Professional Documents
Culture Documents
Ketal Mechanism
Ketal Mechanism
Uploaded by
Kaye Simpson0 ratings0% found this document useful (0 votes)
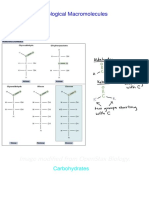
10 views1 pageThe document describes the mechanism for ketal formation. Water acts as a catalyst to facilitate the reaction between an aldehyde and an alcohol, forming an acetal or ketal. The aldehyde and alcohol combine to form a hemiacetal intermediate that then loses water to form the final ketal product, with methanol and water as byproducts.
Original Description:
Original Title
KetalMechanism
Copyright
© Attribution Non-Commercial (BY-NC)
Available Formats
DOC, PDF, TXT or read online from Scribd
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentThe document describes the mechanism for ketal formation. Water acts as a catalyst to facilitate the reaction between an aldehyde and an alcohol, forming an acetal or ketal. The aldehyde and alcohol combine to form a hemiacetal intermediate that then loses water to form the final ketal product, with methanol and water as byproducts.
Copyright:
Attribution Non-Commercial (BY-NC)
Available Formats
Download as DOC, PDF, TXT or read online from Scribd
0 ratings0% found this document useful (0 votes)
10 views1 pageKetal Mechanism
Ketal Mechanism
Uploaded by
Kaye SimpsonThe document describes the mechanism for ketal formation. Water acts as a catalyst to facilitate the reaction between an aldehyde and an alcohol, forming an acetal or ketal. The aldehyde and alcohol combine to form a hemiacetal intermediate that then loses water to form the final ketal product, with methanol and water as byproducts.
Copyright:
Attribution Non-Commercial (BY-NC)
Available Formats
Download as DOC, PDF, TXT or read online from Scribd
You are on page 1of 1
Ketal Formation Mechanism
H2O H O Cl O H HO H OCH3
H H HO O H H2O OCH3
OCH3
CH3OH H2O CH3O OCH3 H CH3O OCH3
OCH3
CH3OH
You might also like
- Exercise Naming AlkenesDocument13 pagesExercise Naming AlkenesUng Hie Huong100% (1)
- PDF Makalah Cuci Tangan - CompressDocument14 pagesPDF Makalah Cuci Tangan - CompressYosa Nanda FermataNo ratings yet
- Acetal Formation: O OCH H CO CH OH HDocument1 pageAcetal Formation: O OCH H CO CH OH Hkalli987No ratings yet
- Carbohydrates: Biochemistry IDocument59 pagesCarbohydrates: Biochemistry IanaNo ratings yet
- Galactose: CH OH CH OHDocument13 pagesGalactose: CH OH CH OHCarla DionisioNo ratings yet
- The Structure and Function of MacromoleculesDocument70 pagesThe Structure and Function of MacromoleculesAhmadMaulanaIfanAkbasNo ratings yet
- Ch05 MacromoleculesCampbellDocument69 pagesCh05 MacromoleculesCampbellCarl Jasper AquinoNo ratings yet
- Scan 26-Aug-2020 PDFDocument1 pageScan 26-Aug-2020 PDFchhaayaachitran akshuNo ratings yet
- PDF Resume Tinjauan Pemrosesan Transaksi Dan Sistem Enterprise Resource Planni DLDocument5 pagesPDF Resume Tinjauan Pemrosesan Transaksi Dan Sistem Enterprise Resource Planni DLNisa AlyaNo ratings yet
- Bio MoleculesDocument21 pagesBio MoleculesHafsa MaryamNo ratings yet
- AlcoholsDocument3 pagesAlcoholsNirav PandeyNo ratings yet
- LJ 3 Apr-4Document3 pagesLJ 3 Apr-4x23dhaneeshkNo ratings yet
- Lecture 2 Cell BiologyDocument27 pagesLecture 2 Cell Biologybobo-girl123No ratings yet
- Cells and Sugars 3-Carbohydrate Structure-StudentDocument8 pagesCells and Sugars 3-Carbohydrate Structure-StudenttyhbbhhNo ratings yet
- CH 2 Indian Eco (1950-1990)Document6 pagesCH 2 Indian Eco (1950-1990)Ronak GellaniNo ratings yet
- 1912127028bio Molecules Study Notes PDFDocument20 pages1912127028bio Molecules Study Notes PDFkarthickumaragNo ratings yet
- Wa0013.Document30 pagesWa0013.VekeMalaNo ratings yet
- Ch. BiomoleculesDocument11 pagesCh. Biomoleculesap39fp3tNo ratings yet
- Biological MacromoleculesDocument8 pagesBiological MacromoleculesRintuNo ratings yet
- ΠΡΟΠΟΝΗΣΗ ΣΤΟ ΣΧΙΝΙΑDocument2 pagesΠΡΟΠΟΝΗΣΗ ΣΤΟ ΣΧΙΝΙΑmctsouNo ratings yet
- I:', ?#T e A-: Obpa3Ai - 3A Hbhhhhthpaii H Hocjiobhhx HapthhpaDocument1 pageI:', ?#T e A-: Obpa3Ai - 3A Hbhhhhthpaii H Hocjiobhhx HapthhpaJelena SimicNo ratings yet
- CarbohydratesDocument27 pagesCarbohydrateseugieniogienioNo ratings yet
- Adobe Scan 17 Aug 2021 SapmDocument3 pagesAdobe Scan 17 Aug 2021 SapmAkash ChauhanNo ratings yet
- XVI A & XVII A Group ElementsDocument6 pagesXVI A & XVII A Group ElementsHarshvardhansingh Rajsingh DeoraNo ratings yet
- Chem ProjectDocument19 pagesChem Projectthealokkumar7No ratings yet
- Carbohydrate For International ClassDocument43 pagesCarbohydrate For International ClasseciNo ratings yet
- Digital OptimizationDocument4 pagesDigital Optimizationeesahasan786No ratings yet
- Expt1 - Nikhil Bhardwaj - 200633 - D7Document6 pagesExpt1 - Nikhil Bhardwaj - 200633 - D7Nikhil BhardwajNo ratings yet
- Organic Compounds and The Atomic Properties of CarbonDocument65 pagesOrganic Compounds and The Atomic Properties of CarbonLindsey La CroixNo ratings yet
- Acids Bases and Salts NotesDocument8 pagesAcids Bases and Salts NotesM SreehithaNo ratings yet
- M.SC I (General Microbiology) Shivaji University, KolhapurDocument14 pagesM.SC I (General Microbiology) Shivaji University, KolhapurPratibha PatilNo ratings yet
- Carbohydrate Synthesis and Study of Carbohydrate-Lectin Interactions Using QCM Biosensors and Microarray TechnologiesDocument21 pagesCarbohydrate Synthesis and Study of Carbohydrate-Lectin Interactions Using QCM Biosensors and Microarray TechnologiesEric ZulfikarNo ratings yet
- Delayen A. Heterocycle 2005, 65 - 1673Document6 pagesDelayen A. Heterocycle 2005, 65 - 1673nileshshitole619No ratings yet
- Chemistry Practical File Class 12 2023Document18 pagesChemistry Practical File Class 12 2023Shivanshu SharmaNo ratings yet
- 1.2 Carbohydrates-1Document40 pages1.2 Carbohydrates-1Hanithra AnanthanNo ratings yet
- Carbonyl Chemistry Tutorial #9 2018-2019 AnswersDocument9 pagesCarbonyl Chemistry Tutorial #9 2018-2019 AnswersZoe NorvilleNo ratings yet
- Chapter 2 - Carbohydrate MetabolismDocument72 pagesChapter 2 - Carbohydrate MetabolismPJ1-0619 Florence Binti IstemNo ratings yet
- Scan Jun 25, 2021Document4 pagesScan Jun 25, 2021Saktheeswaran VNo ratings yet
- Chapter4 Cellularmetabolism 110727073536 Phpapp02Document52 pagesChapter4 Cellularmetabolism 110727073536 Phpapp02onadandan4No ratings yet
- Requirements For Structural Determination of A Carbohydrate A B CDocument20 pagesRequirements For Structural Determination of A Carbohydrate A B CMunir AhmadNo ratings yet
- 2010 Organic Chemistry Assignment 1 AnswersDocument6 pages2010 Organic Chemistry Assignment 1 Answersvokasa4037No ratings yet
- Natural Product: AlkaloidDocument38 pagesNatural Product: AlkaloidWidya AnggrainiNo ratings yet
- Biomolecules Notes From Srikanth SirDocument22 pagesBiomolecules Notes From Srikanth SirSRI SREENo ratings yet
- Structure of MaltoseDocument2 pagesStructure of MaltoseBhavaniNo ratings yet
- Lipid Metabolism FK 2018Document58 pagesLipid Metabolism FK 2018gabriellafelisaNo ratings yet
- KarbohidratDocument75 pagesKarbohidratLolita YudistiraNo ratings yet
- Ipr 2Document13 pagesIpr 2Hindustan Karnataka (Shah)No ratings yet
- Ketone BodiesDocument25 pagesKetone Bodies2K19/BAE/104 ADITI GARGNo ratings yet
- PDF Clinical Pathway Bayi Asfiksia - CompressDocument7 pagesPDF Clinical Pathway Bayi Asfiksia - CompressRahmat RizalNo ratings yet
- Elements Found in Living Things2Document6 pagesElements Found in Living Things2Maevrick HaleyNo ratings yet
- Biomimetics Chemistry: New Field in Chemistry Emerging From The Advancement of BiotechnologyDocument18 pagesBiomimetics Chemistry: New Field in Chemistry Emerging From The Advancement of Biotechnologymalvarez153No ratings yet
- Integrated Catalytic Process For Obtaining Liquid FuelsDocument9 pagesIntegrated Catalytic Process For Obtaining Liquid FuelsClaudia Elizabeth Ruiz DávilaNo ratings yet
- Spanish Product GuideDocument107 pagesSpanish Product GuideVan Ludwing100% (1)
- General EconomyDocument12 pagesGeneral EconomysarfarazNo ratings yet
- Chapter4 Cellularmetabolism 110727073536 Phpapp02 1Document52 pagesChapter4 Cellularmetabolism 110727073536 Phpapp02 1Jinan K. DianNo ratings yet
- Electrophiles Deficient Celt: It III SpeciesDocument9 pagesElectrophiles Deficient Celt: It III SpeciesArmaan GargNo ratings yet
- Oligosaccharides. Polysaccharides. Terpenes.Document182 pagesOligosaccharides. Polysaccharides. Terpenes.Umar M abubakarNo ratings yet
- 1.2.QR - Carbohydrate Structure - UUBiochemDocument2 pages1.2.QR - Carbohydrate Structure - UUBiochemdondavis77No ratings yet
- Chromatography - HPLCDocument25 pagesChromatography - HPLCnajdat alzaatraNo ratings yet
- Cell Biology Topic 4A Carbohydrates and LipidsDocument62 pagesCell Biology Topic 4A Carbohydrates and Lipidscorleone.tamNo ratings yet