Professional Documents
Culture Documents
Carryover Report
Carryover Report
Uploaded by
Riad ManamanniCopyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
Carryover Report
Carryover Report
Uploaded by
Riad ManamanniCopyright:
Available Formats
CARRYOVER REPORT
ACQUI SI TI ON I NFORMATI ON
SystemName: IClass_BSM_FTN_PDA_ELS Acquired By: Natasha
Project Name: SQT_E_BSM_FTN_PDA_ELSD Column serial Number: 017139167157 42
Sample Set Method: 080 Carryover SSM Sample Kit 1 Expiration Date: 11/23/2011 12:00:00 AM EST
Processing Method Carryover Clean PM, Carryover PM Sample Kit 1 Lot Number: W04040902
Report Method Name: Carryover RM Build Version: Empower 2 Software Build 2154 SPs Installed: Service
User Name: Lisa Busher (lbusher) Sample Set Id: 1861 Result Set Id: 1872
Sample Set Name: Carryover SSM Sample Set Start Date: 11/23/2011 4:41:06 PM EST
In the Carryover qualification, two pre-blank injections are performed to determine if the chromatograph is
cleanenough to measure carryover. Those injections are followed by three Caffeine calibrationinjections at a
concentration of (0.2ppm) whichis 0.005 percent of the Challenge injection concentration (4000 ppm).
The Challenge injection is made and is followed bythree post-blank injections.
Carryover is reported as a percent of the Challenge concentration. If the % Carryover of first post-blank injection
is < or = 0.005%the qualification passes.
Carryover Peak Results
Carryover Carryover
SampleName Peak Type %Carryover
Specification Pass Fail
1 PostBlank1 < or = 0.005% Missing PASS
Printed 6:42:59 PM US/Eastern 1/16/2012 Page: 1 of 3
CARRYOVER REPORT
ACQUI SI TI ON I NFORMATI ON
SystemName: IClass_BSM_FTN_PDA_ELS Acquired By: Natasha
Project Name: SQT_E_BSM_FTN_PDA_ELSD Column serial Number: 017139167157 42
Sample Set Method: 080 Carryover SSM Sample Kit 1 Expiration Date: 11/23/2011 12:00:00 AM EST
Processing Method Carryover Clean PM, Carryover PM Sample Kit 1 Lot Number: W04040902
Report Method Name: Carryover RM Build Version: Empower 2 Software Build 2154 SPs Installed: Service
User Name: Lisa Busher (lbusher) Sample Set Id: 1861 Result Set Id: 1872
Sample Set Name: Carryover SSM Sample Set Start Date: 11/23/2011 4:41:06 PM EST
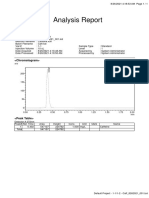
Caffeine
0.004 0.004 0.004
AU
AU
AU
Caffeine
Caffeine
0.002 0.002 0.002
0.000 0.000 0.000
0.00 0.50 1.00 0.00 0.50 1.00 0.00 0.50 1.00
Minutes Minutes Minutes
PreBlank; Vial: 2:A,1; Inj: 1 PreBlank; Vial: 2:A,1; Inj: 2 Standard; Vial: 2:A,2; Inj: 1
Caffeine
Caffeine
0.004 0.004 0.004
AU
AU
AU
0.002 0.002 0.002
0.000 0.000 0.000
0.00 0.50 1.00 0.00 0.50 1.00 0.00 0.50 1.00
Minutes Minutes Minutes
Standard; Vial: 2:A,2; Inj: 2 Standard; Vial: 2:A,2; Inj: 3 Challenge; Vial: 2:A,3; Inj: 1
0.004 0.004 0.004
AU
AU
AU
Caffeine
Caffeine
Caffeine
0.002 0.002 0.002
0.000 0.000 0.000
0.00 0.50 1.00 0.00 0.50 1.00 0.00 0.50 1.00
Minutes Minutes Minutes
PostBlank1; Vial: 2:A,4; Inj: 1 PostBlank2; Vial: 2:A,5; Inj: 1 PostBlank3; Vial: 2:A,6; Inj: 1
Printed 6:42:59 PM US/Eastern 1/16/2012 Page: 2 of 3
CARRYOVER REPORT
ACQUI SI TI ON I NFORMATI ON
SystemName: IClass_BSM_FTN_PDA_ELS Acquired By: Natasha
Project Name: SQT_E_BSM_FTN_PDA_ELSD Column serial Number: 017139167157 42
Sample Set Method: 080 Carryover SSM Sample Kit 1 Expiration Date: 11/23/2011 12:00:00 AM EST
Processing Method Carryover Clean PM, Carryover PM Sample Kit 1 Lot Number: W04040902
Report Method Name: Carryover RM Build Version: Empower 2 Software Build 2154 SPs Installed: Service
User Name: Lisa Busher (lbusher) Sample Set Id: 1861 Result Set Id: 1872
Sample Set Name: Carryover SSM Sample Set Start Date: 11/23/2011 4:41:06 PM EST
Pre-Blank Injections
SampleName Sample Type Vial Injection Name RT Area Height Peak Type %Carryover
1 PreBlank Unknown 2:A,1 1 Caffeine 0.700 Missing
2 PreBlank Unknown 2:A,1 2 Caffeine 0.700 Missing
Caffeine Standards
SampleName Sample Type Vial Injection Name RT Area Height Peak Type %Carryover
1 Standard Standard 2:A,2 1 Caffeine 0.679 5088 4424 Found 0.0050
2 Standard Standard 2:A,2 2 Caffeine 0.679 5114 4468 Found 0.0050
3 Standard Standard 2:A,2 3 Caffeine 0.679 5072 4469 Found 0.0050
Post-Blank Injections
SampleName Sample Type Vial Injection Name RT Area Height Peak Type %Carryover
1 PostBlank1 Unknown 2:A,4 1 Caffeine 0.700 Missing
2 PostBlank2 Unknown 2:A,5 1 Caffeine 0.700 Missing
3 PostBlank3 Unknown 2:A,6 1 Caffeine 0.700 Missing
Printed 6:42:59 PM US/Eastern 1/16/2012 Page: 3 of 3
You might also like
- Internet MKTDocument88 pagesInternet MKTgallcoms100% (1)
- Hacking IoT For Bug Bounties PDFDocument81 pagesHacking IoT For Bug Bounties PDFmarlon_tayagNo ratings yet
- Magazine Fit ChartDocument4 pagesMagazine Fit Chartcloudman81No ratings yet
- The Beatles Reading Comprehension. (Excellent) PDFDocument4 pagesThe Beatles Reading Comprehension. (Excellent) PDFLaura Camelo100% (1)
- Nfpa Compliance Inspection Checklist A361Document8 pagesNfpa Compliance Inspection Checklist A361gerardtrNo ratings yet
- Planner S CurveDocument23 pagesPlanner S CurveMd ShariqueNo ratings yet
- Annexure of Calibration CertificateDocument2 pagesAnnexure of Calibration Certificatevizay237_430788222100% (2)
- Green Book 7edDocument198 pagesGreen Book 7edAjith UtilityNo ratings yet
- Carryover ReportDocument3 pagesCarryover ReportRiad ManamanniNo ratings yet
- Carryover ReportDocument3 pagesCarryover ReportRiad ManamanniNo ratings yet
- CarryoverDocument3 pagesCarryoverRiad ManamanniNo ratings yet
- Carryover ReportDocument3 pagesCarryover ReportRiad ManamanniNo ratings yet
- Tabp Elisa Report: Standard TableDocument8 pagesTabp Elisa Report: Standard TableEka SaputraNo ratings yet
- Carryover RMDocument4 pagesCarryover RMRiad ManamanniNo ratings yet
- System Precision ReportDocument5 pagesSystem Precision ReportRiad ManamanniNo ratings yet
- System Precision ReportDocument5 pagesSystem Precision ReportRiad ManamanniNo ratings yet
- System Precision ReportDocument5 pagesSystem Precision ReportRiad ManamanniNo ratings yet
- LC Calibration Report: Peak: ScopoletinDocument7 pagesLC Calibration Report: Peak: Scopoletinnananana199103No ratings yet
- Diff Trip Time Characteristic:: Test Object - Device SettingsDocument4 pagesDiff Trip Time Characteristic:: Test Object - Device SettingsDuna FlaviuNo ratings yet
- Im MT 002Document5 pagesIm MT 002Emy SumartiniNo ratings yet
- UV Test InjDocument4 pagesUV Test InjRiad ManamanniNo ratings yet
- Im MT 003Document5 pagesIm MT 003Emy SumartiniNo ratings yet
- Tabp Elisa Report: Standard Table Standard (PPB)Document8 pagesTabp Elisa Report: Standard Table Standard (PPB)Eka SaputraNo ratings yet
- HARTIN TRF 1 Sef LVDocument43 pagesHARTIN TRF 1 Sef LVzita aisyahNo ratings yet
- Test Injections UV RMDocument3 pagesTest Injections UV RMRiad ManamanniNo ratings yet
- Im MT 004Document323 pagesIm MT 004Emy SumartiniNo ratings yet
- PerformanceReport-STD BHIOS 4-20200422-133121697Document3 pagesPerformanceReport-STD BHIOS 4-20200422-133121697Luis Avit Medina QuispeNo ratings yet
- Gupta Udit Assign2Document2 pagesGupta Udit Assign2Udit GuptaNo ratings yet
- Instrument CalibratiomnDocument10 pagesInstrument CalibratiomnBhaskar NapteNo ratings yet
- Diff Operating Characteristic in UNIT 2 - DIFF - CONDENSATE EXTRACTION PUMP A - 20BBB07Document6 pagesDiff Operating Characteristic in UNIT 2 - DIFF - CONDENSATE EXTRACTION PUMP A - 20BBB07MühâMméd SàhãdhNo ratings yet
- General Test Information:: Excitation Curve DataDocument2 pagesGeneral Test Information:: Excitation Curve DatajadensfadNo ratings yet
- Test Injections UV RMDocument3 pagesTest Injections UV RMRiad ManamanniNo ratings yet
- FATHOM Data Summary - AME (PVT)Document7 pagesFATHOM Data Summary - AME (PVT)Harold VergaraNo ratings yet
- Bioc2101 Principles of Biochemisrty Scientific Report AssignmentDocument1 pageBioc2101 Principles of Biochemisrty Scientific Report AssignmentHenry OkoyeNo ratings yet
- Battery (New) Run Time CalculatorDocument7 pagesBattery (New) Run Time CalculatorJose Mata RamcharanNo ratings yet
- 140T1 RoscaDocument23 pages140T1 RoscaLizandro ParedesNo ratings yet
- Flow Rate Linearity ReportDocument3 pagesFlow Rate Linearity ReportRiad ManamanniNo ratings yet
- Diff Operating Characteristic - Otf:: Test Object - Differential ParametersDocument3 pagesDiff Operating Characteristic - Otf:: Test Object - Differential ParametersDuna FlaviuNo ratings yet
- PerformanceReport KED BHIOS 4 20190207 145703952Document3 pagesPerformanceReport KED BHIOS 4 20190207 145703952luis avitNo ratings yet
- PerformanceReport-PERFORMANCE SDT BHIOS 4-20240117-085649782 Auditoria Iterna 17012024Document3 pagesPerformanceReport-PERFORMANCE SDT BHIOS 4-20240117-085649782 Auditoria Iterna 17012024luis avitNo ratings yet
- FATHOM Data Summary - PVTDocument6 pagesFATHOM Data Summary - PVTHarold VergaraNo ratings yet
- PerformanceReport STD 20240116 132131403Document3 pagesPerformanceReport STD 20240116 132131403luis avitNo ratings yet
- Test Device: Excitation CurveDocument2 pagesTest Device: Excitation Curveidhaya vickyNo ratings yet
- Diff Operating CharacteristicDocument6 pagesDiff Operating CharacteristicMühâMméd SàhãdhNo ratings yet
- Annual Protection Maintenance On 10MVA Transformer T1 at Chavuma Substation - 2022Document17 pagesAnnual Protection Maintenance On 10MVA Transformer T1 at Chavuma Substation - 2022Friday HaankumbaNo ratings yet
- Enzyme Analysis - ProteaseDocument4 pagesEnzyme Analysis - Proteasedavid.anthony010700No ratings yet
- Flow Rate Linearity ReportDocument3 pagesFlow Rate Linearity ReportRiad ManamanniNo ratings yet
- Date: 15/02/2021 Aim:: Experiment-6Document5 pagesDate: 15/02/2021 Aim:: Experiment-6Vishal ManwaniNo ratings yet
- Carry OverDocument3 pagesCarry OverAdrian MartinezNo ratings yet
- Field CBR at Drishti (R)Document1 pageField CBR at Drishti (R)Sushant MallickNo ratings yet
- Ret670 Diff Slope TestDocument9 pagesRet670 Diff Slope TestkarthikNo ratings yet
- 1-Calculator For OC Tripping Time.Document7 pages1-Calculator For OC Tripping Time.m khNo ratings yet
- +K01 FinalDocument94 pages+K01 FinalMERLYNNo ratings yet
- Test Device: Excitation CurveDocument2 pagesTest Device: Excitation CurveNagaraj VjNo ratings yet
- Nama: ENDANG TRISTAFIANI (221700550) Logistic Regression: Case Processing SummaryDocument5 pagesNama: ENDANG TRISTAFIANI (221700550) Logistic Regression: Case Processing SummaryJuLio MissaNo ratings yet
- Soal EnzymeDocument3 pagesSoal EnzymeluliNo ratings yet
- ILab 300 PDFDocument42 pagesILab 300 PDFS MNo ratings yet
- Injector CarryoverDocument3 pagesInjector CarryoverRiad ManamanniNo ratings yet
- Injector Linearity & Accuracy ReportDocument3 pagesInjector Linearity & Accuracy ReportRiad ManamanniNo ratings yet
- Default Project - 1-1 - 1-2 - Caff - 8262021 - 001Document1 pageDefault Project - 1-1 - 1-2 - Caff - 8262021 - 001Ali RizviNo ratings yet
- KR 01Document2 pagesKR 01Omkar DaveNo ratings yet
- General Test Information:: Excitation Curve DataDocument2 pagesGeneral Test Information:: Excitation Curve Datasikander843562No ratings yet
- Default Project - 1-1 - 1-6 - Caff - 8262021 - 005Document1 pageDefault Project - 1-1 - 1-6 - Caff - 8262021 - 005Ali RizviNo ratings yet
- Data Sheet of TCLP Sludge Testing Environment Laboratory of PT - Sucofindo FOR/ENV-LP/02-01Document4 pagesData Sheet of TCLP Sludge Testing Environment Laboratory of PT - Sucofindo FOR/ENV-LP/02-01Affan MahdyNo ratings yet
- Im MT 015Document5 pagesIm MT 015Emy SumartiniNo ratings yet
- System PrecisionDocument3 pagesSystem PrecisionRiad ManamanniNo ratings yet
- SQ Detector Operators GuideDocument220 pagesSQ Detector Operators GuideRiad ManamanniNo ratings yet
- Excitation Wavelength AccDocument2 pagesExcitation Wavelength AccRiad ManamanniNo ratings yet
- System IQDocument3 pagesSystem IQRiad ManamanniNo ratings yet
- Gradient RMDocument6 pagesGradient RMRiad ManamanniNo ratings yet
- Carryover RMDocument4 pagesCarryover RMRiad ManamanniNo ratings yet
- ELS Detector Response ReportDocument5 pagesELS Detector Response ReportRiad ManamanniNo ratings yet
- System Precision ReportDocument5 pagesSystem Precision ReportRiad ManamanniNo ratings yet
- Sampler Temp AccDocument1 pageSampler Temp AccRiad ManamanniNo ratings yet
- MS Detector Lin Sens RMDocument4 pagesMS Detector Lin Sens RMRiad ManamanniNo ratings yet
- Wavelength Accuracy ReportDocument3 pagesWavelength Accuracy ReportRiad ManamanniNo ratings yet
- Amounts TUVPDA2996Document4 pagesAmounts TUVPDA2996Riad ManamanniNo ratings yet
- Test Injections RMDocument3 pagesTest Injections RMRiad ManamanniNo ratings yet
- Acq SM MP FRMDocument1 pageAcq SM MP FRMRiad ManamanniNo ratings yet
- AcqQSM BSMOQDocument3 pagesAcqQSM BSMOQRiad ManamanniNo ratings yet
- UV Injection Lin Acc RMDocument3 pagesUV Injection Lin Acc RMRiad ManamanniNo ratings yet
- Test Injections UV RMDocument3 pagesTest Injections UV RMRiad ManamanniNo ratings yet
- Qualification Summary RMDocument2 pagesQualification Summary RMRiad ManamanniNo ratings yet
- Acq QSM MP FRMDocument1 pageAcq QSM MP FRMRiad ManamanniNo ratings yet
- Acq Samp Org OQDocument3 pagesAcq Samp Org OQRiad ManamanniNo ratings yet
- Compositional Accuracy ReportDocument2 pagesCompositional Accuracy ReportRiad ManamanniNo ratings yet
- Wavelength Accuracy ReportDocument3 pagesWavelength Accuracy ReportRiad ManamanniNo ratings yet
- System Precision ReportDocument5 pagesSystem Precision ReportRiad ManamanniNo ratings yet
- Gain Linearity ReportDocument3 pagesGain Linearity ReportRiad ManamanniNo ratings yet
- Acq Els MP FRMDocument1 pageAcq Els MP FRMRiad ManamanniNo ratings yet
- Detector Linearity & Sensitivity ReportDocument4 pagesDetector Linearity & Sensitivity ReportRiad ManamanniNo ratings yet
- Test Equip CalDocument2 pagesTest Equip CalRiad ManamanniNo ratings yet
- Final ApprovalDocument1 pageFinal ApprovalRiad ManamanniNo ratings yet
- Acq BSM MP FRMDocument1 pageAcq BSM MP FRMRiad ManamanniNo ratings yet
- ProcSmMpFrm 715002173rBDocument1 pageProcSmMpFrm 715002173rBRiad ManamanniNo ratings yet
- Brochure CompactLogix 5380 PDFDocument4 pagesBrochure CompactLogix 5380 PDFFederico FranchinoNo ratings yet
- Strategic Paper Guide PartsDocument7 pagesStrategic Paper Guide PartsAGOCOY Ashley S.No ratings yet
- GMED May 29 AgendaDocument1 pageGMED May 29 Agendaevalydiawati4937No ratings yet
- HTTPS:WWW - Pci.nic - In:pdf:syllabus M PharmDocument22 pagesHTTPS:WWW - Pci.nic - In:pdf:syllabus M PharmBvayNo ratings yet
- Fellowship Proposal 2018Document4 pagesFellowship Proposal 2018Sheraz Cheema100% (2)
- Third Division: Joseph Harry Walter Poole-Blunden, Petitioner, V. Union Bank of The Philippines, Respondent. DecisionDocument17 pagesThird Division: Joseph Harry Walter Poole-Blunden, Petitioner, V. Union Bank of The Philippines, Respondent. DecisionBiancaDiwaraNo ratings yet
- IBS in SarawakDocument5 pagesIBS in SarawakHani HusainiNo ratings yet
- FAA 8110-9 With InstructionsDocument4 pagesFAA 8110-9 With InstructionsSantiago Henao Naranjo100% (1)
- ThinkPadEdgeE430 - 2012 04 10Document8 pagesThinkPadEdgeE430 - 2012 04 10EchoNo ratings yet
- PWT 2007 - 2598Document8 pagesPWT 2007 - 2598Jan MinkNo ratings yet
- UK Visas & Immigration: Personal InformationDocument5 pagesUK Visas & Immigration: Personal InformationMaria SaeedNo ratings yet
- Last Mile Leap (LML) Test Series ScheduleDocument28 pagesLast Mile Leap (LML) Test Series ScheduleEinfiniteSailorNo ratings yet
- Aplicaciones Filtros de TransmisionDocument2 pagesAplicaciones Filtros de TransmisionMaritza ElizondoNo ratings yet
- Cost Sheet - Pages 16Document16 pagesCost Sheet - Pages 16omikron omNo ratings yet
- E-AP-6 - Bills Discounted and PurchasedDocument3 pagesE-AP-6 - Bills Discounted and PurchasedAung Zaw HtweNo ratings yet
- PAB - Case DigestsDocument20 pagesPAB - Case DigestsJJMONo ratings yet
- NCS TCP GuidelinesDocument32 pagesNCS TCP GuidelinesSandhya SinghNo ratings yet
- Government of Chhattisgarh Public Health Engineering DepartmentDocument67 pagesGovernment of Chhattisgarh Public Health Engineering DepartmentSanjeev BansalNo ratings yet
- Death of EmployeeDocument3 pagesDeath of Employeekashif_337No ratings yet
- Energies 15 06924 v2Document28 pagesEnergies 15 06924 v2qnhunhu266No ratings yet
- Ac MotorsDocument28 pagesAc MotorsShilpa SweetyNo ratings yet
- 10 Acknowledgement SlipDocument1 page10 Acknowledgement SlipumarsaboNo ratings yet
- A Study of Marketing Strategies of Pharmaceutical Industry in IndiaDocument12 pagesA Study of Marketing Strategies of Pharmaceutical Industry in IndiaSaurav PatelNo ratings yet