Professional Documents
Culture Documents
Birimler Tablosu
Birimler Tablosu
Uploaded by
o_f_tOriginal Title
Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
Birimler Tablosu
Birimler Tablosu
Uploaded by
o_f_tCopyright:
Available Formats
YRD.DO.DR.
ATLLA EVCN
A.K.. MHENDSLK FAKLTES
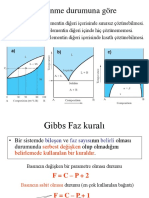
BRMLER TABLOSU
Fiziksel Byklk
Aydnlanma iddeti
Basn
Dipol moment
Elektrik miktar, yk
Elektriksel diren
Elektriksel potansiyel
Elektriksel sa
Enerji ak, s ak
Enerji, i, s miktar
Frekans
Ik iddeti
ndklenme
Kuvvet
Magnetik ak
Birim
Lux
Pascal
Debye
Coulomb
Ohm
Volt
Farad
Watt
Joule
Hertz
Lumen
Henry
Newton
Weber
Fiziksel Byklk
Asal hz
Alan
Aydnlanma younluu
Basn
Dinamik viskozite
Elektriksel alan
Hacim
Hz
vme
Kinematik viskozite
Manyetik alan iddeti
Younluk
SI Birimi
Radyant bl saniye
Metrekare
Candela bl metrekare
Mewton bl metrekare
Newton saniye bl metrekare
Volt bl metre
Metrekp
Metre bl saniye
Metre bl saniyekare
Metrekare bl saniye
Amper bl metre
Kilogram bl metrekp
Fiziksel Byklk
Uzunluk
Alan
Kuvvet
Basn
Basn
Dinamik Viskozite
Kinematik Viskozite
Radyoaktiflik
Enerji
Enerji
Birimin smi
Angstrm
Barn
Dyn (gr.cm.s-2)
Atm
Torr (1 mmHg)
Poise (gr.cm-1.s-1)
Stokes
Curie
erg (gr.cm2.s-2)
Kalori
YRD.DO.DR. ATLLA EVCN
Birim Simgesi
Ix
Pa
D
C
V
F
W
J
Hz
Im
H
N
Wb
Birimin Tanm
cd.sr.m-2
Kg.m-1.s-2 = N.m-2
C.mm. = A.m.s
A.s
Kg.m2.s-3.A-2 = V.A-1
Kg.m2.s-3.A-1 = J.A-1.s-1
A2.s4.kg-1.m-2 = A.s.V-1
Kg.m2.s-3 = J.s-1
Kg.m2.s-2
s-1
cd.sr
2 -2 -2
Kg.m .s .A = V.s.A-1
Kg.m.s-2 = J.m-1
Kg.m2.s-2.A-1 = V.s
Birimin Simgesi
Rad.s-1
m2
cd.m-2
N.m-2
N.s.m-2 = Pascal .s
V.m-1
m3
m.s-1
m.s-2
m2.s-1
A.m-1
Kg.m-3
Birimin Tanm
10-10 m
10-25 m2
10-5 N
101325 kN.m-2
133322 N.m-2
10-1 kg.m-1.s-1
104 m2.s-1
37.109 s-1
10-7 J
4.184 J
A.K.. MHENDSLK FAKLTES
1
YRD.DO.DR. ATLLA EVCN
A.K.. MHENDSLK FAKLTES
Basn Birimleri evrim Tablosu
N.m-2 = Pa
-2
N.m = Pa
Bar
At
Atm
Torr
Psi
Bar
-5
1
105
0,980665.105
1,01325.105
133,3224
6894,7
10
1
0,980625
1,01325
1,3332.10-3
0,0689487
at
1,0197.10-5
1,019716
1
1,033227
1,3595.10-3
0,070305
Atm
0,986923.10-5
0,986923
0,967841
1
1,3158.10-3
0,068046
Torr
7,50062.10-3
750,062
735,559
760
1
51,715
psi
1,4504.10-4
14,504
14,2235
14,696
0,019337
1
Enerji Birimleri evrim Tablosu
J
1
Jolue
cal
cm3.atm
BTU
-4
0,2390
9,488.10
KWh
9,869
2,7778.10
-6
erg
18
10
19
6,20.10
Cal
4,184
3,96340.10
41,293
1,162.10
2,61.10
4,19.107
BTU
1055,66
252,309
1,04186.104
293,24.10-6
6,545.1021
1,056.1010
0,024217
9,59826.10-5
28,15.10-9
6,282.1017
1,0133.106
cm3.atm 0,101325
-3
EV
-7
KWh
3,6.106
8,606.105
3,410.103
35,52.106
2,232.1025
3,6.1013
EV
1,60.10-19
3,83.10-20
1,528.10-22
1,592.10-18
4,48.10-26
1,60.10-12
Erg
10-7
2,39.10-8
9,4697.10-11
9,868.10-11
2,778.10-14
6,24.1011
G Birimleri evrim Tablosu
HP
kpm.s-1
Ibf.ft.s-1
Btu.hr-1
kcal.hr-1
HP
746
75
550
2545
632,61
1,3405.10-3
0,1019716
0,7373
3,412
0,8603
Kpm.s-1
0,013333
9,80665
7,33315
33,9325
8,4345
Ibf.ft.s-1
1,8182.10-3
1,3562
0,13637
4,6273
1,16682
Btu.hr-1
3,9293.10-4
0,4146
29,47.10-3
0,2161
0,252139
Kcal.hr-1
1,5808.10-3
1,16264
0,11856
0,857
3,966
Scaklk Birimleri evrim Tablosu
Fahrenheit (F)
Rankin (R)
Celcius (C)
Scaklk (t)
Kelvin (K)
(tR) 459,69
1,8.(tC) + 32
(tF) + 459,69
1,8.(tC) + 491,,69 1,8.(tK)
(tF 32).5/9
5/9(tR) 273
(tK) - 273
(tF 32).5/9+273 5/9.(tR)
(tC) + 273
YRD.DO.DR. ATLLA EVCN
1,8.(tK 273)+32
A.K.. MHENDSLK FAKLTES
2
YRD.DO.DR. ATLLA EVCN
A.K.. MHENDSLK FAKLTES
Sabitler
Fiziksel sabit
Atomik Ktle Birim
Avagadro Says
Bohr Yarap
Boltzman Sabiti
Debye Sabiti
Elektronun Ktlesi
Elektronun Yk
Faraday Sabiti
Ik Hz
deal Gaz Hacmi
deal Gaz Sabiti
deal Gaz Sabiti
deal Gaz Sabiti
deal Gaz Sabiti
deal Gaz Sabiti
deal Gaz Sabiti
deal Gaz Sabiti
deal Gaz Sabiti
deal Gaz Sabiti
Ntronun Ktlesi
Pi () says
Planck Sabiti
Protonun Ktlesi
Rydberg Sabiti
Yerekimi ivmesi
Simge
u
N
ao
k
D
me
qe
F
c
V
R
R
R
R
R
R
R
R
R
mn
h
mp
R
g
arpan n ek ismi
Sembol
Deeri
1,66056.10 kg
6,0221367.1023 mol-1
5,29177.10-11 m
1,38066.10-23 J.K-1
3,33564.10-30 C.m
9,11.10-31 kg
1,62.10-19 C
9,6487.104 C.mol-1
2,9979.108 m.s-1
22,4136 L
82,06 atm.cm3.mol-1.K-1
1,987 cal.mol-1.K-1
1,987 Btu.Ibmol-1.R-1
1546 (Ibf.ft-2).ft3.Ibmol-1.R-1
1,314 atm.ft3.Ibmol-1.K-1
0,7302 atm. ft3.Ibmol-1.R-1
10,73 psi.ft3.Ibmol-1.R-1
8,31434 J.mol-1.K-1
0,08206 atm.L.mol-1.K-1
1,67495.10-27 kg
3,1415926536
6,62618.10-34 J.s
1,67265.10-27 kg
1,0968.105 cm-1
9,80665 m.s-2
-27
arpan n ek ismi
Sembol
10-1
Desi
101
Deka
da
10-2
Santi
102
Hekto
10-3
Mili
103
Kilo
-6
10
Mikro
10
Mega
10-9
Nano
109
Giga
10-12
Piko
1012
Tera
10-15
Femto
1015
Peta
10-18
Atto
1018
Ekza
YRD.DO.DR. ATLLA EVCN
A.K.. MHENDSLK FAKLTES
3
YRD.DO.DR. ATLLA EVCN
A.K.. MHENDSLK FAKLTES
Greek Alfabesi
Sembol
A
smi
Alfa
Beta
Gama
Delta
Epsilon
Zeta
Eta
Teta
Iota
Kappa
Lamda
M
Sembol
smi
N
N
Ksi
O
o Omikron
Pi
Ro
Sigma
Tau
Upsilon
Fi
Psi
Omega
Acre
Dnm Faktrleri
= 4046,85 m2
Acre
cP (santipuaz)
= 1.10-3 kg.m-1.s-1
= 43560 ft2
cm3
= 2,64172.10-4 gal
Atm
= 1,01325.105 N.m-2
cP
= 2,4191 Ib.ft-1.h-1
Atm
= 14,696 Ibf.in-2
cP
= 6,7197.10-4 Ib.ft-1.s-1
Barrel
= 5,6146 ft3
cSt (santistoks) = 1.10-6 m2.s-1
Barrel
= 42 gal
ft
= 0,3048 m
Barrel
= 0,15899 m3
ft-Ibf
= 1,2851.10-3 Btu
Bar
= 1.105 N.m-2
ft-Ibf
= 0,32383 cal
Bar
= 14,5040 Ibf.in-2
ft-Ibf
= 1,35582 J
Btu
= 251,996 cal
ft-Ibf.s-1
= 4,6262 Btu.h-1
Btu
= 778,17 ft-Ibf
ft-Ibf.s-1
= 1,81818.10-3 hp
Btu
= 1055,06 J
ft2.h-1
= 2,581.10-5 m2.s-1
Btu
= 2,9307.10-4 kWh
ft2.h-1
= 0,2851 cm2.s-1
Btu.Ib-1
= 0,55556 cal.gr-1
ft3
= 2,831684.104 cm3
Btu.Ib-1.F-1
= 1 cal.gr-1.C-1
ft3
= 7,48052 gal
-2
= 28,31684 L
= 2,71948 Btu
-2
-1
Btu.ft .h
Cal
= 3,1546 W.m
= 4,184 J
ft
ft -atm
Cm
= 0,39370 in
ft -atm
= 685,29 cal
Cm
= 0,0328084 ft
ft3-atm
= 2,8692.103 J
cm3
= 3,531467.10-5 ft3
ft3.s-1
= 448,83 gal.min-1
YRD.DO.DR. ATLLA EVCN
A.K.. MHENDSLK FAKLTES
4
YRD.DO.DR. ATLLA EVCN
A.K.. MHENDSLK FAKLTES
Gal
= 0,13368 ft3
Ibmol.ft-2.h-1
= 1,3652.10-4 gmol.cm-2.s-1
Bar
= 106 dyn.cm-2
atm
= 1,033228 kp.cm-2
Btu.ft.h-1.ft-2.F-1
= 4,134.10-3 cal.cm.s-1.cm-1C-1
Btu.ft.h-1.ft-2.F-1 = 0,01731 W.m.h-1.m-2.C-1
Btu.ft.h-1.ft-2.F-1
= 1,488 kcal.m.h-1.m-2.C-1
oz
= 28,35 gr
Btu.h-1.ft-2
= 7,535.10-5 cal.s-1.cm-2
Btu.h-1.ft-2
= 3,154.10-4 W.cm-2
Btu.h-1.ft-2.F-1
= 1,355.10-4 cal.s-1.cm-2.C-1
Btu.h-1.ft-2.F-1
= 5,678.10-4 W.h-1.m-2.C-1
Btu.h-1.ft-2.F-1
= 4,882 kcal.h-1.m-2.C-1
cal.s-1.cm-2.C-1
= 4,187 W.cm-2.C-1
Gal
= 231 in3
= 3,280840 ft
= 60 min
= 3600 s
Hp
= 2544,43 Btu.h-1
= 0,74570 kW
Hp
N.m
= 264,17 gal
= 1.10-5 dyn
= 0,22481 lbf
-2
= 1,4498.10-4 lbf.in-2
= 0,45359237 kg
kcal
= 427 kpm
-3
= 16,018 kg.m
kpm
= 9,807 J
Ib.ft-3
= 0,016018 gr.cm-3
= 100 cP
Ibf.in-2
= 6,89473.103 N.m2
cP
= 0,01 gr.cm-1.s-1
Ibmol.ft-2.h-1
= 1,3652.10-3 kgmol.m-2.s-1
Stoke
= 1.104 m2.s-1
= 2,54 cm
ton
= 1000 kg
in3
= 16,3871 cm3
ton
= 2204,6 lb
= 1.107 erg
yd
= 3 ft
= 0,73756 ft-Ibf
yd
= 0,9144 m
Kg
= 2,20462 Ib
mil
= 5280 ft
KW
= 1,36 BG
Pascal
= 10 dyn.cm-2
KWh
km
= 0,6214 mil
= 3412,1 Btu
= 1.103 m3
L.atm
= 4,187 cal
Lb
= 16 ons
L.atm
= 9,604.10-2 Btu
Yerekimi ivmesi = 9,80665 m.s-2
m3
= 35,3147 ft3
Yerekimi Sabiti = 6,673.10-11 N.m2.kg-1
= 39,3701 in
Ib
Ib.ft
-3
YRD.DO.DR. ATLLA EVCN
A.K.. MHENDSLK FAKLTES
5
YRD.DO.DR. ATLLA EVCN
A.K.. MHENDSLK FAKLTES
GEREK GAZ HAL DENKLEMLER
a.n 2
P + 2 .(v n.b) = n.R.T
V
Van Der Waals
Redlich Kwong
a.n 2
P +
.(V n.b) = n.R.T
T .V .(V + n.b)
R.T
a
P=
Vm b T .Vm 2
Berthelot
Dieterici
P.e
n.a
R .T .V
.(V n.b) = n.R.T
c
A
RT
+
(1
)(
v
B)
v
vT 3
v2
a
A = A O (1 )
v
b
B = B O (1 )
v
P =
BeattieBridgeman
YRD.DO.DR. ATLLA EVCN
A.K.. MHENDSLK FAKLTES
6
YRD.DO.DR. ATLLA EVCN
YRD.DO.DR. ATLLA EVCN
A.K.. MHENDSLK FAKLTES
A.K.. MHENDSLK FAKLTES
7
YRD.DO.DR. ATLLA EVCN
A.K.. MHENDSLK FAKLTES
Tablo 1.2. Virial Sabitleri iin denklemler
B(T)
Van der Waals
Redlich-Kwong
Berthelot
D(T)
b2
b3
a
R.T
a
R.T
9.R.Tk
128.Pk
C(T)
a.b
b +
2
3 2
6.Tk 2
1 2
T
R.T
b
3
3 2
a.b 2
R.T
3 2
Tablo 1.4. Kritik Sabitlerden Hal Parametreleri Denklemleri
Van der Waals
2
Redlich-Kwong
R 2 .Tk
0,42748.
Pk
27.R 2 .Tk
64.Pk
R.Tk
8.Pk
Vk
3.b
3,847.b
3/8
1/3
zk =
Pk .Vk
R.Tk
YRD.DO.DR. ATLLA EVCN
0,086640.
R.Tk
Pk
A.K.. MHENDSLK FAKLTES
8
YRD.DO.DR. ATLLA EVCN
YRD.DO.DR. ATLLA EVCN
A.K.. MHENDSLK FAKLTES
A.K.. MHENDSLK FAKLTES
9
You might also like
- Isıl Işlemler Final 2017Document1 pageIsıl Işlemler Final 2017MuhittinNo ratings yet
- Halilcan Yıldırım 18-159-003 Isıl İşlem Vize CevaplarıDocument6 pagesHalilcan Yıldırım 18-159-003 Isıl İşlem Vize CevaplarıHalilcan YildirimNo ratings yet
- TOZ METALURJİSİ ÇALIŞMA SORULARI 2015 2016 YRD - DOC. YUNUS TÜREN - Karabük Üniversitesi METALURJİDocument7 pagesTOZ METALURJİSİ ÇALIŞMA SORULARI 2015 2016 YRD - DOC. YUNUS TÜREN - Karabük Üniversitesi METALURJİK.SİNEM YAYLANo ratings yet
- Isıl İşlem - 2012 Vize Soruları Ve Cevap AnahtarıDocument3 pagesIsıl İşlem - 2012 Vize Soruları Ve Cevap AnahtarıdownloadmonsterNo ratings yet
- NRL YíntemDocument45 pagesNRL YíntemCako Can100% (2)
- EY-50X CalısmaSoruları Yalıtım r2 40Document5 pagesEY-50X CalısmaSoruları Yalıtım r2 40Facecaylak İngNo ratings yet
- Isıl Işlem SoruDocument2 pagesIsıl Işlem SoruBilim KapsülüNo ratings yet
- Bolum 07 - DovmeDocument60 pagesBolum 07 - DovmeBurla KasimNo ratings yet
- Malzbil Soru Sunumu 19.12.2018Document38 pagesMalzbil Soru Sunumu 19.12.2018Burak ÖlmezNo ratings yet
- Kisa Sinav Calisma SorulariDocument93 pagesKisa Sinav Calisma SorulariHarun CücenNo ratings yet
- Fazlar Demir C TTTDocument176 pagesFazlar Demir C TTTRecep ÇETİNKAYANo ratings yet
- Östemperleme Isıl İşlemiDocument8 pagesÖstemperleme Isıl İşlemiFazlı Fatih MelemezNo ratings yet
- Sertlik Olcme YontemleriDocument42 pagesSertlik Olcme YontemleriOguz GirdapNo ratings yet
- Termal Analiz Yöntemleri 2Document3 pagesTermal Analiz Yöntemleri 2anon_280239516No ratings yet
- Fiziksel Metalurji - Toparlanma Ve RekristalizasyonDocument87 pagesFiziksel Metalurji - Toparlanma Ve RekristalizasyonAdil KocaosmanlarNo ratings yet
- Toz MetalürjisiDocument74 pagesToz MetalürjisiFerhat YlzNo ratings yet
- Metali̇k Profi̇lleri̇n Haddeleme İşlemi̇Document10 pagesMetali̇k Profi̇lleri̇n Haddeleme İşlemi̇Cihan AskinNo ratings yet
- Malzeme II 2020-2021 Bahar Dönemi Vize Sınavı SorularıDocument1 pageMalzeme II 2020-2021 Bahar Dönemi Vize Sınavı Sorularıziya deniz kayaNo ratings yet
- Faz 2Document34 pagesFaz 2aasdcdcNo ratings yet
- Astm TaneDocument5 pagesAstm Tanenecdet_karagülleNo ratings yet
- 03 Hafta Ötektik BileşimDocument35 pages03 Hafta Ötektik BileşimElif ArancıNo ratings yet
- Mb-Malzemeni̇n Mekani̇k Özelli̇kleri̇Document116 pagesMb-Malzemeni̇n Mekani̇k Özelli̇kleri̇Hikmet ÇakmakçıNo ratings yet
- Fiziksel Metalurji Sunu 1Document66 pagesFiziksel Metalurji Sunu 1Ahmet Malik Aşgın100% (2)
- Celigin Isil Isleminin Teorik Ve Pratik EsaslariDocument231 pagesCeligin Isil Isleminin Teorik Ve Pratik Esaslarihyılmaz_42No ratings yet
- KarbürlemeDocument7 pagesKarbürlemeOzan Pekel100% (1)
- Kristal Malzemelerin Dayanimlarinin ArtirilmasiDocument28 pagesKristal Malzemelerin Dayanimlarinin ArtirilmasisnngkcNo ratings yet
- Isıl Işlem SorularıDocument6 pagesIsıl Işlem SorularıaasdcdcNo ratings yet
- Fiziksel Metalurji II Hafta SunumDocument33 pagesFiziksel Metalurji II Hafta Sunumaliaga15No ratings yet
- Demir Dışı Metal Üretimi FinalDocument15 pagesDemir Dışı Metal Üretimi FinalaasdcdcNo ratings yet
- Makel VizeDocument4 pagesMakel VizeAyberk AydogumuNo ratings yet
- Punta Kaynaği - 135 KöşeDocument1 pagePunta Kaynaği - 135 KöşeNazım YıldırımNo ratings yet
- Mukavemet 2 Final 2013 Soru Ve CozumleriDocument8 pagesMukavemet 2 Final 2013 Soru Ve CozumleriCeyhun KuzuNo ratings yet
- Homojen ÇekirdeklenmeDocument9 pagesHomojen ÇekirdeklenmeGökçe DunluNo ratings yet
- Ders Notu 1 TribolojiDocument51 pagesDers Notu 1 TribolojiwwedatcanNo ratings yet
- Metalografik Muayene Ve Sertlik DeneyiDocument9 pagesMetalografik Muayene Ve Sertlik DeneyiSamet ZümbülNo ratings yet
- Fiziksel Metalurji I Yayınım (Difüzyon)Document67 pagesFiziksel Metalurji I Yayınım (Difüzyon)kerasus3530% (1)
- Ta N Mvize CevaplarDocument2 pagesTa N Mvize CevaplarMehmet IşıkNo ratings yet
- Isıl İşlem Deney RaporuDocument8 pagesIsıl İşlem Deney RaporuAhmet TozanNo ratings yet
- Malzeme Muayenesi 5229 PDFDocument51 pagesMalzeme Muayenesi 5229 PDFVolkanKaanÇiçekNo ratings yet
- Fiziksel Metalurji 7 - HaftaDocument1 pageFiziksel Metalurji 7 - HaftajosephbelieveNo ratings yet
- Isıl İşlemlerDocument29 pagesIsıl İşlemlerahmat Sa50% (2)
- Mukavemet - Ii.2007 2008.finalDocument8 pagesMukavemet - Ii.2007 2008.finalCeyhun KuzuNo ratings yet
- Burulma Problemleri̇Document13 pagesBurulma Problemleri̇canNo ratings yet
- Teknik Egitim El KitabiDocument55 pagesTeknik Egitim El KitabiHuseyin HamurcuNo ratings yet
- Kocaeli̇ Üni̇ Muk2 Fi̇nal SoruvecevaplariDocument9 pagesKocaeli̇ Üni̇ Muk2 Fi̇nal SoruvecevaplariCem IşıldakNo ratings yet
- 1) Piknometre Deney Föyü - EEDocument4 pages1) Piknometre Deney Föyü - EEPınarselen TuranNo ratings yet
- TASIMDA MALZEME SEyOMDocument280 pagesTASIMDA MALZEME SEyOMkerasus353No ratings yet
- Takım Malzemeleri Ve SınıflandırılmasıDocument26 pagesTakım Malzemeleri Ve SınıflandırılmasıSedat GüvenNo ratings yet
- Faz Di̇yagramlariDocument100 pagesFaz Di̇yagramlariTunahan BasgedikNo ratings yet
- Malzeme BilgisiDocument88 pagesMalzeme Bilgisiedgar pulgarNo ratings yet
- Ark Kaynağı Elektrod Standartlari Oerlikon Yayınlar, Burhan Oğuz (Kısa Bilgi)Document6 pagesArk Kaynağı Elektrod Standartlari Oerlikon Yayınlar, Burhan Oğuz (Kısa Bilgi)Kadir KoyluNo ratings yet
- Oyal MetalDocument1 pageOyal MetalUfuk KıvançNo ratings yet
- Termal AnalizDocument2 pagesTermal Analizemad hayek100% (1)
- Met. Ve Malz. Muh. Giris BirimlerDocument18 pagesMet. Ve Malz. Muh. Giris BirimleremirterNo ratings yet
- Boyutlar BirimlerDocument7 pagesBoyutlar BirimlersercanerigurNo ratings yet
- Fiziksel SabitlerDocument11 pagesFiziksel SabitlerMehmet CeppiNo ratings yet
- Hafta 1 DersDocument20 pagesHafta 1 DersAlperen GökçeNo ratings yet
- D09u3 Elasti̇k Zem Otr Surk Temel 2020Document5 pagesD09u3 Elasti̇k Zem Otr Surk Temel 2020Alper DumanNo ratings yet
- Boyutlar BirimlerDocument28 pagesBoyutlar BirimlerMehmet KoçNo ratings yet