Professional Documents
Culture Documents
Sameer Unias Physics Tutorials: MHT-CET 2021-22
Uploaded by
Arhaan PenwalaOriginal Title
Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
Sameer Unias Physics Tutorials: MHT-CET 2021-22
Uploaded by
Arhaan PenwalaCopyright:
Available Formats
accelerate towards success
Sameer Unias Physics Tutorials
MHT-CET 2021-22
Time : 60 Min Phy : Kinetic theory of gases and Radiation Marks : 50
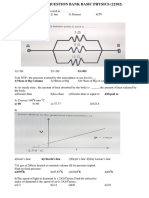
01) Figure shows two flasks connected to each C) Number of collisions per unit time will increase
other. The volume of the flask 1 is twice that of D) Collisions will not change
flask 2. The system is filled with an ideal gas at
temperature 100 K and 200 K respectively. If the 06) At constant temperature on increasing the
mass of the gas in 1 be m then what is the mass of pressure of a gas by 5% will decrease its volume by
the gas in flask 2? A) 4.26%
B) 4.76%
C) 5%
D) 5.26%
07) The mean kinetic energy of a gas at 300 K is
M 100 J. The mean energy of the gas at 450 K is
A)
8 equal to
M A) 100 J
B)
4 B) 150 J
M C) 450 J
C) D) 3000 J
2
D) m
08) If the oxygen (O2) has root mean square velocity
of C ms-1, then root mean square velocity of the
02) The value of universal gas constant is 8.3
hydrogen (H2) will be
J/mole/K, the mean kinetic energy of 32 gm of
oxygen at - 73°C will be A) C ms 1
A) 480 J 1
B) ms 1
B) 2490 J C
C) 4980 J
C) 4 C ms 1
D) The information is incomplete.
C
D) ms 1
03) A cylinder rolls without slipping down an 4
inclined plane, the number of degrees of freedom it
has, is 09) The respective speeds of five molecules are 2,
A) 1 1.5, 1.6, 1.6 and 1.2 km/s. The most probable
B) 2 speed in km/s will be
C) 3 A) 1.31
D) 5 B) 1.58
C) 1.6
04) At a given temperature, if Vrms is the root D) 2
mean square velocity of the molecules of a gas and
Vs the velocity of sound in it, then these are 10) 4 moles of an ideal gas is at 0°C. At constant
pressure it is heated to double its volume, then its
C final temperature will be
related as P
Cv A) 546°C
B) 273°C
3
A) Vrms Vs C) 0°C
D) 136.5°C
B) Vrms Vs 11) At 27°C temperature, the kinetic energy of an
3
ideal gas is E1 . If the temperature is increased to
3
C) Vrms Vs 327°C, then kinetic energy would be
1
D) Vrms Vs A) E1
2
B) 2E1
05) At constant volume, temperature is increased.
Then 1
C) E1
A) Collisions will be in straight lines 2
B) Collision on walls will be less D) 2E1
NEET/AIPMT, JEE-MAIN, MHT-CET
Sameer Unias Physics Tutorials
gas of density is proportional to
12) At which temperature the velocity of O2 A)
molecules will be equal to the velocity of N2
molecules at 0°C? B) 2
A) 39°C 1
C)
B) 40°C
C) 93°C
1
D) Cannot be calculated. D)
2
13) Two gases are at absolute temperatures 300 K
and 350 K respectively. Ratio of average kinetic 20) A box contains n molecules of a gas. How will
energy of their molecules is the pressure of the gas be affected, if the number of
A) 49 : 36 molecules is made 2 n?
B) 36 : 49 A) Pressure will become three times.
C) 7 : 6 B) Pressure will be doubled.
D) 6 : 7 C) Pressure will remain unchanged.
D) Pressure will decrease.
14) A gas at 27°C has a volume V and pressure P.
On heating its pressure is doubled and volume 21) The kinetic energy per gm mol for a diatomic
becomes three times. The resulting temperature of gas at room temperature is
the gas will be 5
A) 162°C A) RT
2
B) 600°C
3
C) 1527°C B) RT
D) 1800°C 2
1
C) RT
15) For hydrogen gas, Cp Cv a and for oxygen 2
gas, Cp Cv b . So the relation between a and b D) 3 RT
is given by 22) For an ideal gas of diatomic molecules,
A) a b 7
B) a 4b A) CP R
2
C) a 16b 5
D) b 16a B) Cp R
2
16) The specific heats at constant pressure is 3
C) Cv R
greater than that of the same gas at constant 2
volume because D) CP Cv 2R
A) the molecular vibration increases more at
constant pressure. 23) What is the ratio of specific heats of constant
B) the molecular attraction increases more at pressure and constant volume for NH3 ?
constant pressure.
A) 1.67
C) at constant volume work is done in expanding
B) 1.28
the gas.
C) 1.44
D) at constant pressure work is done in expanding
D) 1.33
the gas.
24) A vessel is filled with an ideal gas at a pressure
17) The volume of a gas at 21°C temperature and
of 10 atmospheres and temperature 27°C. Half of
768 mm pressure is 1 litre. If the density of the gas
the mass of the gas is removed from the vessel and
is 1.2 gm/litre at NTP, then its mass will be
temperature of the remaining gas is increased to
A) 1.13 gm
87°C. Then the pressure of the gas in the vessel
B) 4 gm
will be
C) 4.21 gm
A) 8 atm
D) 10 gm
B) 7 atm
C) 6 atm
18) Hydrogen gas is filled in a balloon at 20°C. If
D) 5 atm
temperature is made 40 C , pressure remaining
same, what fraction of Hydrogen will come out? 25) Which of the following statement is true?
A) 0.07 A) Given sample of 1 cc of hydrogen and 1 cc of
B) 0.25 oxygen both at NTP; oxygen sample has a large
C) 0.5 number of molecules.
D) 0.75 B) The root mean square speed of the molecules of
different ideal gases, maintained at the same
19) At a given temperature, the pressure of an ideal temperature are the same.
NEET/AIPMT, JEE-MAIN, MHT-CET
Sameer Unias Physics Tutorials
C) Two different gases at the same temperature C) 0.2%
pressure have equal root mean square velocities. D) 0.1%
D) Absolute zero degree temperature is not zero
energy temperature. 33) For ideal gas, which statement is wrong?
A) It obeys Boyle's law.
26) At a given temperature the ratio of r. m. s. B) Internal energy depends on temperature only.
velocities of hydrogen molecule and helium atom C) It follows Vander-Waal's equation.
will be D) It follows PV = RT.
A) 1 : 2
B) 2 : 1 34) In a monoatomic gas, total degree of freedom
C) 1: 2 are because of
A) rotational motion
D) 2 :1 B) translational motion
C) oscillatory motion
27) By what factor the r. m. s. velocity will change, D) vibrational motion
if the temperature is raised from 27°C to 327°C?
A) 1 35) The equation of state corresponding to 8 g of
B) 2 O2 is
C) 2 A) PV = RT/2
D) 2 2 B) PV = RT
C) PV = RT/4
R D) PV = 8RT
28) What is the value of for diatomic gas?
CP
36) The specific heat relation for ideal gas is
A) 2/7 A) CP CV R
B) 3/4
B) CP CV R
C) 3/5
D) 5/7 C) CV /CP R
D) CP /CV R
29) Gas at a pressure P0 in contained is a vessel.
If the masses of all the molecules are halved and 37) The temperature of an ideal gas at atmospheric
their speeds are doubled, the resulting pressure P pressure is 300 K and volume 1m 3. If temperature
will be equal to and volume become double, then pressure will be
P A) 4 105 N / m2
A) 0
2
B) 2 105 N / m2
B) P0
C) 105 N / m2
C) 2P0
D) 4P0 D) 0.5 105 N / m2
30) The kinetic energy of one gram molecule of a 38) A diatomic gas molecule has translational,
gas at normal temperature and pressure rotational and vibrational degrees of freedom. The
is (R 8.31 J / mole K) Cp / Cv is
A) 1.29
A) 3.4 103 J B) 1.33
B) 2.7 102 J C) 1.4
D) 1.67
C) 1.3 102 J
D) 0.56 104 J 39) The value of CV for one mole of neon gas is
7
A) R
31) A perfect gas at 27 C is heated at constant 2
pressure to 327°C. If original volume of gas at 27°C 5
B) R
is V, then volume at 327 C is 2
A) V / 2 3
C) R
B) V 2
C) 2 V 1
D) 3 V D) R
2
32) A gas at the temperature 250 K is contained in
a closed vessel. If the gas is heated through 1 K, 40) Energy of all molecules of a monoatomic gas
then the percentage increase in its pressure will be 3
having a volume V and pressure P is PV . The
A) 0.8% 2
B) 0.4% total translational kinetic energy of all molecules of
NEET/AIPMT, JEE-MAIN, MHT-CET
Sameer Unias Physics Tutorials
a diatomic gas as the same volume and pressure is whole with velocity v, then the pressure exerted by
5 the gas is
A) PV
2 1
A) (c 2 v)2
3 3
B) PV
2 1
B) (c v)2
1 3
C) PV
2 1
C) (c v)2
D) 3 PV 3
1 2
D) c
41) Mean kinetic energy per degree of freedom of 3
gas molecules is
3 47) The temperature of the hydrogen at which the
A) RT
2 average speed of its molecules is equal to that of
1 oxygen molecules at a temperature of 31°C, is
B) KT A) - 264°C
2 B) - 254°C
3 C) - 235°C
C) kT
2 D) - 216°C
D) kT
48) At a pressure of 24 105 dyne /cm2 , the
R volume of O2 is 10 litre and mass is 20 gm. The r.
42) For a gas, 0.67 . This gas is made up of
CV m. s. velocity will be
molecules which are A) 400 m/s
A) polyatomic. B) 600 m/s
B) diatomic. C) 800 m/s
C) monoatomic. D) Data is incomplete.
D) mixture of diatomic and polyatomic molecules.
49) The average translational kinetic energy of O2
43) A diatomic molecule has how many degrees of (molar mass 32) molecules at a particular
freedom? temperature is 0.048 eV. The translational kinetic
A) 3
energy of N2 (molar mass 28) molecules in eV at
B) 4
C) 5 the same temperature is
D) 6 A) 0.768
B) 0.048
44) The root mean square speed of hydrogen C) 0.003
molecules at 300 K is 1930 m/s. Then the root D) 0.0015
mean square speed of oxygen molecules at 900 K
will be 50) Two vessels having equal volume contains
A) 643 m /s molecular hydrogen at one atmosphere and helium
at two atmospheres respectively. If both samples
B) 836 m /s are at the same temperature, the mean velocity of
C) 1930 3 m /s hydrogen molecules is
1930 A) 2 times that of helium.
D) m /s B) half that of helium.
3
C) twice that of helium.
D) equal to that of helium.
45) The translational kinetic energy of gas molecule
for one mole of the gas is equal to
1
A) RT
2
2
B) KT
3
2
C) RT
3
3
D) RT
2
46) Consider a gas with density and c as the
root mean square velocity of its molecules
contained in a volume. If the system moves as
NEET/AIPMT, JEE-MAIN, MHT-CET
You might also like
- P6 Home AssignmentDocument5 pagesP6 Home AssignmentMemoona GullNo ratings yet
- Kinetic Molecular Theory of GasesDocument6 pagesKinetic Molecular Theory of GasesDanny PhantomNo ratings yet
- Thermo & KTGDocument9 pagesThermo & KTGAvik DasNo ratings yet
- E10 Questions PDFDocument7 pagesE10 Questions PDFmail2sgarg_841221144No ratings yet
- XII Physics Irfan SanjraniDocument4 pagesXII Physics Irfan Sanjranijaipal singhNo ratings yet
- CC Physical Chem Test PaperDocument4 pagesCC Physical Chem Test PaperLight MayNo ratings yet
- Ch-10, Ch-1 MCQDocument2 pagesCh-10, Ch-1 MCQanis.zamanNo ratings yet
- C2 Home AssignmentDocument2 pagesC2 Home AssignmentMemoona GullNo ratings yet
- VMCCCCCCCCCCC Sir PDFDocument7 pagesVMCCCCCCCCCCC Sir PDFTG ADIROCKNo ratings yet
- 4 M/s 6m/s 2s 0m/s 2m/s 6 2 4 2 4 2 6 2: F 30N F 2400 N - M/s M/sDocument4 pages4 M/s 6m/s 2s 0m/s 2m/s 6 2 4 2 4 2 6 2: F 30N F 2400 N - M/s M/sMiguel FotsoNo ratings yet
- Btech Model QuestionsDocument22 pagesBtech Model QuestionsAkshayKannanNo ratings yet
- Mock Test 9-02-09-2021Document21 pagesMock Test 9-02-09-2021nagendra_gm100% (1)
- 11th NEW CHEMISTRY 13-06-2021Document9 pages11th NEW CHEMISTRY 13-06-2021Rishi ParmaniNo ratings yet
- Thermal and Chemical Effects of Electric CurrentDocument2 pagesThermal and Chemical Effects of Electric CurrentOrbit MBBS PreparationNo ratings yet
- 11TH Jee Level 1Document4 pages11TH Jee Level 1sayalis1604No ratings yet
- VVIMP QUESTIONS Basic Physics (22102)Document5 pagesVVIMP QUESTIONS Basic Physics (22102)Prathamesh Nawale100% (1)
- Model QuestionDocument16 pagesModel QuestionSudhir JhaNo ratings yet
- 2022 Mock JEE Main-3 - PaperDocument22 pages2022 Mock JEE Main-3 - PaperAshish GuleriaNo ratings yet
- Exercise - I: (Only One Option Is Correct)Document9 pagesExercise - I: (Only One Option Is Correct)RishabhNo ratings yet
- Chemistry EM IMPDocument53 pagesChemistry EM IMPdharmishthakaneriya4No ratings yet
- XI Sample Papers 2023Document33 pagesXI Sample Papers 2023Bhavya JangidNo ratings yet
- Full Length Paper-2: PhysicsDocument16 pagesFull Length Paper-2: PhysicsMehbob QureshiNo ratings yet
- Thermodynamics (Assignment)Document10 pagesThermodynamics (Assignment)Mudit KNo ratings yet
- Exam 1 AnswersDocument6 pagesExam 1 AnswersAdham EmadNo ratings yet
- 09 Gaseous State EngDocument5 pages09 Gaseous State EngMr XNo ratings yet
- Nand Foundation Academy, Shegaon: Xii - A DivDocument11 pagesNand Foundation Academy, Shegaon: Xii - A DivSanket PatilNo ratings yet
- Gaseous State PDFDocument4 pagesGaseous State PDFramanji1021No ratings yet
- RFT 21Document16 pagesRFT 21Faraz KhanNo ratings yet
- UNIT 1 PHYS Kinetic Theory, Heat TransferDocument16 pagesUNIT 1 PHYS Kinetic Theory, Heat TransferOnaje OneNo ratings yet
- XI Sample Papers 2023Document33 pagesXI Sample Papers 2023Bhavya JangidNo ratings yet
- IOM Model Exam 2067-05-19Document4 pagesIOM Model Exam 2067-05-19Bibek BhattaraiNo ratings yet
- Kinetic Theory of Gases (KTG) DPPDocument5 pagesKinetic Theory of Gases (KTG) DPPDhruv Kumar TailorNo ratings yet
- Module 2 Exam Practice Exam Physics: This Is Exam Number 1. 1. 2 3 4 5 6 7 8 9 10 B C B A A A A B B ADocument79 pagesModule 2 Exam Practice Exam Physics: This Is Exam Number 1. 1. 2 3 4 5 6 7 8 9 10 B C B A A A A B B AJowiNo ratings yet
- Part - A (Physics) : Jee Main 2019 - 8 April - Morning Shift MathongoDocument39 pagesPart - A (Physics) : Jee Main 2019 - 8 April - Morning Shift MathongoAparaNo ratings yet
- Molecular SpeedsDocument6 pagesMolecular SpeedsDanny PhantomNo ratings yet
- QuestionsDocument19 pagesQuestionsVIRESH SUTARNo ratings yet
- H CH SO O H: Kinetic Theory of GasesDocument5 pagesH CH SO O H: Kinetic Theory of GasesRishabhNo ratings yet
- Question PaperDocument19 pagesQuestion PaperSaad NadafNo ratings yet
- Key Star MDCAT Crash Test # 2Document12 pagesKey Star MDCAT Crash Test # 2Ahmed SaudNo ratings yet
- KTG (Objective HCV V-2) 04.01.22Document2 pagesKTG (Objective HCV V-2) 04.01.22Quark classes kanpurNo ratings yet
- Fiitjee - Jee (Main) : Physics, Chemistry & Mathematics Phase-4Document13 pagesFiitjee - Jee (Main) : Physics, Chemistry & Mathematics Phase-4Abhinav GuptaNo ratings yet
- Physics Thermodynamics: GgliveDocument12 pagesPhysics Thermodynamics: GgliveChinmaya VastradNo ratings yet
- Exam 1Document5 pagesExam 1Adham EmadNo ratings yet
- CY2301Document11 pagesCY2301Prarabdha SharmaNo ratings yet
- Electro Chemistry: Multiple Choice QuestionsDocument140 pagesElectro Chemistry: Multiple Choice QuestionsjitendratrivediNo ratings yet
- KTG UpdatedDocument5 pagesKTG Updateddeejam123No ratings yet
- DPP3 Gaseous State BansalDocument2 pagesDPP3 Gaseous State BansalBhushanNo ratings yet
- Module 2 Exam Practice Exam Physics: This Is Exam Number 1. 1. 2 3 4 5 6 7 8 9 10 B C B A A A A B B ADocument74 pagesModule 2 Exam Practice Exam Physics: This Is Exam Number 1. 1. 2 3 4 5 6 7 8 9 10 B C B A A A A B B AAmila FernandoNo ratings yet
- Chapter 3 Atomic StructreDocument3 pagesChapter 3 Atomic StructreManahil PariNo ratings yet
- Jeee Mains 2022 Question PaperDocument46 pagesJeee Mains 2022 Question Paperkaustabh2005No ratings yet
- Half Yearly Practice Paper ChemistryDocument9 pagesHalf Yearly Practice Paper ChemistrySOHAN DASNo ratings yet
- Kinetic Theory of GasesDocument8 pagesKinetic Theory of GasesAditya SallyNo ratings yet
- Nitya Academy: PVT - LTDDocument19 pagesNitya Academy: PVT - LTDRk kashyapNo ratings yet
- Sample Paper - 1: Class 11-PhysicsDocument6 pagesSample Paper - 1: Class 11-PhysicsAkshitNo ratings yet
- AIIMS - 1995: Solved PaperDocument11 pagesAIIMS - 1995: Solved PaperipsitaNo ratings yet
- Physics Sample Paper 10 - 231221 - 091030Document19 pagesPhysics Sample Paper 10 - 231221 - 091030derexax756No ratings yet
- 1st Year ENTRY TEST PAPER MT-2Document3 pages1st Year ENTRY TEST PAPER MT-2Shahzad AslamNo ratings yet
- West Bengal State University: B.Sc./Part-I/Hons./CEMA-II/2017Document4 pagesWest Bengal State University: B.Sc./Part-I/Hons./CEMA-II/2017SwwwwwNo ratings yet
- A Modern Course in Statistical PhysicsFrom EverandA Modern Course in Statistical PhysicsRating: 3.5 out of 5 stars3.5/5 (2)
- Pipe 2Document349 pagesPipe 2Herson Fronda BucadNo ratings yet
- Ideal and Real SolutionsDocument57 pagesIdeal and Real SolutionsAhammed Hussain MadhaniNo ratings yet
- Conversion of Thermal Radiation Into Chemical EnergyDocument8 pagesConversion of Thermal Radiation Into Chemical Energymaryam afzaliNo ratings yet
- Motion Mountain Volume1Document603 pagesMotion Mountain Volume1Saleem KhanNo ratings yet
- Fluid MexDocument29 pagesFluid Mexcool kidNo ratings yet
- Gravity 1Document25 pagesGravity 1Ana Marie ValenzuelaNo ratings yet
- Phy - Dpp-Dual Nature of Radiation and MatterDocument10 pagesPhy - Dpp-Dual Nature of Radiation and MatterPIYUSH SHARMANo ratings yet
- Acceleration Calculation Description Result Unit 9.81Document12 pagesAcceleration Calculation Description Result Unit 9.81venkateswaranNo ratings yet
- Module Lesson 2 PDFDocument8 pagesModule Lesson 2 PDFAbbygail HernandezNo ratings yet
- QC 1 5Document13 pagesQC 1 5Isabel EsquijoNo ratings yet
- Ell701 Assignment1Document1 pageEll701 Assignment1Hamza RazaNo ratings yet
- MPTA B2c 2017 Sheave - Pulley Balancing Final April 2018 PDFDocument9 pagesMPTA B2c 2017 Sheave - Pulley Balancing Final April 2018 PDFJorge A VilalNo ratings yet
- Chapters 10-11 Conceptual QuestionsDocument5 pagesChapters 10-11 Conceptual QuestionsShasha Jain100% (1)
- Seader Henley 2011 Chap03Document54 pagesSeader Henley 2011 Chap03AyushaNo ratings yet
- PHY2 - Chapter22 - Electric FieldDocument39 pagesPHY2 - Chapter22 - Electric FieldChiến PhạmNo ratings yet
- Last+Minute+Revision +chemistry+ (Abhinav+Sir+ +10!03!2021)Document303 pagesLast+Minute+Revision +chemistry+ (Abhinav+Sir+ +10!03!2021)AISHA AHAMMEDNo ratings yet
- Statistical Physics Phy632-Tutorial: Sharifah Hafizah Binti Syed Ab AzizDocument26 pagesStatistical Physics Phy632-Tutorial: Sharifah Hafizah Binti Syed Ab AzizSharifahHafizahAl-jufNo ratings yet
- International Physics Olympiad IPhO 1967 2019Document1,575 pagesInternational Physics Olympiad IPhO 1967 2019SanikNo ratings yet
- Fluid Mechanics Chapter 6Document34 pagesFluid Mechanics Chapter 6Ricky Mak100% (1)
- Candid Icse Physics 10Document85 pagesCandid Icse Physics 10YoNo ratings yet
- Lectures 20 and 21: Quantum Mechanics in 3D and Central PotentialsDocument13 pagesLectures 20 and 21: Quantum Mechanics in 3D and Central PotentialsJefersonNo ratings yet
- January 2019 QP Paper 2 Edexcel Maths B IGCSEDocument32 pagesJanuary 2019 QP Paper 2 Edexcel Maths B IGCSEMinn Thant ZarniNo ratings yet
- DMX4203 - 1-3 - Bernoulli DerivationDocument9 pagesDMX4203 - 1-3 - Bernoulli DerivationG.G. PRABHATH CHINTHAKANo ratings yet
- Some Important Essays and Short Questions in PhysicsDocument2 pagesSome Important Essays and Short Questions in PhysicsSURESH BABU M PhyNo ratings yet
- 52 Graphene Photonics Plasmonics and OptoelecctronicsDocument12 pages52 Graphene Photonics Plasmonics and OptoelecctronicsHabibe DurmazNo ratings yet
- 01-LINAC ComponentsDocument11 pages01-LINAC ComponentsMohamed AbdullahNo ratings yet
- Rotational Divergent PDFDocument12 pagesRotational Divergent PDFraiz123No ratings yet
- Issn 2225-6717 выпуск 52 2021: Etkin, Valery A. Levin, Elizabetha Khmelnik, Solomon I. Kireev, Valery YuDocument180 pagesIssn 2225-6717 выпуск 52 2021: Etkin, Valery A. Levin, Elizabetha Khmelnik, Solomon I. Kireev, Valery YusolikNo ratings yet
- Engineering PhysicsDocument33 pagesEngineering PhysicsLeony PanjaitanNo ratings yet
- PHYS U1 Mock Exam 4Document16 pagesPHYS U1 Mock Exam 4David-Benjamin OgilvieNo ratings yet