Professional Documents
Culture Documents
Unit 3 Review
Uploaded by
819551Original Description:
Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
Unit 3 Review
Uploaded by
819551Copyright:
Available Formats
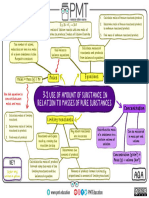
SCH3U0
Unit 3 Chemical Quantities - Review
Chapter 5
5.1 Solving for moles. What is a mole? What is a mole based on?
Know what a molecule, formula unit, atom, ion and Avogadro’s constant are.
n=N
NA
5.2 n=m To determine molar mass (M). Add up the elements in the compound from
M the atomic numbers on the periodic table.
• Atomic mass is the average mass in atomic mass units of one atom of the element.
Molar mass is the mass in grams of one mole of the element. (6.02 x 1023 atoms)
Chapter 6
6.1 Percentage Composition % of = mass (g) element x 100 molar mass (element) x 100
element mass (g) compound molar mass (compound)
OR
6.2 Empirical Formula 1. Assume % = 100 g (convert % to mass (g)) or use masses given.
2. Solve for moles of each element n= mass (g) For each
molar mass of element element in
the compound.
3. Divide by lowest moles to get a whole number ratio
(Use fractions if necessary)
6.2 Molecular formula Factor = Molecular molar mass multiply the factor by the empirical
Empirical molar mass formula to get the molecular formula.
6.2 Hydrates 1. Treat the anhydrous and water as two separate compounds.
2. Solve for moles of each.
3. Divide by lowest moles to solve for “X” number of water molecules per
formula unit.
To determine the mass of either the water portion or anhydrous portion of the
hydrate multiply the mass of the whole hydrate by the decimalized percent of the
portion you are solving for.
Chapter 7
7.1 Stoichiometry 4 Steps 1. Balance the equation
2. Solve for moles of given
3. Mole ratio of known → to moles of unknown
4. Solve for the unknown (mass or particles)
7.2 Limiting Reactant 4 Steps 1. Balance the equation
2. Solve for moles of ALL givens
3. Mole ratio of ALL givens → to moles of the SAME unknown
Determine the limiting reactant from the reactant that given the
smallest moles of product.
4. Solve for the unknown (mass or particles)
7.3 Percentage Yield Use stoichiometry to determine the theoretical yield % Yield = actual yield x 100
theoretical yield
Review Questions pg 339-343 1-15, 34-38
You might also like
- Mole ConceptDocument24 pagesMole ConceptRaju SinghNo ratings yet
- General Chemistry I Handout 6Document5 pagesGeneral Chemistry I Handout 6Roxan Oxima ClabriaNo ratings yet
- Grade 11 ChemistryDocument17 pagesGrade 11 ChemistryKevin George100% (1)
- Machine Learning AdvancedDocument12 pagesMachine Learning Advanceddhruvit100% (2)
- StoichiometryDocument63 pagesStoichiometryAlice KirklandNo ratings yet
- Chem 1 Percentage Composition and Chemical FormulasDocument10 pagesChem 1 Percentage Composition and Chemical FormulasCrizhae OconNo ratings yet
- Excel 2022 by Joseph ThompsonDocument207 pagesExcel 2022 by Joseph ThompsonquoazNo ratings yet
- Vacuum Engineering Calculations, Formulas, and Solved ExercisesFrom EverandVacuum Engineering Calculations, Formulas, and Solved ExercisesRating: 4.5 out of 5 stars4.5/5 (2)
- 6.3.6 Lab - Build A CI-CD Pipeline Using JenkinsDocument15 pages6.3.6 Lab - Build A CI-CD Pipeline Using JenkinsWilly DinataNo ratings yet
- Coxeter Graphs and Towers of Algebras, Goodman, de La Harpe and JonesDocument152 pagesCoxeter Graphs and Towers of Algebras, Goodman, de La Harpe and Jonesanon-494847100% (6)
- Mole Concept WorksheetDocument10 pagesMole Concept WorksheetNaman VermaNo ratings yet
- Lesson Plan in StoichiometryDocument7 pagesLesson Plan in StoichiometryLoreen Jane AbogadoNo ratings yet
- What You Need To Know About JavaScript (Ebook)Document66 pagesWhat You Need To Know About JavaScript (Ebook)buoncri100% (1)
- Lif Co BooksDocument8 pagesLif Co BooksAsok Kumar0% (1)
- 10.1 Science Notebook (Answer Key)Document5 pages10.1 Science Notebook (Answer Key)Black arab GaladimaNo ratings yet
- Stem3 ReviewerDocument27 pagesStem3 ReviewerantigajulsNo ratings yet
- CHEM NotesDocument4 pagesCHEM NotesShayne BonayonNo ratings yet
- Equal To Ar or MR of The Substance.: - Numerically This IsDocument2 pagesEqual To Ar or MR of The Substance.: - Numerically This IsM. Faaz JehangiriNo ratings yet
- CHM01 Co4 Lesson1 StoichiometryDocument2 pagesCHM01 Co4 Lesson1 Stoichiometrylucifer angelNo ratings yet
- StoichiometryDocument65 pagesStoichiometryAnonymous P1iMibNo ratings yet
- Chmstry G8th, U5 Calculation Based On FormulaDocument11 pagesChmstry G8th, U5 Calculation Based On Formulasisejeremiah5No ratings yet
- Chapter 3 Notes - StoichiometryDocument4 pagesChapter 3 Notes - StoichiometrypolickNo ratings yet
- 3.2. Use of Amount of Substance in Relation To Masses of Pure SubstancesDocument1 page3.2. Use of Amount of Substance in Relation To Masses of Pure Substancesameliarazaq12No ratings yet
- GEN-CHEM-1 G11 Module3 Q1W3 MODDocument15 pagesGEN-CHEM-1 G11 Module3 Q1W3 MODDaniel Corcino100% (1)
- Week12 Mole Student 2019Document35 pagesWeek12 Mole Student 2019api-4915646430% (1)
- 4 Molecular and Empirical FormulasDocument5 pages4 Molecular and Empirical FormulasCris CorsinoNo ratings yet
- Calculations in IB Chemistry SL and HL: Purpose The Calculation NotesDocument12 pagesCalculations in IB Chemistry SL and HL: Purpose The Calculation NotesCicy IrnaNo ratings yet
- Chapter 4 Calculations ANSWERSDocument2 pagesChapter 4 Calculations ANSWERSmollicksoham2907No ratings yet
- ch7 06 Chapter 3Document19 pagesch7 06 Chapter 3Tinenenji SinyindaNo ratings yet
- Mole Concept Percent Composition and Empirical and Molecular FormulaDocument2 pagesMole Concept Percent Composition and Empirical and Molecular Formuladestro bernardNo ratings yet
- Mole ConceptDocument25 pagesMole Conceptsrk78No ratings yet
- The Mole and Chemical Calculations: Pure ChemistryDocument2 pagesThe Mole and Chemical Calculations: Pure Chemistryyuxiang32No ratings yet
- Unit 9 - The Chemical BondDocument5 pagesUnit 9 - The Chemical BondjessieNo ratings yet
- 3chemistry 12Document23 pages3chemistry 12Faustino AbucayonNo ratings yet
- Mole Concept WorkbookDocument57 pagesMole Concept WorkbookBriana BartonNo ratings yet
- Topic 1 Stoichiometric Relationships PDFDocument7 pagesTopic 1 Stoichiometric Relationships PDFDiksha SinghNo ratings yet
- SyllabusDocument2 pagesSyllabusHarshil ChordiaNo ratings yet
- Chemistry SK 015: Amal Hayati Binti JamaliDocument69 pagesChemistry SK 015: Amal Hayati Binti JamaliDaniel LimNo ratings yet
- AP Chem1Document1 pageAP Chem1sassyhaitianNo ratings yet
- IB Chemistry SL and HLDocument108 pagesIB Chemistry SL and HLVed JoshiNo ratings yet
- Chemistry 4 Compounds and StiochimetryDocument7 pagesChemistry 4 Compounds and StiochimetryRosely PaquiteNo ratings yet
- G11ek3 Lectures in Gen Chem 1 Weeks 3 4lc 9 14Document3 pagesG11ek3 Lectures in Gen Chem 1 Weeks 3 4lc 9 14FMangonon, Jarrah Mae B.No ratings yet
- Summary of Sk016 Define/ State Calculation Remark: Consant Avogadro Particles of NumberDocument8 pagesSummary of Sk016 Define/ State Calculation Remark: Consant Avogadro Particles of NumbertimNo ratings yet
- Percent CompositionDocument2 pagesPercent CompositionKazuya 2302No ratings yet
- Summary of Units Chemistry Class XI.2022-23Document73 pagesSummary of Units Chemistry Class XI.2022-23shilpa07.nairNo ratings yet
- Tugas KimiaDocument2 pagesTugas KimiaAries RahmawatiNo ratings yet
- StoichiometryDocument40 pagesStoichiometryMariana Grace Ustang TafaibNo ratings yet
- Stoichiometry and SolutionsDocument17 pagesStoichiometry and SolutionsyoonglespianoNo ratings yet
- Empirical Formula, Molecular Formula, and Percentage CompositionDocument7 pagesEmpirical Formula, Molecular Formula, and Percentage CompositionPatrixiah Monicah Mareight BaronNo ratings yet
- Stoichiometry & Titration: Submitted byDocument14 pagesStoichiometry & Titration: Submitted byGezem GigantoNo ratings yet
- Complete Notes of Puc First YearDocument182 pagesComplete Notes of Puc First YeardanielakhilkumarNo ratings yet
- Physical Science Quantitative Aspects of Chemical ChangeDocument20 pagesPhysical Science Quantitative Aspects of Chemical ChangeTanaka ChirawuNo ratings yet
- Tools - For - Problem - Solving (Appendix B), R.K. Malik's Newton Classes PDFDocument48 pagesTools - For - Problem - Solving (Appendix B), R.K. Malik's Newton Classes PDFMoindavis DavisNo ratings yet
- Adv Chem Week 3Document13 pagesAdv Chem Week 3Venice Gwyn ChavezNo ratings yet
- Mole ProtocolDocument1 pageMole ProtocolMarin PesicNo ratings yet
- Chapter 9 - The Mole (Part-2)Document2 pagesChapter 9 - The Mole (Part-2)Yasser AliNo ratings yet
- Additional NotesDocument4 pagesAdditional NotesThalia Diosenne ArabesNo ratings yet
- CalculationsDocument6 pagesCalculationsAlvinNo ratings yet
- Chemistry: Abul FazalDocument51 pagesChemistry: Abul FazalRania ZahidNo ratings yet
- Stoichiometry: Stoichen ElementDocument55 pagesStoichiometry: Stoichen ElementKoonj LashariNo ratings yet
- Notes CH 5 Chemical Calculations 2022-1Document20 pagesNotes CH 5 Chemical Calculations 2022-1johhnylawrence924No ratings yet
- Chem Handout StoichDocument3 pagesChem Handout StoichAlexander DolinNo ratings yet
- General ChemistryDocument10 pagesGeneral Chemistryhehe xdNo ratings yet
- Amatac Report in ChemDocument58 pagesAmatac Report in ChemNica Rose GrozenNo ratings yet
- Mole and Mass RatiosDocument10 pagesMole and Mass RatiosJanne Lorraine Garcia-EleazarNo ratings yet
- Roman Collections 2016 PDFDocument66 pagesRoman Collections 2016 PDFRiekea Astika Putri GultomNo ratings yet
- UNIT3 Obj QuestionDocument32 pagesUNIT3 Obj QuestionSoumen MitraNo ratings yet
- Eckersley Interview With John EckersleyDocument2 pagesEckersley Interview With John EckersleyDuncanNo ratings yet
- RevMan 5.3 User GuideDocument105 pagesRevMan 5.3 User Guidefidodido2000No ratings yet
- Sip h323 InterworkingDocument14 pagesSip h323 Interworkingniljos19No ratings yet
- The Rapture: Download PDFDocument8 pagesThe Rapture: Download PDFerwin1961No ratings yet
- Christ in MeDocument3 pagesChrist in MeDon CurtisNo ratings yet
- Cisco Aironet 1530 Series Outdoor Access Point: Sleek, Innovative, Flexible, ProvenDocument9 pagesCisco Aironet 1530 Series Outdoor Access Point: Sleek, Innovative, Flexible, Provenj_benz44No ratings yet
- Dmoi TabletDocument412 pagesDmoi Tabletbszw5eys5vNo ratings yet
- BBA 202 (EDC) - Business CommunicationDocument2 pagesBBA 202 (EDC) - Business Communicationfaheemmsd7No ratings yet
- Core DiscipleshipDocument19 pagesCore Discipleshipgemmasitchon1980No ratings yet
- Hindu TextsDocument9 pagesHindu TextsZoth BernsteinNo ratings yet
- Using Birt Report Studio IserverDocument288 pagesUsing Birt Report Studio IserverpskopjeNo ratings yet
- MorphemeDocument11 pagesMorphemebadak83No ratings yet
- 400-101 326QDocument112 pages400-101 326QOriginal Kerala SunniNo ratings yet
- 5 Сынып Директорлық БақылауDocument5 pages5 Сынып Директорлық БақылауМолдир ТазабекNo ratings yet
- Authors PurposeDocument3 pagesAuthors Purposeapi-341067643No ratings yet
- Syllabus 2. Introduction To The CourseDocument13 pagesSyllabus 2. Introduction To The CourseSkull-FaceNo ratings yet
- Comfort Sensor Using Fuzzy Logic and ArduinoDocument1 pageComfort Sensor Using Fuzzy Logic and ArduinoAlfredo MartinezNo ratings yet
- Bach Invention 04 A4Document2 pagesBach Invention 04 A4Enzo LunaNo ratings yet
- Qm4 U3 Grammar WorksheetDocument1 pageQm4 U3 Grammar WorksheetІрина ГладійNo ratings yet
- Native and Non-Native VarietyDocument23 pagesNative and Non-Native VarietyFatima MunawarNo ratings yet
- Javascript WeirdDocument209 pagesJavascript WeirdAdnan KhanNo ratings yet
- The Analysis of Compression in PoetryDocument19 pagesThe Analysis of Compression in PoetryelgianerNo ratings yet