Professional Documents
Culture Documents
Peng-Robinson Equation of State
Uploaded by
ANDRES CAMILO LEYTON ALVAREZ0 ratings0% found this document useful (0 votes)
13 views2 pagesThe document summarizes the Peng-Robinson equation of state for calculating properties of a pure fluid substance, in this case Freon 12. It provides the critical temperature and pressure of Freon 12, along with other relevant properties. It then shows the calculations of the equation of state constants and coefficients based on these properties, which are used to solve the cubic equation of state and determine the volume of the fluid at different pressures and temperatures.
Original Description:
Original Title
PR-EOS V2.2 (3)
Copyright
© © All Rights Reserved
Available Formats
XLSX, PDF, TXT or read online from Scribd
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentThe document summarizes the Peng-Robinson equation of state for calculating properties of a pure fluid substance, in this case Freon 12. It provides the critical temperature and pressure of Freon 12, along with other relevant properties. It then shows the calculations of the equation of state constants and coefficients based on these properties, which are used to solve the cubic equation of state and determine the volume of the fluid at different pressures and temperatures.
Copyright:
© All Rights Reserved
Available Formats
Download as XLSX, PDF, TXT or read online from Scribd
0 ratings0% found this document useful (0 votes)
13 views2 pagesPeng-Robinson Equation of State
Uploaded by
ANDRES CAMILO LEYTON ALVAREZThe document summarizes the Peng-Robinson equation of state for calculating properties of a pure fluid substance, in this case Freon 12. It provides the critical temperature and pressure of Freon 12, along with other relevant properties. It then shows the calculations of the equation of state constants and coefficients based on these properties, which are used to solve the cubic equation of state and determine the volume of the fluid at different pressures and temperatures.
Copyright:
© All Rights Reserved
Available Formats
Download as XLSX, PDF, TXT or read online from Scribd
You are on page 1of 2
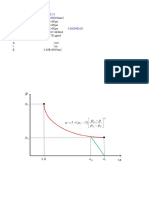
Peng-Robinson Equation of State for a Pure Fluid
Professional courtesy from G. Bolaños (School of ChE - Univalle - Colombia)
Properties Intermediate calculations
Substance Tc (K) Pc (MPa) w
Freon 12 385 4.14 0.179 Rg = 8.314
Tr = 0.9221
State Pr = 0.4995
T (K) 355 fw = 0.6421
P (MPa) 2.068
Equation-of-state constants
Three real roots (1 or 2 phase region) a= 1190078.8
Z V Fugacity b= 60.15
cm3/gmol MPa
0.704210 1005.06 1.59018 Coefficients in EOS polynomial: Z3 + A*Z2 + B
Stable phase has the
0.169293 241.62 1.79487 lower fugacity alfa = 0.2825
0.084350 120.39 1.73538 beta = 0.0421
Fugacity ratio = 0.91633
A= -0.9579
B= 0.1929
One real root (1 phase region) C= -0.0101
Z V Fugacity
cm3/gmol MPa Equation-of-state roots (Thank god Cardano d
Err:502 Err:502 Err:502 Q= 0.03764
R= -0.01356
M= -2.938E-05
Instrucciones:
Llene la información de las casillas en verde. Los resultados One real root calculation
se ven en las casillas en amarillo. S T
Para calcular presión de vapor dada la temperatura, use el SOLVER Err:502 Err:502
(Vaya al menú DATA y seleccione SOLVER), y haga que cambie
P hasta que la casilla C18 sea la unidad. Three real roots calculation
Theta Roots
0.126723 0.704210
calculations
cm3 MPa/(gmol K) Ideal gas constant
Reduced temperature
Reduced pressure
Accentric factor function
MPa cm6/gmol2
cm3/gmol
OS polynomial: Z3 + A*Z2 + B*Z + C = 0
e roots (Thank god Cardano did not die at the stake!)
If M > 0 there is one real root
If M < 0 there are three real roots
Root
Err:502
Roots
0.169293 0.084350
You might also like
- Mass Transfer Operations by Treybal Solution ManualDocument229 pagesMass Transfer Operations by Treybal Solution ManualASIM RIAZ57% (21)
- Thermal Stress in a Deformable BodyDocument11 pagesThermal Stress in a Deformable BodyKristelle GinezNo ratings yet
- Beam DesignDocument21 pagesBeam DesignKent XyrellNo ratings yet
- Zebra Current Capacity Calculation SheetDocument1 pageZebra Current Capacity Calculation SheetbinodeNo ratings yet
- Shell and tube heat exchanger designDocument32 pagesShell and tube heat exchanger designdesignselvaNo ratings yet
- Column Design CalculationsDocument307 pagesColumn Design CalculationsJuly Mar KobyashiNo ratings yet
- XII-M-1 JEE Chemistry FinalDocument312 pagesXII-M-1 JEE Chemistry FinalADITYA PRATAP100% (1)
- Industrial Flow MeasurementDocument337 pagesIndustrial Flow MeasurementRaphael Salis100% (1)
- Assignment CSTT 1Document21 pagesAssignment CSTT 1KHÁNH VÕ ĐĂNGNo ratings yet
- Zebra Current Capacity Calculation SheetDocument1 pageZebra Current Capacity Calculation Sheetbinode100% (1)
- Material properties and equations of state for concreteDocument50 pagesMaterial properties and equations of state for concreteSujin Kumar100% (1)
- Propo SED One-Store Y Timbe R Store: Analysis For PurlinsDocument10 pagesPropo SED One-Store Y Timbe R Store: Analysis For PurlinsDara MoralesNo ratings yet
- II.2 Set-2 Differential Protection For 380KV Dhahiyah Line-1 P546Document4 pagesII.2 Set-2 Differential Protection For 380KV Dhahiyah Line-1 P546Jay WinNo ratings yet
- Model Answers of FractionationDocument16 pagesModel Answers of FractionationAhmed Mohamed KhalilNo ratings yet
- 2 - Rainfall Analysis2 - Rainfall Analysis With Excel - Doc With ExcelDocument6 pages2 - Rainfall Analysis2 - Rainfall Analysis With Excel - Doc With ExcelAbu Zafor100% (1)
- A684 Cable Sizing Calculations PDFDocument1 pageA684 Cable Sizing Calculations PDFamrit100% (1)
- Learning Plan For Metallic BondingDocument6 pagesLearning Plan For Metallic BondingFrancisca Catacutan100% (1)
- Science: Quarter 3 - Module 2: The Phase ChangeDocument24 pagesScience: Quarter 3 - Module 2: The Phase ChangeJomari Palang-at Carnecer67% (3)
- Natural Bend Radius Cal Rev. C PDFDocument3 pagesNatural Bend Radius Cal Rev. C PDFhamid sobirin100% (2)
- Chemical Principles The Quest For Insight 7th Edition Atkins Solutions Manual 1Document71 pagesChemical Principles The Quest For Insight 7th Edition Atkins Solutions Manual 1jessica100% (35)
- Evaporador PDFDocument31 pagesEvaporador PDFAdriano RafaelNo ratings yet
- Plantilla TQDocument8 pagesPlantilla TQhitayosara fernandezNo ratings yet
- Peng-Robinson Equation of State For A Pure Fluid: Properties Intermediate CalculationsDocument2 pagesPeng-Robinson Equation of State For A Pure Fluid: Properties Intermediate CalculationsNICOLE DAYANA FERNANDEZ VARGASNo ratings yet
- PreosDocument15 pagesPreosRitesh Dev MaityNo ratings yet
- The Compressibility Factor Equation of StateDocument10 pagesThe Compressibility Factor Equation of StatesantanuNo ratings yet
- Chem Principles 7e ISM Focus 03 Even FINALDocument62 pagesChem Principles 7e ISM Focus 03 Even FINALSelma MeloNo ratings yet
- In Service Welding Calculation (Confidential)Document1 pageIn Service Welding Calculation (Confidential)Rudi DaNo ratings yet
- ME2112 Tutorial 1 Key StepsDocument12 pagesME2112 Tutorial 1 Key StepsdinodanoNo ratings yet
- Atzin Abigail TseDocument31 pagesAtzin Abigail TseAbi AtzinNo ratings yet
- Pr-2 Teknik Produksi I: Catur Kristiawan 124.16.021Document8 pagesPr-2 Teknik Produksi I: Catur Kristiawan 124.16.021Reynold AvgNo ratings yet
- Al-Mg intermetallic compound ratiosDocument10 pagesAl-Mg intermetallic compound ratiosdervis berk congerNo ratings yet
- Sheet Mid 000Document20 pagesSheet Mid 000kareemshdefat00No ratings yet
- Design of Suspended Two-Way Slab, S-2Document5 pagesDesign of Suspended Two-Way Slab, S-2John Harold CastroNo ratings yet
- Piezo Material Properties Data Sheet 20201112Document1 pagePiezo Material Properties Data Sheet 20201112Trương Anh DuyNo ratings yet
- Design of Slab On Grade at Basement, SOGDocument5 pagesDesign of Slab On Grade at Basement, SOGJohn Harold CastroNo ratings yet
- Example 3-10 - Smith, VNess, Abbott, Swihart - 8th Ed - Ver 2 EJERCICIO8Document3 pagesExample 3-10 - Smith, VNess, Abbott, Swihart - 8th Ed - Ver 2 EJERCICIO8Eyes in MirrorNo ratings yet
- H Is Higher Than The Initial Concentration of GNDocument3 pagesH Is Higher Than The Initial Concentration of GNEmine SöylemezNo ratings yet
- Peng-Robinson Equation of State for Calculating FugacitiesDocument2 pagesPeng-Robinson Equation of State for Calculating FugacitiesZain Ul AbedinNo ratings yet
- Cristy Nicole Buiza ChE 130 Problem Set 5A 1Document13 pagesCristy Nicole Buiza ChE 130 Problem Set 5A 1Xavier AbainzaNo ratings yet
- DC Tachometer Generator Specs and OptionsDocument3 pagesDC Tachometer Generator Specs and OptionsJUANJAUNNo ratings yet
- App4-Comp Factor ZDocument5 pagesApp4-Comp Factor ZNasrul AdliNo ratings yet
- Bottomholemodaloil GGDocument3 pagesBottomholemodaloil GGvictor javier nuñez0% (1)
- Chapter 3.7Document31 pagesChapter 3.7m-6835040No ratings yet
- 2019 Quiz 1Document2 pages2019 Quiz 1VinNo ratings yet
- Rob RCD.3Document3,258 pagesRob RCD.3RobbieNo ratings yet
- Heat Transfer CalculationsDocument4 pagesHeat Transfer CalculationsCabagnot Piolo JuliusNo ratings yet
- Thermodynamics of Multicomponent Systems SolutionsDocument5 pagesThermodynamics of Multicomponent Systems SolutionsHistorianNo ratings yet
- B X L or D A: Calculation of Earth Grid Conductor Area As Per IEEE-80 258.40 3.744 475.39 SQMMDocument1 pageB X L or D A: Calculation of Earth Grid Conductor Area As Per IEEE-80 258.40 3.744 475.39 SQMMVamsi ManojNo ratings yet
- Answer Key and Detailed SolutionsDocument13 pagesAnswer Key and Detailed SolutionsIshu PatelNo ratings yet
- DESIGN OF TWO-WAY SLAB Page16Document1 pageDESIGN OF TWO-WAY SLAB Page16Keana Lauren Lamaton AbuanNo ratings yet
- Hoja Caracteristica Trasistor 2N6059Document5 pagesHoja Caracteristica Trasistor 2N6059Alejandro G SantosNo ratings yet
- Chemical Principles The Quest For Insight 7Th Edition Atkins Solutions Manual Full Chapter PDFDocument36 pagesChemical Principles The Quest For Insight 7Th Edition Atkins Solutions Manual Full Chapter PDFpauline.wilson221100% (12)
- Junction Between The Large End of A Cone and A Cylinder Without A KnuckleDocument3 pagesJunction Between The Large End of A Cone and A Cylinder Without A KnuckleEng-CalculationsNo ratings yet
- Valve Pit (2.5X2.5)Document8 pagesValve Pit (2.5X2.5)rachanaNo ratings yet
- Extra Exercise - Water Pollution 2Document10 pagesExtra Exercise - Water Pollution 2Johnson116No ratings yet
- Problem Set 1 Ay2016-2017Document1 pageProblem Set 1 Ay2016-2017Hanna MarineNo ratings yet
- Datasheet - HK De3l20u 418701Document7 pagesDatasheet - HK De3l20u 418701cairo carNo ratings yet
- Heater (1 Juli 2023)Document145 pagesHeater (1 Juli 2023)habbibrachmanNo ratings yet
- Low Drop Power Schottky Rectifier: Main Products CharacteristicsDocument5 pagesLow Drop Power Schottky Rectifier: Main Products CharacteristicsAndres GalvezNo ratings yet
- Design of Two-Way Slab: Structural Calculation Residential BuildingDocument1 pageDesign of Two-Way Slab: Structural Calculation Residential Buildingfrancis sebastian lagamayoNo ratings yet
- Junction Between The Large End of A Cone and A Cylinder With A KnuckleDocument4 pagesJunction Between The Large End of A Cone and A Cylinder With A KnuckleEng-CalculationsNo ratings yet
- P MMHG: Constantes de Antoine Parámetros Binarios: (G - G) (Cal/Mol K) Parámetros Binarios: AlfaDocument14 pagesP MMHG: Constantes de Antoine Parámetros Binarios: (G - G) (Cal/Mol K) Parámetros Binarios: AlfaXiime WalburgNo ratings yet
- Deflection calculator beam analysisDocument5 pagesDeflection calculator beam analysisbrayanNo ratings yet
- Contact Stress Two CylindersDocument3 pagesContact Stress Two CylindersEng-CalculationsNo ratings yet
- I. Tài Liệu Thiết Kế I.1. tài liệu công trình: tc tt tc ttDocument4 pagesI. Tài Liệu Thiết Kế I.1. tài liệu công trình: tc tt tc ttNguyễn Đức HiếuNo ratings yet
- CTB-GSB 10% CBR & 150 MSA TrafficDocument7 pagesCTB-GSB 10% CBR & 150 MSA TrafficNM2104TE06 PRATHURI SUMANTHNo ratings yet
- REINFORCED CONCRETE BEAM DESIGNDocument13 pagesREINFORCED CONCRETE BEAM DESIGNderick balloganNo ratings yet
- Homework 1 - 2021-IIDocument2 pagesHomework 1 - 2021-IIANDRES CAMILO LEYTON ALVAREZNo ratings yet
- Homework 1 - 2021-IIDocument2 pagesHomework 1 - 2021-IIANDRES CAMILO LEYTON ALVAREZNo ratings yet
- New Doc 2020-02-18 13.06.53 - 20200218132326Document32 pagesNew Doc 2020-02-18 13.06.53 - 20200218132326ANDRES CAMILO LEYTON ALVAREZNo ratings yet
- International Communications in Heat and Mass Transfer: Marco PizzarelliDocument7 pagesInternational Communications in Heat and Mass Transfer: Marco PizzarelliANDRES CAMILO LEYTON ALVAREZNo ratings yet
- Taller 1Document14 pagesTaller 1ANDRES CAMILO LEYTON ALVAREZNo ratings yet
- Demostracion Capa Semiinfinita PDFDocument4 pagesDemostracion Capa Semiinfinita PDFANDRES CAMILO LEYTON ALVAREZNo ratings yet
- 8Document12 pages8ANDRES CAMILO LEYTON ALVAREZNo ratings yet
- Handouts 6 Residual PropertiesDocument17 pagesHandouts 6 Residual PropertiesAnonymous btFsuBkbwNo ratings yet
- Fluid Diff Gage Finale1Document4 pagesFluid Diff Gage Finale1Nope Not NeilNo ratings yet
- Compressor Choke or Stonewall - EnggcyclopediaDocument11 pagesCompressor Choke or Stonewall - EnggcyclopediabitterteaNo ratings yet
- Specific Heat of GasesDocument22 pagesSpecific Heat of Gasesthinkiit0% (1)
- Materials Science and Engineering Concept Check Part3 PDFDocument26 pagesMaterials Science and Engineering Concept Check Part3 PDF李宛芸No ratings yet
- Fluid Flow TypesDocument3 pagesFluid Flow TypesrijintomNo ratings yet
- Gas DensityDocument5 pagesGas DensityAdi SupriadiNo ratings yet
- E 69 CC 2 A 7093 DC 133Document10 pagesE 69 CC 2 A 7093 DC 133Ammar AlaghbariNo ratings yet
- Pneumatic Conveying DesignDocument7 pagesPneumatic Conveying DesignMolly0630No ratings yet
- P - 18.2 (Cyb - 1.4) ,.............................................................Document13 pagesP - 18.2 (Cyb - 1.4) ,.............................................................David Torres RiveraNo ratings yet
- Thin FilmsDocument68 pagesThin FilmsNaveenNo ratings yet
- 02 Mechanical Properties of Fluids One Shot (LMR Series)Document110 pages02 Mechanical Properties of Fluids One Shot (LMR Series)ashunaik409No ratings yet
- Fouling FactorsDocument6 pagesFouling FactorsDayo IdowuNo ratings yet
- Lab 6 Heat of VaporizationDocument6 pagesLab 6 Heat of Vaporizationapi-458764744No ratings yet
- Reliability of Core Analysis & PVT Data-3Document1 pageReliability of Core Analysis & PVT Data-3Aditya KurniawanNo ratings yet
- Fluid Mechanics Lecture Week 6 Chapter 2 PPT With NotesDocument40 pagesFluid Mechanics Lecture Week 6 Chapter 2 PPT With NotesJake CanlasNo ratings yet
- Acs CGD 5b00989Document39 pagesAcs CGD 5b00989प्रधुम्नमहतNo ratings yet
- Wa0001Document9 pagesWa0001Shargeesh .sNo ratings yet
- Chemical Element Atoms Crystalline MoleculesDocument3 pagesChemical Element Atoms Crystalline MoleculesReign AckermannNo ratings yet
- 1 QuestionsDocument37 pages1 QuestionsCarla Jem Delos SantosNo ratings yet
- Quantifying Effects of Supercharging When Using LWD and Final PDFDocument46 pagesQuantifying Effects of Supercharging When Using LWD and Final PDFTran Dang SangNo ratings yet
- Patent Application Publication (10) Pub. No.: US 2012/0047941 A1Document8 pagesPatent Application Publication (10) Pub. No.: US 2012/0047941 A1Jay patelNo ratings yet