Professional Documents
Culture Documents
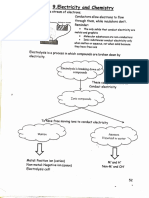
TOPIC Products Obtained by Electrolysis
Uploaded by
vivek dave0 ratings0% found this document useful (0 votes)
7 views2 pagesCopyright
© © All Rights Reserved
Available Formats
PDF, TXT or read online from Scribd
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
© All Rights Reserved
Available Formats
Download as PDF, TXT or read online from Scribd
0 ratings0% found this document useful (0 votes)
7 views2 pagesTOPIC Products Obtained by Electrolysis
Uploaded by
vivek daveCopyright:
© All Rights Reserved
Available Formats
Download as PDF, TXT or read online from Scribd
You are on page 1of 2
Products Obtained by Electrolysis
Sr. Material for Anode Cathode Results Reactions
No. Electrolysis
1. fused NaCl graphite steel • sodium metal on Reaction:
cathode NaCl→ Na+ +Cl−
• chlorine gas on
anode. Reaction at anode: oxidation
graphite :
-
Cl(l) →1/2 Cl2(g) + e−
Reaction at cathode : reduction
steel :
Na+(Aq ) + e− →Na(s)
2. graphite graphite • only water is
Dilute aqueous electrolysed
solution of NaCl • hydrogen gas at
cathode
• oxygen gas at anode
3. graphite graphite • Cl2 gas is produced
Concentrated at anode
aqueous solution • H2 gas is produced
of NaCl at cathode
• NaOH is produced in
the solution
4. copper copper • copper electrode is
CuSO4 solution active electrode
(i) using copper • Copper anode
electrodes undergoes, oxidation
during electrolysis.
• With the progress of
electrolysis, mass of
Products Obtained by Electrolysis
anode decrease and
mass of cathode
increases.
• Impure Cu is
purified by
electrolysis method
• Impurities of
inactive metals like
Au, Pt present in
impure copper
anode do not
• undergo oxidation.
These impurities fall
on the bottom of the
electrode when they
become free from
anode and is called
anode mud.
5. CuSO4 solution Pt or Pt or • water undergoes
(ii)using inactive graphite graphite oxidation, giving
electrodes inactive inactive oxygen at anode
electrod electrodes • Cu+2 ions undergo
es reduction at
cathode, they are
replaced by H+(aq)
• copper deposits on
cathode, oxygen
liberates at anode
• H2SO4 is produced
in the solution.
You might also like
- ElectrolysisDocument20 pagesElectrolysisGowriram RamNo ratings yet
- Voltaic Cell and Electrolytic Cell: by 5S3 Jocelyne LewDocument9 pagesVoltaic Cell and Electrolytic Cell: by 5S3 Jocelyne LewJocelyne LewNo ratings yet
- ElectroDocument48 pagesElectroMang friesNo ratings yet
- ElectrolyisDocument5 pagesElectrolyisMukeshNo ratings yet
- Electrometallurgy 3: Laval University, Quebec City, Canada Fathi - Habashi@arul - Ulaval.caDocument28 pagesElectrometallurgy 3: Laval University, Quebec City, Canada Fathi - Habashi@arul - Ulaval.caBasilia YulianiNo ratings yet
- Summary - ElectrolysisDocument7 pagesSummary - ElectrolysisKeertana SNNo ratings yet
- Groupings of Elements in The Periodic Table Group I A - The Alkali Metals NaDocument6 pagesGroupings of Elements in The Periodic Table Group I A - The Alkali Metals NaGlad YsNo ratings yet
- ELECTROCHEMISTRYDocument14 pagesELECTROCHEMISTRYmohamed komiNo ratings yet
- IGCSE CoordSci Electricity and Chemistry L2Document35 pagesIGCSE CoordSci Electricity and Chemistry L2dung luongtuanNo ratings yet
- Corrosion (All)Document59 pagesCorrosion (All)trongstaNo ratings yet
- Two More Uses of Electrolysis: When Electrodes Are Not InertDocument2 pagesTwo More Uses of Electrolysis: When Electrodes Are Not InertShahid Ur RehmanNo ratings yet
- ELECTROLYSIS Notes Condensed 2Document3 pagesELECTROLYSIS Notes Condensed 2Diya ShahNo ratings yet
- All About ElectrochemistryDocument23 pagesAll About ElectrochemistryROY JAIVIN A/L SANTHANA DAS MoeNo ratings yet
- Electrochemistry Ol Notes 2Document7 pagesElectrochemistry Ol Notes 2Ahmed SherifNo ratings yet
- 1.4 BI CompleteDocument9 pages1.4 BI CompleteRawid GoshanNo ratings yet
- CorrosionDocument25 pagesCorrosionmahmoud EissaNo ratings yet
- Electricity and Chemistry - Unit 4Document30 pagesElectricity and Chemistry - Unit 4Cloud WtafNo ratings yet
- Lec 10-13Document66 pagesLec 10-13BhushanNo ratings yet
- Experiment 2 - Electrolytic CellDocument8 pagesExperiment 2 - Electrolytic CellafifiNo ratings yet
- Chemistry 4 Electrolysis QP Level Cie For Class 10 11Document24 pagesChemistry 4 Electrolysis QP Level Cie For Class 10 11Maryam KhanNo ratings yet
- Corrosion MaterialDocument25 pagesCorrosion MaterialrahmadNo ratings yet
- C6 Book AnswersDocument4 pagesC6 Book AnswersvijahatNo ratings yet
- ElectroplatingDocument17 pagesElectroplatingShirlyn HeeNo ratings yet
- 8 ElectrolysisDocument15 pages8 ElectrolysisArmaanNo ratings yet
- Metals and Non MetalsDocument15 pagesMetals and Non Metalskebepef613No ratings yet
- Electrolysis O LevelDocument17 pagesElectrolysis O LevelInnocent EbilNo ratings yet
- C12 Electrochemistry IgcseDocument33 pagesC12 Electrochemistry IgcseAnna DixonNo ratings yet
- Corrosion & Its Control & Its ControlDocument28 pagesCorrosion & Its Control & Its ControlHiren KumarNo ratings yet
- Electrochemistry - EnglishDocument6 pagesElectrochemistry - Englishchamath p.s.a.d amakaraNo ratings yet
- Electrolysis of Aqeous Solutions (Copper Sulfate) PosterDocument1 pageElectrolysis of Aqeous Solutions (Copper Sulfate) Posternnilam1308No ratings yet
- Ch13 ElectrolysisDocument35 pagesCh13 ElectrolysishahaNo ratings yet
- Chapter 8 ElectrochemistryDocument3 pagesChapter 8 Electrochemistrysitinur qahirahNo ratings yet
- CSEC Chemistry - ElectrochemistryDocument7 pagesCSEC Chemistry - ElectrochemistryCornflakes Toasted100% (1)
- Application of Electrolytic Cells Lesson 11Document24 pagesApplication of Electrolytic Cells Lesson 11Rosmaini MohamadNo ratings yet
- ElectrochemistryDocument38 pagesElectrochemistryShannon SmithNo ratings yet
- Screenshot 2022-06-19 at 3.39.23 PMDocument47 pagesScreenshot 2022-06-19 at 3.39.23 PMWalaa AdelNo ratings yet
- Frequently Asked Questions in SPM Standard Answers: Chemistry SPM - Quick Review F4Document1 pageFrequently Asked Questions in SPM Standard Answers: Chemistry SPM - Quick Review F4Sze Hao CheahNo ratings yet
- ElectrolysisDocument22 pagesElectrolysisDebbie BerriesNo ratings yet
- Chapter 6b Electrolysis of Aqueous SolutionDocument16 pagesChapter 6b Electrolysis of Aqueous SolutionKavitha ThayagarajanNo ratings yet
- ElectrolysisDocument24 pagesElectrolysisstudent purposesNo ratings yet
- ElectrolysisDocument3 pagesElectrolysisMohit RawatNo ratings yet
- SGA MP 415 W6 - 1 Corrosion 1Document33 pagesSGA MP 415 W6 - 1 Corrosion 1Phillip WeyersNo ratings yet
- Chapter 4Document11 pagesChapter 4J.K HomerNo ratings yet
- Electrolysis NotesDocument8 pagesElectrolysis NotesJ Nyumah TengonghNo ratings yet
- Electrolysis Cheat Sheet: by ViaDocument2 pagesElectrolysis Cheat Sheet: by Viaaziz ahmadNo ratings yet
- Application of ElectrolysisDocument19 pagesApplication of Electrolysisazka shahidNo ratings yet
- Chemistry Grade 11 Chapter VIIIDocument44 pagesChemistry Grade 11 Chapter VIIIJ.K HomerNo ratings yet
- Electrolysis of Silver SulphateDocument5 pagesElectrolysis of Silver SulphateJackson_de_Roz_6005100% (1)
- Electrochemical ProcessesDocument19 pagesElectrochemical Processeshrishikeshdeka18No ratings yet
- ElectrolysisDocument20 pagesElectrolysisapi-381901275% (4)
- CHAPTER 7 9th CLASS Long QuestionsDocument12 pagesCHAPTER 7 9th CLASS Long Questionssomerakhan2023No ratings yet
- Chemistry - Notes Icse 10Document25 pagesChemistry - Notes Icse 10Suneet MohanNo ratings yet
- Study Material Class 10 Chapter 3 2017 PDFDocument10 pagesStudy Material Class 10 Chapter 3 2017 PDFKaran Pratap67% (3)
- ElectrochemistryDocument16 pagesElectrochemistryitsshaunboteNo ratings yet
- Pin Điện Hóa - Bùi Thị Thảo NguyênDocument68 pagesPin Điện Hóa - Bùi Thị Thảo NguyênĐặng Xuân ThắngNo ratings yet
- Electrolysis: of Molten Ionic CompoundsDocument16 pagesElectrolysis: of Molten Ionic CompoundsHafiz Abdul RehmanNo ratings yet
- Electricity ND ChemistryDocument23 pagesElectricity ND ChemistryFilza SiddiqNo ratings yet
- Analysis of the New Metals: Titanium, Zirconium, Hafnium, Niobium, Tantalum, Tungsten and Their AlloysFrom EverandAnalysis of the New Metals: Titanium, Zirconium, Hafnium, Niobium, Tantalum, Tungsten and Their AlloysNo ratings yet
- Organometallic Mechanisms and Catalysis: The Role of Reactive Intermediates in Organic ProcessesFrom EverandOrganometallic Mechanisms and Catalysis: The Role of Reactive Intermediates in Organic ProcessesNo ratings yet
- Preparation of Alkyl HalideDocument1 pagePreparation of Alkyl Halidevivek daveNo ratings yet
- Chemical KineticsDocument3 pagesChemical Kineticsvivek daveNo ratings yet
- Unit-III C PesticidesDocument11 pagesUnit-III C Pesticidesvivek daveNo ratings yet
- KimayaDocument4 pagesKimayavivek daveNo ratings yet
- Unit-Iii C PesticidesDocument11 pagesUnit-Iii C Pesticidesvivek daveNo ratings yet
- Nitro Compounds and Benzene Diazonium SaltDocument7 pagesNitro Compounds and Benzene Diazonium Saltvivek daveNo ratings yet
- SilicatesDocument7 pagesSilicatesvivek daveNo ratings yet
- (Section A) - Answer The Following For 1 Mark.Document6 pages(Section A) - Answer The Following For 1 Mark.vivek daveNo ratings yet
- Topic-0 Development of ChemistryDocument2 pagesTopic-0 Development of Chemistryvivek daveNo ratings yet
- Topic Faraday's Law and Electrolytic CellsDocument7 pagesTopic Faraday's Law and Electrolytic Cellsvivek daveNo ratings yet
- PAPER-307 Unit-Iii C: PesticidesDocument13 pagesPAPER-307 Unit-Iii C: Pesticidesvivek daveNo ratings yet
- Topic Faraday's Law and Electrolytic CellsDocument7 pagesTopic Faraday's Law and Electrolytic Cellsvivek daveNo ratings yet
- Solid State NotesDocument15 pagesSolid State Notesvivek daveNo ratings yet
- Topic Different Types of CellsDocument2 pagesTopic Different Types of Cellsvivek daveNo ratings yet
- Chapter 3 Electrochemistry Topic 1 Electrochemical Cell, Electrodes and E.M.F. SeriesDocument8 pagesChapter 3 Electrochemistry Topic 1 Electrochemical Cell, Electrodes and E.M.F. Seriesvivek daveNo ratings yet
- Topic Nernst Equation and Concentration CellDocument6 pagesTopic Nernst Equation and Concentration Cellvivek daveNo ratings yet
- Chapter 3 Electrochemistry Topic 1 Electrochemical Cell, Electrodes and E.M.F. SeriesDocument8 pagesChapter 3 Electrochemistry Topic 1 Electrochemical Cell, Electrodes and E.M.F. Seriesvivek daveNo ratings yet
- Chapter 3 Electrochemistry Topic ElectrochemistryDocument16 pagesChapter 3 Electrochemistry Topic Electrochemistryvivek daveNo ratings yet
- Factors Affecting SolubilityDocument5 pagesFactors Affecting Solubilityvivek daveNo ratings yet
- Topic Nernst Equation and Concentration CellDocument6 pagesTopic Nernst Equation and Concentration Cellvivek daveNo ratings yet
- Chapter 3 Electrochemistry Topic ElectrochemistryDocument16 pagesChapter 3 Electrochemistry Topic Electrochemistryvivek daveNo ratings yet
- Alkyl Halide Jee Neet PDFDocument3 pagesAlkyl Halide Jee Neet PDFvivek daveNo ratings yet
- Topic Different Types of CellsDocument2 pagesTopic Different Types of Cellsvivek daveNo ratings yet
- Hydrogen SensorDocument4 pagesHydrogen Sensorvivek daveNo ratings yet
- Write The Answer in One SentenceDocument2 pagesWrite The Answer in One Sentencevivek daveNo ratings yet
- Hydrogen SensorDocument4 pagesHydrogen Sensorvivek daveNo ratings yet
- IterDocument6 pagesItervivek daveNo ratings yet
- Scan 001Document5 pagesScan 001vivek daveNo ratings yet
- A Paradigm of Fragile Earth in Priestleys Bell JaDocument6 pagesA Paradigm of Fragile Earth in Priestleys Bell Ja2104007No ratings yet
- Bromocresol GreenDocument9 pagesBromocresol GreenNur ShahirahNo ratings yet
- (Review) Degradation of Chlorophyll During Processing of Green VegetablesDocument8 pages(Review) Degradation of Chlorophyll During Processing of Green VegetablesAndrés GómezNo ratings yet
- CH 4 EnergeticsDocument35 pagesCH 4 Energeticsthat guyNo ratings yet
- Heidolph Rotary Evaporators CatalogDocument15 pagesHeidolph Rotary Evaporators CatalogMuhammad AminNo ratings yet
- Unit 8Document25 pagesUnit 8Kinnari PatelNo ratings yet
- Lubi Permeate Pump DatasheetDocument2 pagesLubi Permeate Pump DatasheetVenkatesh SivaramanNo ratings yet
- Practical 2 RDTDocument20 pagesPractical 2 RDTDoraemon CuteNo ratings yet
- Ketenes, Allenes and Related Compounds - Volume 1 (1980) 1980 PDFDocument492 pagesKetenes, Allenes and Related Compounds - Volume 1 (1980) 1980 PDFhemberNo ratings yet
- College Notes Unit-1 Solid StateDocument24 pagesCollege Notes Unit-1 Solid StateRamanujam JNo ratings yet
- Voltatex t050 Cleaning Agent - DatasheetDocument2 pagesVoltatex t050 Cleaning Agent - DatasheetAlexandru CanturNo ratings yet
- Class X Science Practice Test Ak 2022-23Document15 pagesClass X Science Practice Test Ak 2022-23Tanish MehtaNo ratings yet
- Chemical Process MCQs PDFDocument72 pagesChemical Process MCQs PDFAman Kumar TiwariNo ratings yet
- Report of TitrationDocument7 pagesReport of TitrationSafwan BakrmanyNo ratings yet
- Line Symbols Service Code Work Breakdown Structure Valve Symbols Line SymbolsDocument346 pagesLine Symbols Service Code Work Breakdown Structure Valve Symbols Line SymbolsEnrique100% (1)
- 860 Moldable Polymer GasketingDocument2 pages860 Moldable Polymer GasketingprajanegaraNo ratings yet
- Cheese: Microbiology of Cheese: December 2016Document13 pagesCheese: Microbiology of Cheese: December 2016Anh MinhNo ratings yet
- Glossary of ForgingDocument46 pagesGlossary of ForgingBHAVESH JOSHINo ratings yet
- Colour Suture Material Colour: MersuturesDocument1 pageColour Suture Material Colour: MersuturesK.P. MOHITHRAJNo ratings yet
- ASTM D5186 App Note AromaticsDocument8 pagesASTM D5186 App Note Aromaticsmustak kadivarNo ratings yet
- Development of An Electrochemical Metal-Ion Biosensor Using Self-Assembled Peptide NanofibrilsDocument7 pagesDevelopment of An Electrochemical Metal-Ion Biosensor Using Self-Assembled Peptide NanofibrilsRenan Guilherme de Oliveira GuihNo ratings yet
- Distillation Formal Lab ReportDocument8 pagesDistillation Formal Lab ReportPeter Ickes100% (2)
- Effect of Using Potassium Bisulfate As Food Preservative Under Various ConditionsDocument15 pagesEffect of Using Potassium Bisulfate As Food Preservative Under Various Conditionsmihir khabiyaNo ratings yet
- Determining Integrity of Seals For Medical Packaging by Visual InspectionDocument4 pagesDetermining Integrity of Seals For Medical Packaging by Visual Inspectionรอคนบนฟ้า ส่งใครมาให้ สักคนNo ratings yet
- Kelvin Lau MWI +RRDocument18 pagesKelvin Lau MWI +RRUniversityJCNo ratings yet
- Series 200 Oven Family User's ManualDocument53 pagesSeries 200 Oven Family User's Manualrolo5000No ratings yet
- Mempelajari Sifat Minyak Sawit Dari Brondolan Pada Perlakuan Lama Sterilisasi Dan Jumlah Water Dillution Yang BerbedaDocument7 pagesMempelajari Sifat Minyak Sawit Dari Brondolan Pada Perlakuan Lama Sterilisasi Dan Jumlah Water Dillution Yang BerbedaAfrizal HasibuanNo ratings yet
- White Wax 50 G White Petrolatum 950 G To Make 1000 GDocument3 pagesWhite Wax 50 G White Petrolatum 950 G To Make 1000 GRoseanne Legaspi CasayuranNo ratings yet
- SCIENCE Most Important Questions (Prashant Kirad)Document40 pagesSCIENCE Most Important Questions (Prashant Kirad)aladdinvszafar100% (1)
- Cambridge IGCSE: CHEMISTRY 0620/42Document16 pagesCambridge IGCSE: CHEMISTRY 0620/42Renesh PatelNo ratings yet