare 2° amine (as major product) which of the following reactions can be used up :
CH3
+ – LiAlH4 + CH3 –
NC N
(A) (B) OH
CH3
(C) NH3 / (D) O + 1. MeNH2
Cl 2. H2(Pd)
8. Select correct statement(s) out of following?
O NH
(A) is more reactive than towards reaction with PhMgBr
Cl
Cl2 (aq)
(B) O
h
O (major product)
Conc. H 2SO 4
(C) 2 (Major)
(Catalytic)
PhNHNH 2
(D) D-glucose + D-mannose + D-fructose only one osazone
9. Select correct statement(s) out of following for compound given below :
Br(x)
O Br(y)
Br(z)
(A) Br(y) is the most reactive Br towards SN2 reaction in this compound.
(B) Br(z) is the most reactive Br towards SN1 reaction in this compound.
(C) 16 stereoisomers are theoretically possible for this compound.
(D) This compound is capable of showing geometrical isomerism
10. Identify reactions give naturally occuring amino acids?
(1)H–CN (1) Red 'P' + X2
(A) NH (B) Me – CH2 – COOH (2) NH
(2)H3O
3
O Cl O O
(1) CH – C C
C CH3
Me OEt
N Na (1) NaOI
(C) (2) H3O (D)
C (2) H
Cl (3) NH3
O
ORGANIC CHEMISTRY / GR # 07 (Set-3) E-3/7
JEE (MAIN + ADVANCED) 2023
GUIDED REVISION LEADER + ENTHUSIAST COURSE
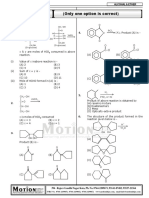
(iii) Paragraph Type
This section contains 01 paragraph each describing theory, experiment, data etc. Each question of
a paragraph has only one correct answer among the four choices (A), (B), (C) and (D).
Paragraph for Questions 11 and 12
N 2H4 OH
C6H10O
(A)
(1) CH3MgBr Conc. H2SO4 (1) O3
(B) (C) (D) OH ,
(E)
(2) H2O , -H2O (2) Zn, H2O Intra Molecular
Aldol condensation
(dicarbonyl
compound)
(1) Li (CH3)2Cu
(2) H2O
Zn - Hg
(F)
HCl
# Compound (A) has (S) - configuration at the chiral center.
# All organic products are major in above reaction scheme
11. Select the correct statement ?
(A) Formation of Compound (F) involves 1, 2-Addition.
(B) Compound (C) on reaction with HBr or HBr, R2O2 gives same organic product.
(C) Compound (A) on reaction with NaCN / HCN followed by H2O gives enantiomers pair as
a product.
Br
(D) Compound (C) can also be obtained as major product when heated with
O K
12. Select the correct one
O
(A) Compound (F) = (B) Compound (C) = CHO
O OH
CH3
(C) Compound (A) = (D) Compound (B) =
H
ORGANIC CHEMISTRY / GR # 07 (Set-3) E-4/7
JEE (MAIN + ADVANCED) 2023
GUIDED REVISION LEADER + ENTHUSIAST COURSE
(iv) Matching List Type
This Section contains 2 multiple choice questions. Each question has matching lists. The codes for
the lists. have choices (A), (B), (C) and (D) out of which ONLY ONE is correct. 4(0)
13. Match the following question :
List - I List-II
(Reaction) (Products)
CH3
(P) HOOC COOH (1) Diastereomers
H D
C 2H 5
CH3
(Q) HOOC COOH (2) Racemic mixture
Ph
HOOC COOH
(R) (3) Meso compound
CH3
COOH
(S) (4) CO2 gas will evolve
COOH
Select CORRECT code for your answer :
(A) (P)1, 4 ; (Q)2, 4 ; (R)1, 4 ; (S)3 (B) (P)1, 4 ; (Q)2, 4 ; (R)1, 4 ; (S)3,4
(C) (P)1, 4 ; (Q)2,3,4 ; (R)1, 4 ; (S)3 (D) (P)1, 4 ; (Q)2, 4 ; (R)1, 3 ; (S)3
14. Mark the correct option assuming monobromination in all reactions (include stereo isomers
wherever possible)
List I List II
Br2
(P)
hv
(1) Even number of optically active products
Br2
(Q)
hv
(2) Odd number of total products obtained
after fractional distillation
Br2
(R)
hv
(3) Even number of total products
Br2
(S)
hv
(4) Even number of optically inactive products
(A) P1,2,4 ; Q2,3,4 ; R1,3,4 ; S1,3,4
(B) P1,3,4 ; Q1,2,3 ; R2,3,4 ; S2,3,4
(C) P1,3,4 ; Q2,3,4 ; R2,3,4 ; S1,3,4
(D) P2,3,4 ; Q1,2,4 ; R1,3,4 ; S2,3,4
ORGANIC CHEMISTRY / GR # 07 (Set-3) E-5/7
JEE (MAIN + ADVANCED) 2023
GUIDED REVISION LEADER + ENTHUSIAST COURSE
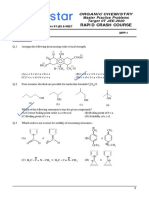
SECTION-III : (Integer Value Correct Type)
This section contains 06 questions. The answer to each question is a single digit Integer, ranging from
0 to 9 (both inclusive) 4(–1)
1. Electrodes are immersed in a solution of pH ‘X’ which is prepared by using 0.1 M formic acid
(Ka = 10–4) and 0.1 M sodiumformate in equal volume. To this solution amino acid, ascorbic
pKa = 4 pKb = 4
NH2
acid, a naturally occurring amino. acid HO—C—CH2—CH is added.
O C O
OH
pKa = 2
Assorbic acid pKa values
Then, if net migration of amino acid is towards anode, then your answer is (X + 1). If migra-
tion is towards is cathode, then your answer is (X + 2) . If migration is neither towards anode
nor towards cathode then your answer is X.
O
2.
Ph—CH=CH—C—CH=CH—Ph
CH2(COOEt)2 (1 mole)
NaOEt
(A)
(Six membered ring)
How many oxygen atoms are present in (A) ?
3. How many equivalents of base are consumed in the following reaction ?
Br2 / OH /
CH3CHO HCBr3 HCOO
(1equivalent)
4. In the given reaction sequence, find number of possible 5 and 6 membered Lactone(s) having
different boiling points :
(i) LiAlH4
'X' (Lactone)
(ii) H2O
Molecular
formula = C6H10O2 (iii) Red P / HI
ORGANIC CHEMISTRY / GR # 07 (Set-3) E-6/7
JEE (MAIN + ADVANCED) 2023
GUIDED REVISION LEADER + ENTHUSIAST COURSE
5. How many of the following compound will give silver mirror with Tollen's reagent ?
CH2OH
H OH
(a) Me–CHO (b) Ph–CHO (c) H (d) Ph–NH–OH
OH H
HO OD
H OH
CH 2OH
HO OH
H COOH
H H O OH
H HO H H OH
(e) H O CH2OH (f) Me–CCH (g) OH H (h)
H OH
OH H HO HO CH 3
O
OH H H H COOH
H
H OH
(i) HCOOH (j) Me–COOH
6. How many theoretically possible dipeptides can be produced if we have only Glycine and
Alanine ?
ORGANIC CHEMISTRY / GR # 07 (Set-3) E-7/7
You might also like
- Alln GRPDocument7 pagesAlln GRPrajkaran3765No ratings yet
- Allen Guided Revision Organic ChemistryDocument74 pagesAllen Guided Revision Organic ChemistrymemepepedankNo ratings yet
- GRP #01Document7 pagesGRP #01Rishav Kumar 10 CNo ratings yet
- Carbonyl Compounds - XII PDFDocument39 pagesCarbonyl Compounds - XII PDFMathoholic OjaNo ratings yet
- Carbonyl Compounds 1654147338445Document33 pagesCarbonyl Compounds 1654147338445Harsh VardhanNo ratings yet
- Guided Plan-6 (E)Document7 pagesGuided Plan-6 (E)abhiraw30062005No ratings yet
- Organic Chemistry Guided Revision Plan-Score AdvancedDocument4 pagesOrganic Chemistry Guided Revision Plan-Score AdvancedNamchrahsiNo ratings yet
- Class Test Aldol and CannizaroDocument9 pagesClass Test Aldol and Cannizaroalomrobi07No ratings yet
- GRP #08Document4 pagesGRP #08rajkaran3765No ratings yet
- Alcohol, Phenol & EtheRDocument8 pagesAlcohol, Phenol & EtheRdevvratchoudhary11989No ratings yet
- SECTION-I (Multiple Choice Questions) : IIT - JEE: 2015 Crash Course (C-14) Date: Topic: AldehydesDocument9 pagesSECTION-I (Multiple Choice Questions) : IIT - JEE: 2015 Crash Course (C-14) Date: Topic: AldehydesSachin DedhiaNo ratings yet
- Aldehyde Ketone Carboxylic Acid and TriangleDocument25 pagesAldehyde Ketone Carboxylic Acid and Trianglejiknown6No ratings yet
- GRP #04Document7 pagesGRP #04rajkaran3765No ratings yet
- Alcohols & Ethers Exercise - IDocument5 pagesAlcohols & Ethers Exercise - IVasudev ArchakNo ratings yet
- Quanta Chemistry: Csir-Net - Iit-Gate - Iit-Jam - Other Msc. Entrance An Institute of Chemical SciencesDocument6 pagesQuanta Chemistry: Csir-Net - Iit-Gate - Iit-Jam - Other Msc. Entrance An Institute of Chemical Sciencesnityananada ChemNo ratings yet
- CarbonylDocument27 pagesCarbonylbpvx4dyqsxNo ratings yet
- Vibrant Academy: (India) Private LimitedDocument6 pagesVibrant Academy: (India) Private LimitedRk ChaudharyNo ratings yet
- Answer Test-1 ON CARBONYL COMPOUNDS.Document4 pagesAnswer Test-1 ON CARBONYL COMPOUNDS.Ishita AgarwalNo ratings yet
- Class Test-1 - Phenol - Without AnswerDocument4 pagesClass Test-1 - Phenol - Without AnswershouryatrialNo ratings yet
- Set 3 Oc GRP 8 Student Copy EngDocument4 pagesSet 3 Oc GRP 8 Student Copy EngAaditya KumarNo ratings yet
- Exercise - I (Only One Option Is Correct) : CH - OhDocument5 pagesExercise - I (Only One Option Is Correct) : CH - Ohd anjilappaNo ratings yet
- Aldehydes & KetonesDocument9 pagesAldehydes & Ketoneskrishna janamNo ratings yet
- Guided Revision: Sot Type 4 (-1) 1Document3 pagesGuided Revision: Sot Type 4 (-1) 1Shubham RajNo ratings yet
- Class Test 6 Reaction of Alkene Ozonolysis Hydrocarbon Without AnswerDocument5 pagesClass Test 6 Reaction of Alkene Ozonolysis Hydrocarbon Without AnswershouryatrialNo ratings yet
- Jumbo Home Test-1 - FinalDocument59 pagesJumbo Home Test-1 - FinalSayali SachinNo ratings yet
- Ocd PP Special On Taut Omer Is MDocument3 pagesOcd PP Special On Taut Omer Is MKartik YadavNo ratings yet
- NQE 2009 Chemistry SolutionsDocument10 pagesNQE 2009 Chemistry Solutionsaleth felicianoNo ratings yet
- Guided Plan-5 (E)Document4 pagesGuided Plan-5 (E)abhiraw30062005No ratings yet
- IIT-JAM 2016 With SolutionDocument25 pagesIIT-JAM 2016 With SolutiongauravNo ratings yet
- GRP #05Document4 pagesGRP #05rajkaran3765No ratings yet
- 2B-Alcohols, Ethers & Phenols - FINAL - 06!03!14 (86-112)Document27 pages2B-Alcohols, Ethers & Phenols - FINAL - 06!03!14 (86-112)udaysrinivasNo ratings yet
- SCH 2358 - Organic Synthesis - Print ReadyDocument4 pagesSCH 2358 - Organic Synthesis - Print ReadyDerick CheruyotNo ratings yet
- Iit Jam Named Reactions RearrangementsDocument8 pagesIit Jam Named Reactions RearrangementsVickyNo ratings yet
- Oc Taas Quiz 6 StudentDocument4 pagesOc Taas Quiz 6 Studentattackerasp1234No ratings yet
- AEP QuestionsDocument8 pagesAEP QuestionsArihant BansalNo ratings yet
- Aldehydes and Ketones: TopicDocument13 pagesAldehydes and Ketones: Topicitzpratyush123098No ratings yet
- Alcohols Phenols EtherDocument55 pagesAlcohols Phenols EtherAnanya AgrawalNo ratings yet
- Name The Following Compounds According To IUPAC System of Nomenclature (Document2 pagesName The Following Compounds According To IUPAC System of Nomenclature (AbubakarNo ratings yet
- Chemistry Alcohol, Ether & Phenol CPP Combine PDFDocument10 pagesChemistry Alcohol, Ether & Phenol CPP Combine PDFVanshika LudhaniNo ratings yet
- GRP #13Document4 pagesGRP #13rajkaran3765No ratings yet
- Aromatic Compounds - QuestionDocument14 pagesAromatic Compounds - Questionhrishik guptaNo ratings yet
- Atp Mega RevDocument14 pagesAtp Mega RevSURAKSHA PATELNo ratings yet
- JEE Advanced General Organic Chemistry Important QuestionsDocument18 pagesJEE Advanced General Organic Chemistry Important QuestionsVaNo ratings yet
- IIT JAM Chemistry Test PaperDocument15 pagesIIT JAM Chemistry Test PaperAnil Kumar100% (1)
- Chemistry: Section - IDocument11 pagesChemistry: Section - ISailendra Narayan SahuNo ratings yet
- C Sol Ch-23 Alcohols, Phenols and EthersDocument6 pagesC Sol Ch-23 Alcohols, Phenols and Ethersmysoftinfo.incNo ratings yet
- PyqsDocument20 pagesPyqsHardik JoshiNo ratings yet
- Rapid Crash Course: Single CorrectDocument8 pagesRapid Crash Course: Single CorrectHudsun HornetNo ratings yet
- C - Ch-24 - Aldehydes, Ketones and Carboxylic AcidsDocument22 pagesC - Ch-24 - Aldehydes, Ketones and Carboxylic Acidsmysoftinfo.incNo ratings yet
- Jumbo Test-2Document5 pagesJumbo Test-2prithvijeetopNo ratings yet
- Iit-Examination Paper-2009 Code 4 Paper 2: InstructionsDocument18 pagesIit-Examination Paper-2009 Code 4 Paper 2: InstructionspoulasNo ratings yet
- Iit Jee 2010 Part 2Document20 pagesIit Jee 2010 Part 2premsempireNo ratings yet
- I Am Sharing 'Assignment-3 Organic' With YouDocument28 pagesI Am Sharing 'Assignment-3 Organic' With YouKriti GargNo ratings yet
- 13 DPP 04P Elimination Excel 1664524624713Document6 pages13 DPP 04P Elimination Excel 1664524624713Jatin SindhwaniNo ratings yet
- GRP #12Document4 pagesGRP #12rajkaran3765No ratings yet
- 12th Chemistry Paper - I (Question Paper)Document4 pages12th Chemistry Paper - I (Question Paper)shaurya7rNo ratings yet
- Answer Advanced Assignment 02 Organic ChemistryDocument2 pagesAnswer Advanced Assignment 02 Organic ChemistryIshita AgarwalNo ratings yet
- Chemistry: Practice ProblemsDocument2 pagesChemistry: Practice ProblemsAnonymous vRpzQ2BLNo ratings yet
- Assignment 07 ON CARBONYL COMPOUNDSDocument2 pagesAssignment 07 ON CARBONYL COMPOUNDSIshita AgarwalNo ratings yet
- Solution Manual for The Elements of Polymer Science and EngineeringFrom EverandSolution Manual for The Elements of Polymer Science and EngineeringRating: 4 out of 5 stars4/5 (3)
- 5.1 Synthesis of A Haloalkane Risk AssessmentDocument4 pages5.1 Synthesis of A Haloalkane Risk AssessmentAdiNo ratings yet
- Chemistry 5Document23 pagesChemistry 5Suman DasNo ratings yet
- Diesel Exhaust FluidDocument7 pagesDiesel Exhaust FluidCISHAC FPNo ratings yet
- Dinitrol - TDS - Protecting - 447 - GB-2L70F (32,38 e 39)Document2 pagesDinitrol - TDS - Protecting - 447 - GB-2L70F (32,38 e 39)Dewton EspíndolaNo ratings yet
- 20-04-2024 - SR C-120 - Jee-Mains - WTM-30 - Q PaperDocument16 pages20-04-2024 - SR C-120 - Jee-Mains - WTM-30 - Q Paperbsahil2007No ratings yet
- Ddu Hardik Report DduDocument62 pagesDdu Hardik Report DduharshtejashprajapatiNo ratings yet
- Msds-Hansa Ese 6960Document6 pagesMsds-Hansa Ese 6960bua.t siampro.netNo ratings yet
- Aldehydes and Ketones - JEE Main 2024 January Question Bank - MathonGoDocument6 pagesAldehydes and Ketones - JEE Main 2024 January Question Bank - MathonGoRoshan AhamedNo ratings yet
- Periodic Table WorksheetDocument1 pagePeriodic Table Worksheettanishknandal2009No ratings yet
- Edmeston SX Welding Recommendations - Rev - 02-MO Okt 2020Document4 pagesEdmeston SX Welding Recommendations - Rev - 02-MO Okt 2020Yahya Kharis Abidul AzizNo ratings yet
- Influence of Coke Calcining Level On Anode Real Density LC and OtherDocument6 pagesInfluence of Coke Calcining Level On Anode Real Density LC and OtherSudhir PatelNo ratings yet
- Physical Metallurgy Vol. 1 2 y 3Document55 pagesPhysical Metallurgy Vol. 1 2 y 3veronicaNo ratings yet
- S1.4.4 Empirical Formula Past Paper Problems (Questions)Document4 pagesS1.4.4 Empirical Formula Past Paper Problems (Questions)mariaar281106No ratings yet
- C11 - Unit 1 HomeworkDocument24 pagesC11 - Unit 1 HomeworkMapleSkyNo ratings yet
- Unit 1 Electrochemistry - REVISION 2024Document28 pagesUnit 1 Electrochemistry - REVISION 2024handungeselmaNo ratings yet
- Literature Review On ZeolitesDocument5 pagesLiterature Review On Zeolitesafmzsbnbobbgke100% (2)
- Milady Foundations Chapter-06 ChemistryDocument54 pagesMilady Foundations Chapter-06 ChemistryKathleen KathleenNo ratings yet
- Cot 1 - Factors Affecting Solubility - First Quarter - 2223Document69 pagesCot 1 - Factors Affecting Solubility - First Quarter - 2223Fatima Abacan ReyesNo ratings yet
- Detailed Notes - Topic 8 Energetics I - Edexcel Chemistry A-LevelDocument8 pagesDetailed Notes - Topic 8 Energetics I - Edexcel Chemistry A-LeveleleinaleepileNo ratings yet
- GROUP 1 - CHE 026L LAS 9 - Chemical ReactionsDocument7 pagesGROUP 1 - CHE 026L LAS 9 - Chemical ReactionsShiena MosquedaNo ratings yet
- Test - 1Document9 pagesTest - 1zoyakhan.rkeNo ratings yet
- Organic Chemistry Some Basic Principles and TechniquesDocument139 pagesOrganic Chemistry Some Basic Principles and TechniquesSanjana KumariNo ratings yet
- Chemistry Fourteenth 14Th 2022 Jason Overby Full Chapter PDF ScribdDocument67 pagesChemistry Fourteenth 14Th 2022 Jason Overby Full Chapter PDF Scribdamanda.swarr169100% (5)
- 2022 Hsiao, High Retention Supercapacitors Using Carbon Nanomaterials-Iron Oxide-Nickel-IronDocument14 pages2022 Hsiao, High Retention Supercapacitors Using Carbon Nanomaterials-Iron Oxide-Nickel-Ironheri rustamajiNo ratings yet
- HiBallast Operational TipsDocument23 pagesHiBallast Operational TipsPopa CristiNo ratings yet
- Chemistry Ecet by AdithyaDocument125 pagesChemistry Ecet by AdithyaKUNUSOTH SAIKUMARNo ratings yet
- Solvent-Free Methods in NanocatalysisDocument347 pagesSolvent-Free Methods in NanocatalysisojodeimprentaNo ratings yet
- Polymer Nanocomposite FoamsDocument20 pagesPolymer Nanocomposite FoamsNguyễn KhánhNo ratings yet
- Advances in Synthesis Gas Methods Technologies and Applications Volume 4 Syngas Process Modelling and Apparatus Simulation Mohammad Reza Rahimpour Full ChapterDocument68 pagesAdvances in Synthesis Gas Methods Technologies and Applications Volume 4 Syngas Process Modelling and Apparatus Simulation Mohammad Reza Rahimpour Full Chapterteresa.vanhorn907100% (4)
- pdf2 2022Document71 pagespdf2 2022Джавахир ГасымоваNo ratings yet